Search API
The Minnesota Health Department today confirmed that Beyfortus™ (Nirsevimab) is available to order via the Minnesota Vaccine for Children (MnVFC).
Beyfortus is the first U.S. FDA-approved extended half-life monoclonal antibody offering passive immunization to infants to prevent lower respiratory tract infections caused by the respiratory syncytial virus (RSV).
Even though Beyfortus is not a vaccine, the VFC program voted to include it in the VFC available vaccines to help ensure that all MnVFC-eligible children can access it at no cost.
Many Minnesota hospital pharmacies are enrolled in the MnVFC program because they manage vaccines for their co-located clinics.
These hospitals can provide inpatient Beyfortus as part of this program. Still, they may need to adjust their practices to include monitoring inpatient unit refrigerator temperatures and documenting MnVFC eligibility in their records.
Nationwide, Beyfortus is part of the U.S. Vaccines For Children program and will be available for the 23-24 RSV season.
The U.S. CDC says administration will start in early October 2023.
Note that babies born between April and September will receive doses early in fall 2023, in addition to babies born during the RSV season, which runs into 2024.

A company developing polysaccharide conjugate vaccines against serious bacterial threats announced today it received a grant award of up to $3 million for developing a Group B Streptococcus (GBS) vaccine.
The U.S. National Institute of Allergy and Infectious Diseases issued the grant to Omniose, whose research operations were established at BioGenerator Labs in St. Louis, MO. The company was previously known as VaxNewMo.
Timothy Cooke, CEO, commented in a press release on September 27, 2023, “GBS infections are an important health problem globally, and there is a broad consensus for a vaccine solution.”
Omniose aims to develop a GBS vaccine with the highest efficacy using a vastly simplified production process.
The Omniose platform uses synthetic biology with enzymatic rather than chemical methods to produce GBS polysaccharide conjugate vaccines.
The one-step enzymatic process occurs in a single re-engineered E. coli cell and retains 100% of sialic acid residues on the polysaccharide for each capsular polysaccharide conjugate.
Retention of sialic acid residues has been shown to be critical for eliciting optimal functional antibody responses towards certain GBS serotypes post-vaccination.
