Search API
GSK plc today announced that the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended expanding the approval of GSK’s respiratory syncytial virus (RSV) vaccine Arexvy for the prevention of lower respiratory tract disease (LRTD) caused by RSV from adults aged 60 and above to include adults aged 50-59 years at increased risk for RSV disease.
GSK said in a press release on July 29, 2024, that today’s positive opinion is the first time that CHMP has recommended an indication for adults aged 50-59 for an RSV vaccine. The European Commission’s final decision is expected by September 2024.
The U.S. FDA approved Arexvy for adults aged 50-59 at increased risk of RSV in June 2024.
Since June 2023, Arexvy has been approved in Europe for adults aged 60 and over to prevent RSV-LRTD.
GSK has also filed regulatory submissions to expand the use of its RSV vaccine to adults aged 50-59 at increased risk in Japan and other geographies, with regulatory decisions undergoing review.
Trials evaluating the immunogenicity and safety of the vaccine in adults aged 18-49 at increased risk due to certain underlying medical conditions and in immunocompromised adults aged 18 and over are expected to read out in H2 2024.
Adults with underlying medical conditions, such as chronic obstructive pulmonary disease, asthma, heart failure, and diabetes, are at increased risk for severe consequences from an RSV infection. RSV can exacerbate these conditions and lead to pneumonia, hospitalization, or death.
In the United States, three approved RSV vaccines are available in July 2024.
Versatope Therapeutics Incorporated announced today it has received a Phase 2 Small Business Innovation Research (SBIR) grant for up to $3 million over three years from the U.S. NIH's National Institute of Allergy and Infectious Diseases.
On July 29, 2024, Versatope confirmed it will use the grant (#R44AI181242) to develop a bi-specific malaria vaccine using a target that blocks the initial malaria infection and transmission.
The Company says the novel, dual-acting vaccine may offer a more robust approach than the current World Health Organization (WHO) certified single-acting malaria vaccines.
Versatope was also awarded a Stage I grant from the MassVentures SBIR Targeted Technologies program.
