Search API
In the past two years, H5 influenza virus subtypes have caused severe disease in birds and mammals in the United States. Because of various media reports, there is concern about the risk of these viruses spreading to humans, which could generate another pandemic.
On June 27, 2024, the U.S. Centers for Disease Control and Prevention (CDC) confirmed that the risk to people is very low. However, the U.S. government continues its multi-year effort to prepare for this risk.
Moderna, Inc. today announced a project award of $176 million to accelerate the development of mRNA-based pandemic influenza vaccines. The award program is within the U.S. Department of Health and Human Services (HHS).
The project award will support the late-stage development of an mRNA-based vaccine to enable the licensure of a pre-pandemic vaccine against the H5 influenza virus.
This new HHS agreement also includes additional options to prepare and accelerate responses to future public health threats.
"mRNA vaccine technology offers advantages in efficacy, speed of development, and production scalability and reliability in addressing infectious disease outbreaks, as demonstrated during the COVID-19 pandemic," said Stéphane Bancel, Chief Executive Officer of Moderna, in a press release on July 2, 2024.
In July 2023, Moderna initiated a Phase 1/2 study to generate safety and immunogenicity data for the investigational pandemic influenza vaccine (mRNA-1018) in healthy adults. The study includes vaccine candidates against H5 and H7 avian influenza viruses.
Results from the study are expected in 2024 and will inform Phase 3 development plans.
Currently, there are U.S. FDA-approved pandemic vaccines (Audenz) and various development initiatives underway, funded by the U.S. government.
Furthermore, the CDC has confirmed annual flu shots may not be effective against these influenza subtypes.

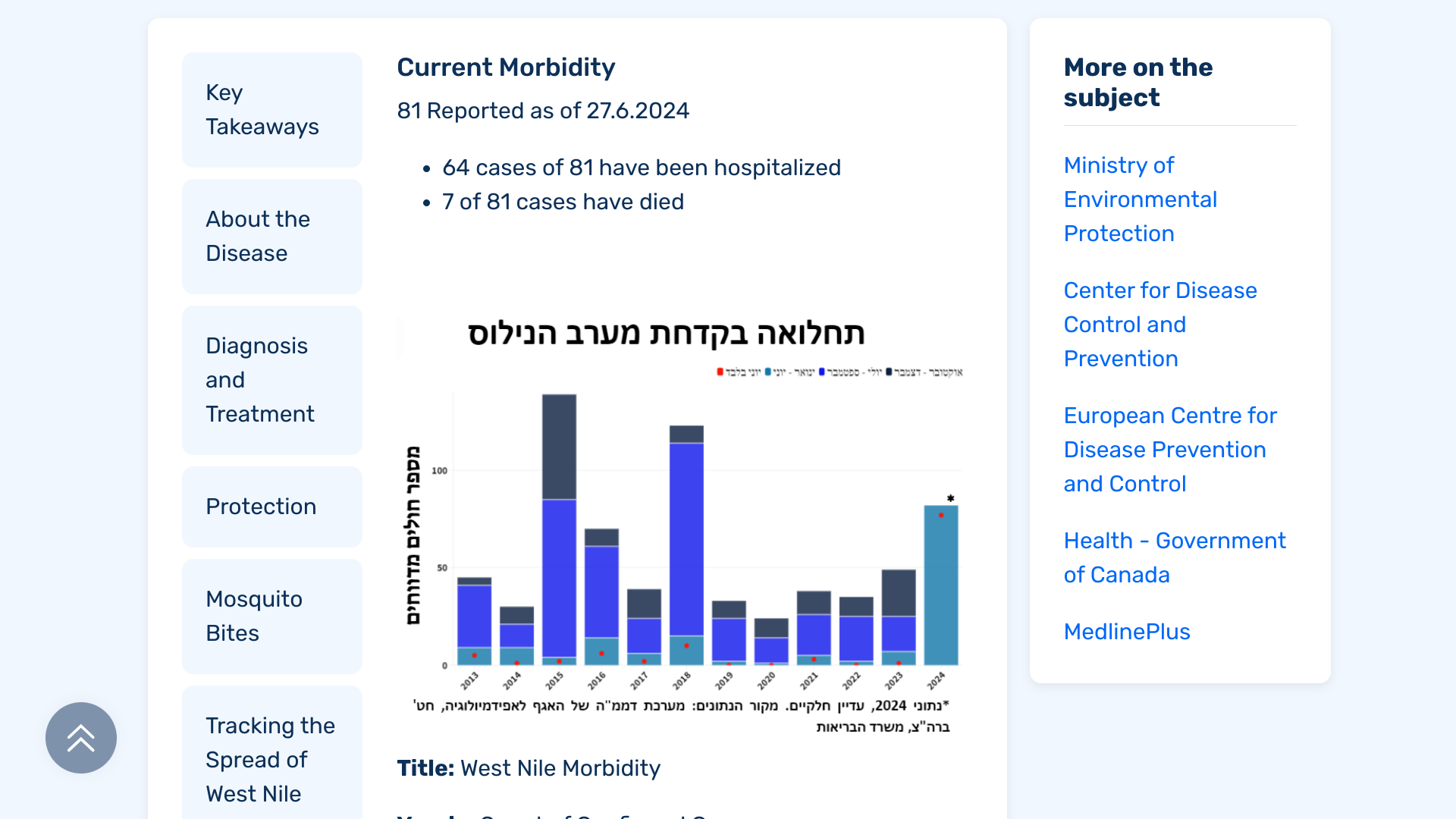
