Search API
The Philadelphia Department of Public Health reported today that there is a cluster of measles among unvaccinated residents.
As of January 4, 2024, in addition to an index case, three locally acquired cases have been confirmed, with two of those cases being hospitalized and released.
Furthermore, there are at least two more possible cases that have been hospitalized.
If you visited any of these sites on the days listed at this link, you may have been exposed to measles.
These measles cases are connected with the notice issued on December 23, 2023.
Health Commissioner Dr. Cheryl Bettigole commented in a press release, “Unfortunately, we are seeing cases of measles that have spread to vulnerable individuals, including young children, due to people declining vaccination and also failing to adhere to quarantine recommendations."
The Health Department is strongly recommending that anyone who may have been exposed to measles should quarantine themselves by staying home and away from others.
Additionally, people who have not received both doses of a Measles, Mumps, and Rubella (MMR) vaccine should talk with their healthcare provider about getting caught up.

AstraZeneca and Sanofi recently announced China's National Medical Products Administration approved Beyfortus™ (Nirsevimab-alip) for the prevention of respiratory syncytial virus (RSV) lower respiratory tract infection (LRTI).
Beyfortus is the first approved single-dose (50mg) preventive option to protect infants born healthy at term, preterm, or with specific health conditions that make them vulnerable to severe RSV disease entering or during their first RSV season.
Beyfortus is anticipated to be available in China during the upcoming 2024-2025 RSV season. Its administration can be timed to the start of the local RSV season.
RSV is the most common cause of LRTD in infants, including bronchiolitis and pneumonia, and is also a leading cause of hospitalisation in all infants. China ranks among countries with a high prevalence of RSV infections.
Iskra Reic, Executive Vice President, Vaccines and Immune Therapies, AstraZeneca, commented in a press release on January 2, 2024, "Beyfortus represents the first opportunity to prevent serious respiratory disease due to RSV for all infants in China."
"We look forward to making Beyfortus available for the 2024/25 season."
While Beyfortus was approved in the United States in 2023, its availability has been limited. As of December 14, 2023, the manufacturers committed 1.4 million doses for delivery in the U.S. before February 2024.
Canada, the European Union, and the United Kingdom have also approved Beyfortus.

Pfizer Canada announced the Health Canada approved its bivalent Respiratory Syncytial Virus (RSV) vaccine ABRYSVO™ Vaccine.
ABRYSVO is indicated for the prevention of lower respiratory tract disease caused by RSV in individuals 60 years of age and older by active immunization, and the prevention of lower respiratory tract disease and severe lower respiratory tract disease caused by RSV in infants from birth through 6 months of age by active immunization of pregnant women.
"Vaccines are considered to be the most effective tool in public health for preventing illness and can help reduce the stress on our healthcare system and professionals, including nurses, doctors, and others on the frontline," said Dr. Darine El-Chaâr, Maternal Fetal Medicine physician at The Ottawa Hospital, in a press release on January 4, 2024.
"Vaccines administered through maternal immunization can also help play a critical role in decreasing the gap of vulnerability in the first few months of an infant's life, as well as helping to protect their mothers, who may be at increased risk of severe disease compared with non-pregnant women."
RSV is a common respiratory virus that causes mild disease with cold-like symptoms in the fall and winter months.
In more vulnerable populations, like adults 60 and over, an RSV infection can be severe, especially among those with underlying respiratory or cardiac conditions.
For infants, RSV infection can result in respiratory distress, especially in those less than six months of age and those with higher risk factors such as congenital heart or lung disease or prematurity.
Pfizer Canada is currently assessing the availability timeline for ABRYSVO and is committed to bringing a supply of this vaccine to Canadians as quickly as possible.
As of January 5, 2024, RSV vaccines and monoclonal antibody therapy that deliver passive immunization to infants are approved for use in the United States.