Conventional chemical methods are complex and lead to reduced levels of sialic acid residues through oxidation, which can have a negative impact on protection, says the company.
Group B Streptococcus is a leading cause of invasive bacterial infections in neonates and older adults in the U.S. It causes an estimated 90,000 infant deaths and 46,000 stillbirths worldwide, according to the U.S. CDC. There are currently no vaccines to prevent GBS.

The September KFF COVID-19 Vaccine Monitor recently asked the public about their intention to get an influenza vaccine, new respiratory syncytial virus (RSV) vaccines, and the updated COVID-19 vaccine during the 2023-2024 respiratory season.
A “tripledemic” scenario in which all three viruses peak together could impact millions of people simultaneously.
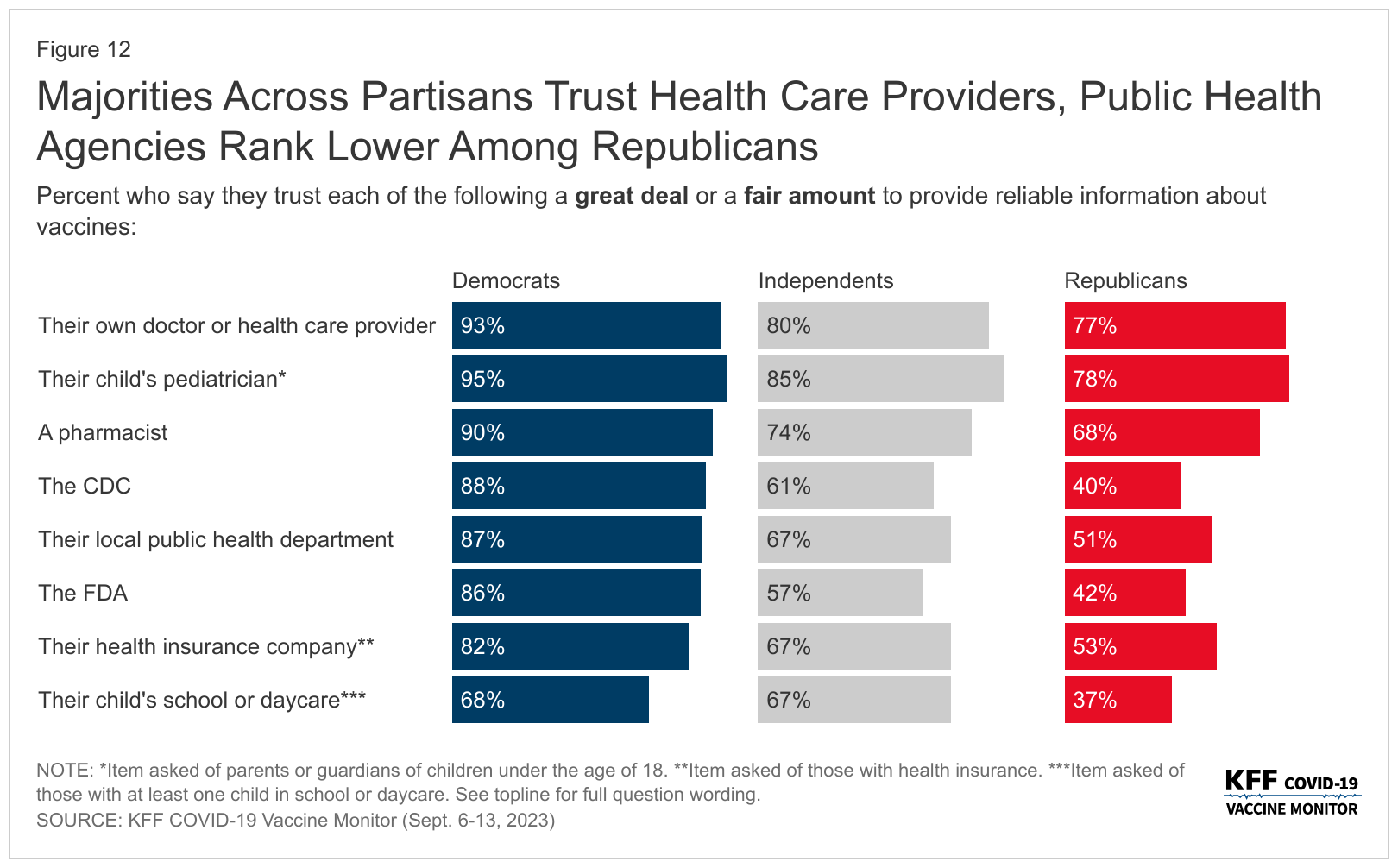
According to the KFF poll announced on September 27, 2023, most adults (58%), including three-quarters of adults 65 and older, say they will get a flu shot this year.
And about half of adults say they either will “definitely get” (23%) or “probably get” (23%) the new vaccine for COVID-19 that was approved on September 12.
In addition, 58% of adults 60 and older say they will either “definitely get” or “probably get” the new RSV vaccine recommended for their age group.
The U.S. Centers for Disease Control and Prevention (CDC) recently recommended that most people get vaccinated this year.
From a trust perspective, this poll offered good news for healthcare providers.
Healthcare providers were ranked as the most trusted sources of information about vaccines again, as most adults (68%) say they usually keep up-to-date with the vaccines their provider recommends.
Unfortunately, government sources of vaccine information like the CDC, local public health departments, and the U.S. FDA fare much lower.
The complete KFF poll results are linked here.
This article's headline was updated on Sept. 28, 2023.
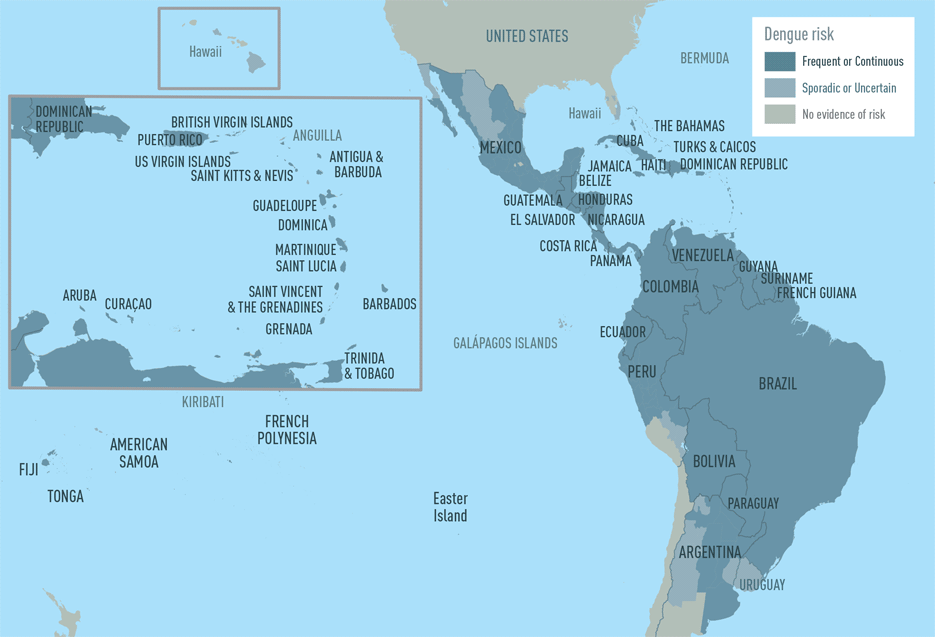
Some countries are reporting increased dengue outbreaks in Central and South America, Mexico, and the Caribbean during 2023. Dengue can become severe within a few hours. Severe dengue is a medical emergency, usually requiring hospitalization.
The U.S. Center for Disease Control and Prevention (CDC) issued a Level 1 Travel Health Notice on September 25, 2023, to alert international travelers of this mosquito-transmitted health risk.
According to the CDC, dengue is endemic in about 125 countries.