West Nile fever cases have been reported in Israel for many years and generally occur between June and November each year. That annual trend changed in 2024, with fatal cases reported earlier than usual.
The Israeli Ministry of Health (MOH) recently reported that 81 people have been diagnosed with West Nile fever in 2024. Sixty-four people have been hospitalized.
As of July 1, 2024, a total of 7 people who were diagnosed with the virus died.
All of these West Nile cases have been reported in the center of Israel.
The MOH says the risk of significant morbidity is among older adults and people with immunosuppression.
To reduce exposure to mosquitoes, the MOH recommends using mosquito repellents and appropriate measures to keep mosquitoes away in living rooms, as well as turning on fans in the place of residence.
The U.S. CDC says there are no licensed vaccines or medicines to prevent or treat West Nile virus in humans. However, several vaccine candidates have shown safety and immunogenicity in clinical trials.
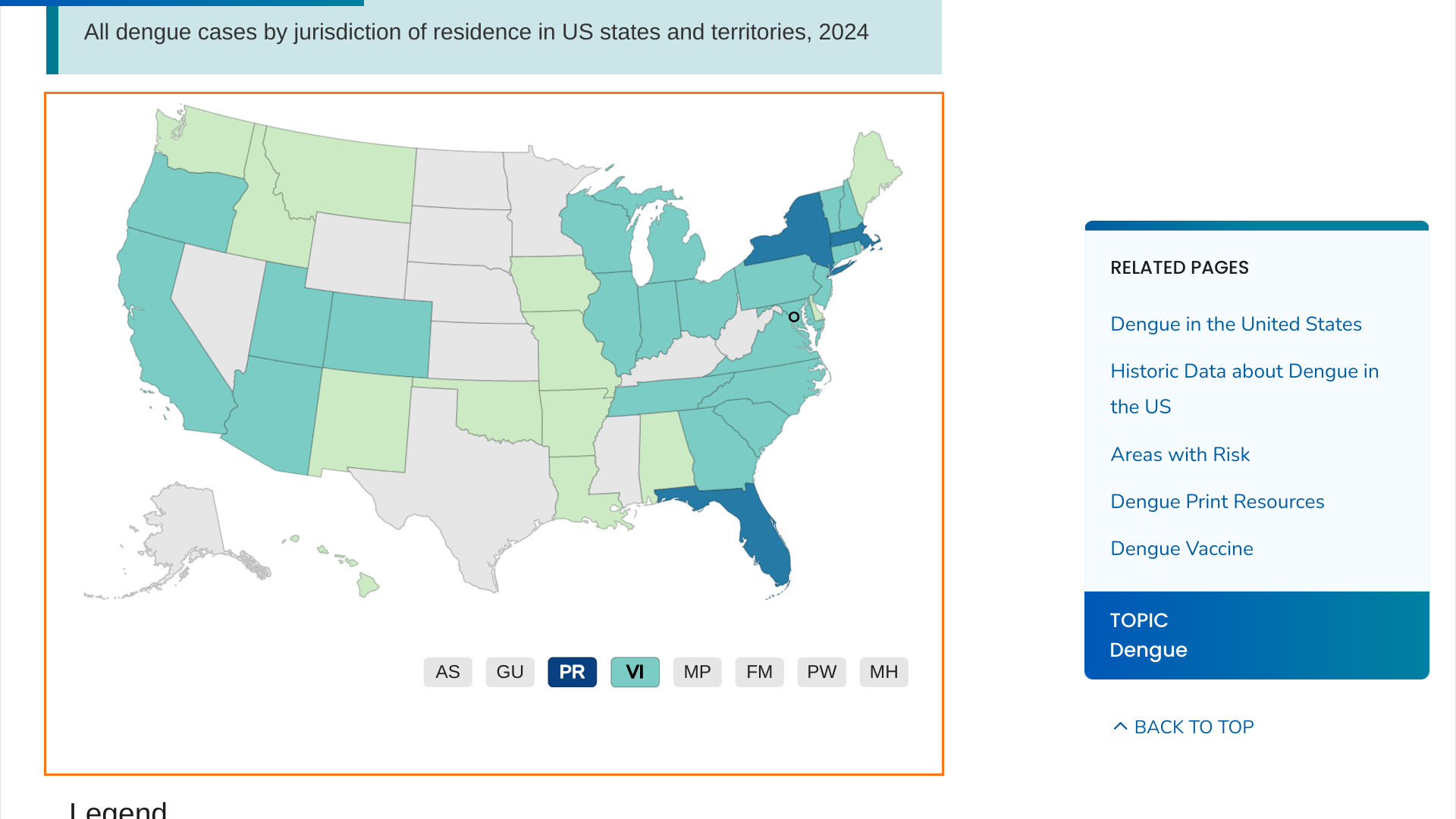
When the U.S. Centers for Disease Control and Prevention (CDC) published a Health Advisory (CDCHAN-00511) on June 25, 2024, it indicated that dengue fever posed a health risk in southeast Florida, New York, and Puerto Rico.
However, according to new CDC data, Massachusetts has reported 50 dengue cases this year.
While the CDC did not disclose whether these dengue cases were locally acquired, it can be assumed that they are travel-related since the mosquitoes that spread dengue are not found that far north in the United States.
However, countries in the Region of the Americas have reported a record-breaking number of dengue cases, exceeding the highest number ever recorded in a single year.
From a local guidance perspective, the Massachusetts Health Department (MDH) says there is no vaccine (Dengvaxia is no longer available in the U.S.); the best way to protect yourself is to not get bitten by mosquitoes.