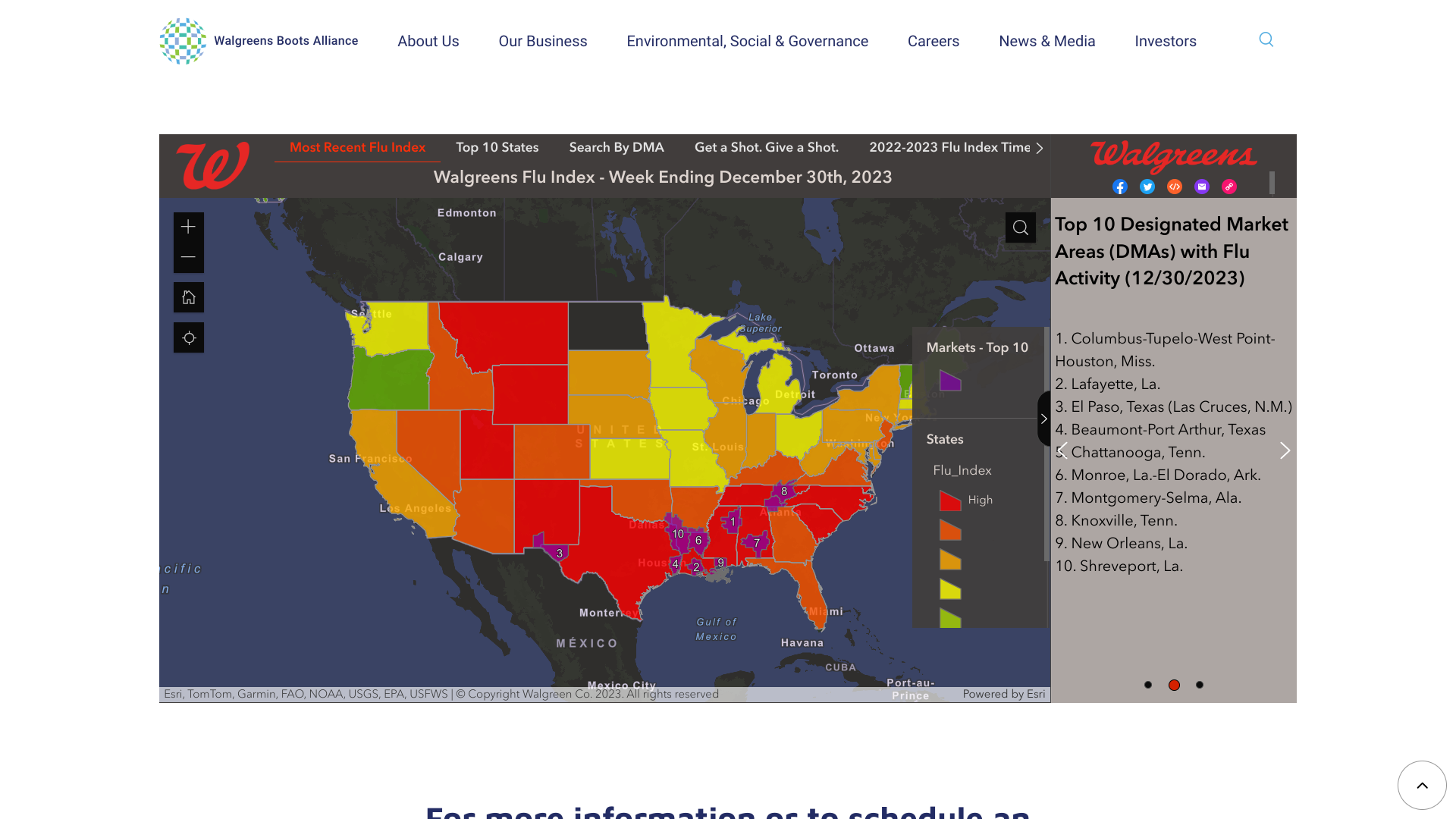
The Walgreens Flu Index® recently rendered updated information regarding influenza activity compiled using retail prescription data for antiviral medications used to treat influenza across Walgreens pharmacy locations.
As of December 30, 2023, the Index identified the top ten cities impacted by the flu last week:
- Columbus-Tupelo-West Point-Houston, Miss.