The countries listed below have recently reported higher-than-usual dengue cases, and travelers visiting these countries may be at increased risk of catching this serious disease.
Argentina
Colombia
Cuba
Guadeloupe
Guatemala
Jamaica
Martinique
Nicaragua
Panama
Peru
In the U.S., 44 jurisdictions have reported about 880 dengue cases in 2023. The majority of these dengue cases were detected in Florida and Puerto Rico.
Since October 2022, two dengue vaccines have been authorized in various countries.
A novel rabies vaccine candidate designed to produce a more robust immune response in an accelerated timespan compared to existing rabies vaccines today announced it has progressed to late-stage clinical research.
YS Biopharma Co., Ltd. confirmed on September 26, 2023, that it has enrolled the first subject in its Phase 3 clinical trial of the PIKA Rabies Vaccine candidate.
The trial will assess the safety, immunogenicity, and lot-to-lot consistency of the PIKA Rabies Vaccine and is expected to include an estimated 4,500 subjects.
Dr. Muhammad Ahmad, the Principal Investigator at Central Park Teaching Hospital in Lahore, Pakistan, where the first subject has been enrolled, commented in a press release, "This marks an important step forward in our collective efforts to develop a novel and powerful vaccine which leverages recent immunological advancements."
"We are optimistic that these results will help shape the future of vaccine interventions and aid in treating a pressing global public health issue."
This experimental vaccine may add clinical value to the currently approved rabies vaccines as sporadic breakthrough infections (i.e., rabies cases in people vaccinated) have been reported, according to The Lancet Infectious Disease in December 2022.
Rabies is a viral disease characterized by an almost 100% mortality rate upon the onset of clinical symptoms.
The virus is responsible for approximately 59,000 human fatalities annually in over 150 countries, primarily in Asia and Africa.
In the U.S., bites from bats, not dogs, are the leading source of rabies infections.
The U.S. CDC says before visiting high-risk rabies destinations, discuss vaccination options with a healthcare provider.

The World Health Organization (WHO) Director-General, Dr. Tedros Adhanom Ghebreyesus, recently launched the TB Vaccine Accelerator Council to facilitate the development, licensing, and using new Tuberculosis (TB) vaccines.
The 100-year-old Bacille Calmette-Guérin (BCG) is the only licensed TB vaccine, with over ten versions available globally.
While it provides moderate efficacy in preventing severe forms of TB in infants and young children, it does not adequately protect adolescents and adults, who account for the majority (>90%) of TB transmission globally, said the WHO on September 22, 2023.
In a press release, Dr. Tedros Adhanom Ghebreyesus commented, "Today, we have knowledge and tools they could only have dreamed of."
"The political declaration countries approved today, and the targets they have set, are a commitment to use those tools, and develop new ones, to write the final chapter in the story of TB."
The Council aims to identify innovative sustainable financing, market solutions, and partnerships across public, private, and philanthropic sectors. It will leverage various agencies to strengthen commitment and actions for novel TB vaccine development and access.
The U.S. Centers for Disease Control and Prevention (CDC) reported that TB outbreaks increased by 5% in 2022, with 60 U.S. states, the District of Columbia, and territories provisionally reporting 8,300 TB cases last year.
According to the CDC, TB is caused by Mycobacterium tuberculosis. The bacteria usually attack the lungs, but TB bacteria can attack any body part.
As a result, two TB-related conditions exist: latent TB infection and TB disease. If not treated properly, TB disease can be fatal.
In the U.S., Merck's TICE BCG vaccine has limited availability.

The U.S. Centers for Disease Control and Prevention (CDC) recently announced it had granted 13 funding awards to establish a first-of-its-kind national network, the Outbreak Analytics and Disease Modeling Network (OADMN).
The OADMN's goal is to improve speed, accuracy, and use of data & analytics during health emergencies, which is an important step towards ensuring Americans have the information they need to keep themselves and their families safe during outbreaks.
Many of these awardees are leading a consortium of collaborators to design, prototype, test, and scale up advances in data modeling tools and technology that can be used to support public health decision-makers at all levels of government.
"The collaboration with our public health, private, and academic partners over the last year to advance the science of disease forecasting and deliver decision support to leaders has been instrumental in improving outbreak response," said Dr. Dylan George, Director, Center for Forecasting and Outbreak Analytics, in a press release on September 22, 2023.
The grantees will be organized into three categories of performance, supporting three priority program actions:
- Innovators: these grantees will develop methods, tools, technologies, and other products to support emergency response.
- Integrators: these grantees will take lessons learned and techniques from Innovators and test them in small-scale deployments.
- Implementors: these grantees will take tested best practices and scale up to larger jurisdictions and partners.
The funding 13 recipients comprising OADMN in September 2023 include:
Emory University
Northeastern University
The University of North Carolina at Chapel Hill
Kaiser Permanente Southern California
Carnegie Mellon University
University of Michigan School of Public Health
University of California, San Diego
University of Minnesota
Clemson University
University of Utah
International Responder Systems
The University of Texas at Austin
The Johns Hopkins University
Note: The OADMN effort is in addition to the U.S. government's Disease X initiative.