MDH's website states if you have recently traveled to a region where mosquito-borne diseases are common and have any related symptoms, you should call your healthcare provider immediately and explain your travel history and symptoms.
Valneva SE announced today that the European Commission (EC) has granted marketing authorization in Europe for the IXCHIQ® vaccine, which is used to prevent diseases caused by the chikungunya virus in adults.
IXCHIQ® is the world’s only licensed chikungunya vaccine.
The EC decision marks the third approval the Company has received for IXCHIQ® following approval from the U.S. FDA in late 2023 and Health Canada last month.
On July 1, 2024, Valneva stated it expects to deliver the first vaccine doses in Europe in the fourth quarter of 2024.
Juan Carlos Jaramillo, M.D., Chief Medical Officer of Valneva, commented in a press release, “The EC approval marks a crucial milestone toward making this vaccine available to as many European citizens as possible .... It is critical to provide a vaccine solution not only to European travelers going to endemic chikungunya areas, such as South America or Africa.
Valneva has also submitted a Marketing Authorization Application to the UK Medicines and Healthcare products Regulatory Agency and the Brazilian Health Regulatory Agency, with potential approval in 2024.
Moderna, Inc. today announced that the European Medicines Agency's (EMA) Committee for Medicinal Products for Human Use (CHMP) has adopted a positive opinion recommending the granting of marketing authorization in the European Union (EU) for mRESVIA® (mRNA-1345), an mRNA respiratory syncytial virus (RSV) vaccine, to protect older adults.
Following the CHMP's positive opinion, the European Commission will decide on the authorization of mRESVIA.
In the EU, RSV is estimated to cause approximately 160,000 hospital admissions in adults each year.