- Lafayette, La.
- El Paso, Texas (Las Cruces, N.M.)
- Beaumont-Port Arthur, Texas
- Chattanooga, Tenn.
- Monroe, La.-El Dorado, Ark.
- Montgomery-Selma, Ala.
- Knoxville, Tenn.
- New Orleans, La.
- Shreveport, La.
Previously, the U.S. CDC reported seasonal influenza activity was elevated in most parts of the country.
As of December 29, 2023, outpatient respiratory illness was above baseline for the eighth consecutive week and in all 10 HHS Regions.
From a prevention perspective, the CDC encourages most people to get an annual flu shot.
And for those who catch the flu, prescription antiviral drugs that treat influenza are available at most pharmacies in the U.S.
Sanofi Canada recently announced the National Advisory Committee on Immunization's annual Statement on Seasonal Influenza Vaccine for 2024-2025 continues to recommend Fluzone® High-Dose Quadrivalent among the preferential influenza vaccines to protect adults 65 years of age and older against influenza.
Delphine Lansac, General Manager, Vaccines Canada, Sanofi, commented in a press release on January 3, 2023, "This long-standing recommendation as a preferential influenza vaccine for adults 65 years of age and older speaks to the science that Fluzone® High-Dose Quadrivalent is built on and demonstrates our continued leadership in vaccines."
"It's been a groundbreaking year, where over 50% – more than one in two – seniors who received influenza vaccines in Canada were immunized with Fluzone® High-Dose Quadrivalent Influenza Vaccine."
Fluzone® High-Dose Quadrivalent Influenza Vaccine is designed to protect seniors with four times the active component dosage of a standard flu shot.
To date, Fluzone® High-Dose is available in more than 20 countries, including the United States, Canada, and Germany.
As of December 23, 2023, over 155 million flu shots had been distributed in the U.S. this season.

Invivyd, Inc. today announced that it has requested Emergency Use Authorization (EUA) from the U.S. Food and Drug Administration (FDA) for VYD222, a broadly neutralizing, half-life extended monoclonal antibody (mAb) candidate, for the pre-exposure prevention of COVID-19 in immunocompromised adults and adolescents.
This is essential news since about 9 million people in the U.S. may not adequately respond to current COVID-19 vaccines.
Initially authorized in late 2020, there are no mAbs currently authorized by the FDA targeting COVID-19. mAb treatments are effective when they block the SARS-CoV-2 beta coronavirus that causes COVID-19 from entering cells in the human body.
"We are tremendously pleased by the fact that VYD222 continues to demonstrate in vitro neutralization activity against the latest dominant variant, JN.1, as well as other prevalent SARS-CoV-2 strains," said Dave Hering, Chief Executive Officer of Invivyd, in a press release on January 3, 2023.
"We believe that the demonstrated durability of VYD222 is reflective of our strategy to select antibody candidates that target conserved epitopes to achieve our stated goal of keeping pace with viral evolution."
The EUA submission was based on positive initial results from the CANOPY Phase 3 pivotal clinical trial of VYD222 and ongoing in vitro neutralization activity against relevant variants such as JN.1.
Previous COVID-19 mAb news is posted by Precision Vaccinations.

ImmunityBio, Inc. announced today a recent capital raise that provides significant financial resources to accelerate the Company's commercialization efforts and expand its pipeline within the broader urological cancer space.
The Company's pipeline is based on broad immunotherapy and cell therapy platforms designed to attack cancer and infectious pathogens by activating the innate and adaptive branches of the immune system in an orchestrated manner.
On January 2, 2023, the Company confirmed up to $320 million royalty financing and equity investment by Oberland Capital, with $210 million of gross proceeds received at closing on December 29, 2023.
The proceeds will also fund ongoing business operations and clinical trials expanding Anktiva® (N-803) indications into multiple solid tumors.
ImmunityBio's commercialization efforts are in anticipation of potential U.S. Food and Drug Administration ("FDA") approval of Anktiva in combination with Merck's Bacillus Calmette-Guérin (BCG) vaccine for the treatment of patients with BCG-unresponsive non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ with or without Ta or T1 disease.
The Company announced on March 26, 2023, that the FDA had set a user fee goal date for Anktiva's Biologics License Application resubmission of April 23, 2024.
Anktiva® (N-803) is an investigational therapy, and no safety or efficacy has been established by any Health Authority or Agency, including the FDA.
"This transaction raises significant capital for the Company to support important growth plans, yet with limited equity dilution and with a cap on total payments tied to the initial investment," said Richard Adcock, Chief Executive Officer and President of ImmunityBio, in a press release.
"Besides providing a capital source at a key inflection point for ImmunityBio, this investment demonstrates strong confidence by Oberland Capital in our future, and in particular in the potential value of Anktiva in bladder cancer, as well as the direction of our clinical pipeline."
In aggregate, $850 million in capital was raised in 2023, with $320 million from institutional investors and $530 million from its founder, Patrick Soon-Shiong, M.D.
The investment from Oberland Capital takes the form of a $300 million Revenue Interest Purchase Agreement ("RIPA") that is non-dilutive to current investors, of which $200 million was funded at closing, and $100 million is to be funded contingent upon FDA approval of the Company's BLA for Anktiva in combination with BCG for NMIBC, and subject to other terms and conditions as outlined in the RIPA.
Under the terms of the RIPA, Oberland Capital will have a right to receive initially tiered single-digit royalty payments on net sales of the Company's products, which are capped at a multiple of their investment. In addition, the Company has entered into a purchase agreement with Oberland Capital for the private placement of 2,432,894 shares issued at closing, representing $10 million of gross proceeds based on the trailing 30-trading days VWAP. Oberland Capital can also purchase an additional $10 million of common stock at a future date.
In connection with the RIPA, the Company and Nant Capital entered into amendments to extend the maturity dates of certain existing promissory notes with an aggregate principal amount of approximately $505 million from December 31, 2024, to December 31, 2025, and to allow Nant Capital to convert up to an aggregate of $380 million of principal, plus accrued and unpaid interest, into shares of common stock at a price per share equal to a 75% premium to the closing market price on January 3, 2024. Nant Capital and the RIPA Purchaser Agent also concurrently entered into a Subordination Agreement, pursuant to which the Notes were subordinated to the Company's obligaCompany'sthe Purchasers under the RIPA.

The journal Vaccine recently published a manuscript entitled "Thermostable bivalent filovirus vaccine protects against severe and lethal Sudan ebolavirus and marburgvirus infection."
This publication describes the preclinical efficacy of a novel, single-vial, bivalent thermostabilized vaccine providing 100% protection in the most rigorous non-human primate challenge models against Sudan ebolavirus (SUDV) and Marburg marburgvirus (MARV) infections.
Recent outbreaks have occurred in Africa, with increased frequency in 2023.
There are currently no approved vaccines or therapeutics for either SUDV or MARV infections.
However, vaccines are available for Zaire ebolavirus (EBOV) infections in 2024, but they provide no protection against SUDV or MARV infection.
"Filoviruses such as EBOV, SUDV, and MARV are some of the most lethal viruses known, and they are endemic in areas of the world where the power supply and distribution network can be uncertain, says the World Health Organization.
A thermostabilized vaccine in a single vial format would significantly enhance any public health response to a new outbreak, at its source," stated Axel Lehrer, Ph.D., Associate Professor, Department of Tropical Medicine, Medical Microbiology and Pharmacology, University of Hawaiʻi at Mānoa, in a press release.
"Our work to date has demonstrated the feasibility of rapid and efficient manufacturing, as well as the ability to thermostabilize multiple antigens that can then be stored for extended times at temperatures exceeding 100 degrees Fahrenheit."
"The use of a bivalent vaccine has the potential to both prevent future infections with these pathogens and potentially mitigate future outbreak events, potentially using an accelerated dosing regimen."
The thermostabilized filovirus vaccine program continues to advance with the support of a National Institute of Health grant and a Small Business Innovation Research grant awarded to Soligenix, Inc.