"The positive opinion from the EMA CHMP for mRESVIA highlights the innovation and adaptability of our mRNA platform," said Stéphane Bancel, Chief Executive Officer of Moderna, in a press release on June 28, 2024. "mRESVIA safeguards older adults against severe RSV outcomes and is uniquely offered in a pre-filled syringe to enhance ease of administration...."
In May 2024, the U.S. Food and Drug Administration (FDA) approved mRESVIA, which uses the same lipid nanoparticles as the Moderna COVID-19 vaccines.
As of June 30, 2024, the FDA has approved three vaccines and one monoclonal antibody (Beyfortus) to prevent RSV in people.
As of May 22, 2024, the CDC's RSVVaxView reported that the overall RSV vaccination rate among pregnant women was about 17.8%, and an estimated 24.4% of adults 60 years and older reported receiving an RSV vaccine.

Since the start of 2024, the Democratic Republic of Congo (DRC) has reported over 20,000 mpox cases, with more than 1,000 deaths, primarily affecting children.
In June 2024, the U.S. CDC issued a Level 2 Alert reporting a mpox outbreak in 25 out of 26 DTC provinces, including urban areas.
According to media sources, authorities in the DRC have responded to this outbreak by approving the use of two new vaccines.
AfricaNews.com reported on June 28, 2024, that emergency use authorization had been issued for the Jynneos® vaccine, developed by Bavarian Nordic, and LC16, produced by KM Biologics.
LC16 is a 3rd generation, live attenuated vaccine containing live vaccinia virus (LC16m8 strain) used to prevent smallpox and mpox.
The DRC decision follows rigorous evaluation by relevant authorities and stakeholders involved in the authorization process.
JYNNEOS (MVA-BN®, IMVAMUNE®) is a two-dose vaccine based on a live, attenuated vaccinia virus, Modified Vaccinia Ankara, and has been offered in the United States since May 2022.

Merck announced today that the U.S. CDC’s Advisory Committee on Immunization Practices (ACIP) unanimously voted to recommend CAPVAXIVE™ (Pneumococcal 21-valent Conjugate Vaccine) as an option for adults 65 years of age and older for pneumococcal vaccination.
Additionally, shared clinical decision-making is recommended regarding using a supplemental dose of CAPVAXIVE for adults 65 and older who have completed their vaccine series with both PCV13 and PPSV23.
“CAPVAXIVE represents an innovative approach to invasive pneumococcal disease prevention in adults, as it is specifically designed to help protect against the strains that cause the majority of severe disease in adults 65 years of age and older,” said Dr. Eliav Barr, senior vice president, Merck Research Laboratories, in a press release on June 27, 2024.
“The ACIP vote recognizes the clinical profile of CAPVAXIVE for adults in the U.S., and we look forward to the CDC’s final, published recommendations.”

PharmaJet® today announced that their Tropis® Intradermal (ID) Needle-free System will be used in a house-to-house polio immunization campaign.
Over a quarter million PharmaJet’s needle-free intradermal syringes have been provided to support this initiative.
The campaign will be conducted in two rounds to reduce the immunity gap significantly against type-2 poliovirus. Young children will receive the needle-free polio vaccine and novel oral polio vaccine (nOPV2) to achieve 95% coverage in each round.
The polio campaign, a collaboration of the African Field Epidemiology Network, WHO, UNICEF, BMGF, GAVI, and U.S. CDC, targets over 170,000 children in Somalia.
The most recent evidence for human circulating vaccine-derived polio virus-2 was in March 2024.
Through the Somalia Emergency Action Plan, the country will continue to work with humanitarian partners to reach about 1.5 million zero-dose children, most of whom live in the country’s highly populated central and southern areas.
Paul LaBarre, Vice President of Global Business Development at PharmaJet, commented in a press release on June 27, 2024, “In Somalia, we are eager to build on previous house-to-house campaign experience that demonstrates how needle-free enables vaccination teams to move quickly and achieve high coverage without the burden of sharps waste management and with reduced vaccine volume and cold chain logistics.”
The U.S. CDC reissued a Global Polio Alert on May 23, 2024, regarding polio outbreaks and poliovirus detections in 34 countries. The CDC recommends that visitors to these countries be fully vaccinated against polio.