"We appreciate the recognition and support of the NIH and MassVentures team to advance the development of Versatope's technology platform and to help take the company to the next stage of development," said Christopher Locher, CEO of Versatope, in a press release.
As of July 2024, two malaria vaccines are being deployed in various countries.
For example, the African country of Côte d'Ivoire recently became the first nation to deploy the R21/Matrix-M™ vaccine.
"The introduction of the R21/Matrix-M™ malaria vaccine in Côte d'Ivoire marks a breakthrough in the fight to protect vulnerable children against a leading cause of death across the region while reinforcing our mission to create innovative vaccines that improve public health," said John Jacobs, Novavax Inc.'s President and CEO, said in a press release on July 15, 2024.
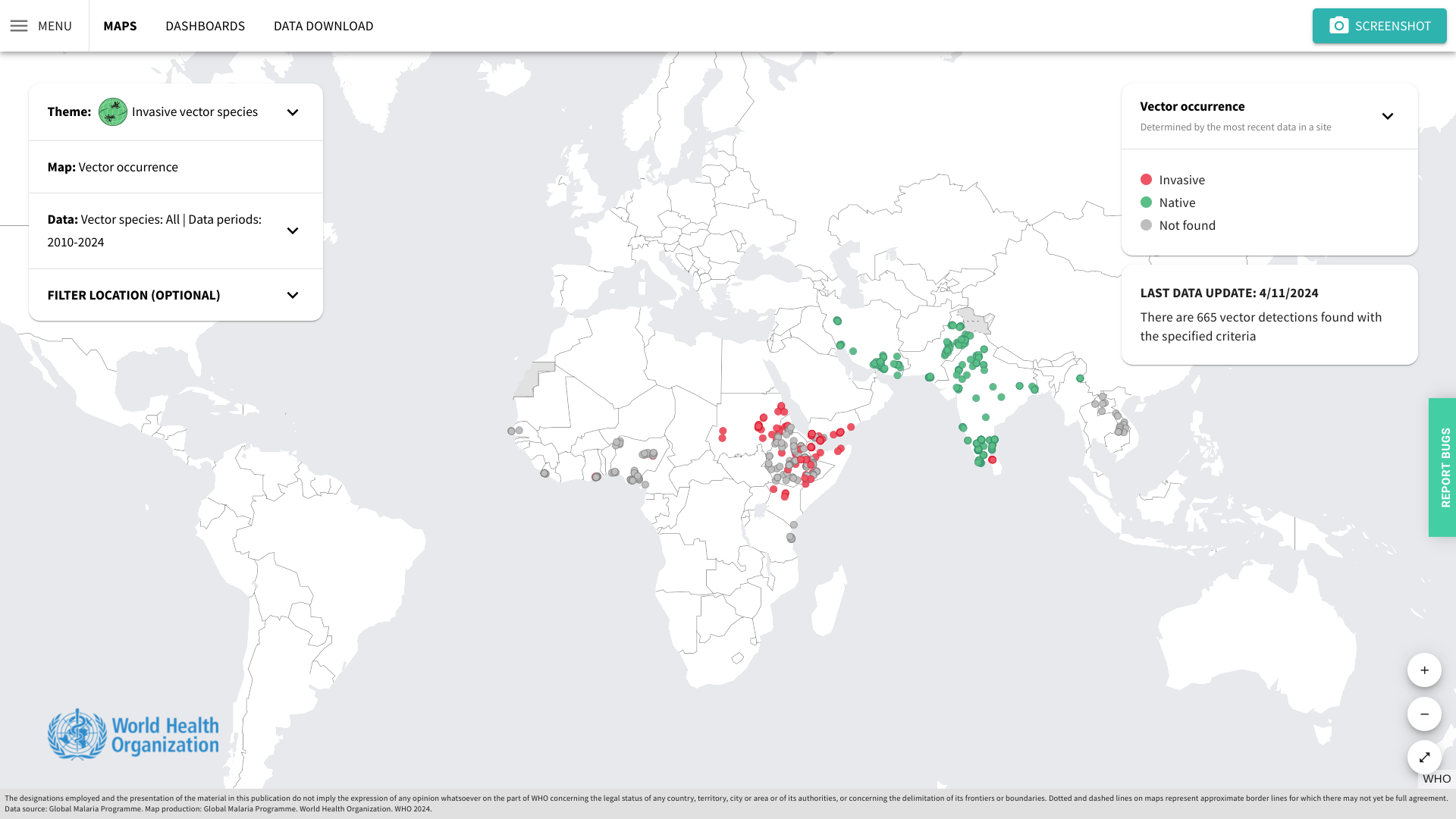
A new World Health Organization (WHO) project aims to accelerate the development and accessibility of human avian influenza (H5N1) messenger RNA (mRNA) vaccine candidates.
The Argentinian manufacturer Sinergium Biotech is leading this effort, leveraging the WHO and the Medicines Patent Pool mRNA Technology Transfer Programme.
Sinergium Biotech has developed candidate H5N1 vaccines and aims to establish proof-of-concept in preclinical models. Once the preclinical data package is concluded, the technology, materials, and expertise will be shared with other vaccine manufacturing partners, accelerating the development of H5N1 vaccine candidates and bolstering pandemic preparedness efforts.
The mRNA Technology Transfer Programme, jointly developed by WHO and MPP, was launched in July 2021.
"This initiative exemplifies why WHO established the mRNA Technology Transfer Programme – to foster greater research, development, and production in low- and middle-income countries so that when the next pandemic arrives, the world will be better prepared to mount a more effective and more equitable response," said Dr. Tedros Adhanom Ghebreyesus, WHO Director-General, in a press release on July 29, 2024.
Avian influenza viruses are a public health risk due to their global circulation in birds and mammals. According to the WHO, they have the potential to cause a future human pandemic.
However, the WHO says avian influenza viruses are currently a low risk for most people.
In the United States, avian influenza vaccines have been developed, candidates are under development, and one has been U.S. FDA-approved.

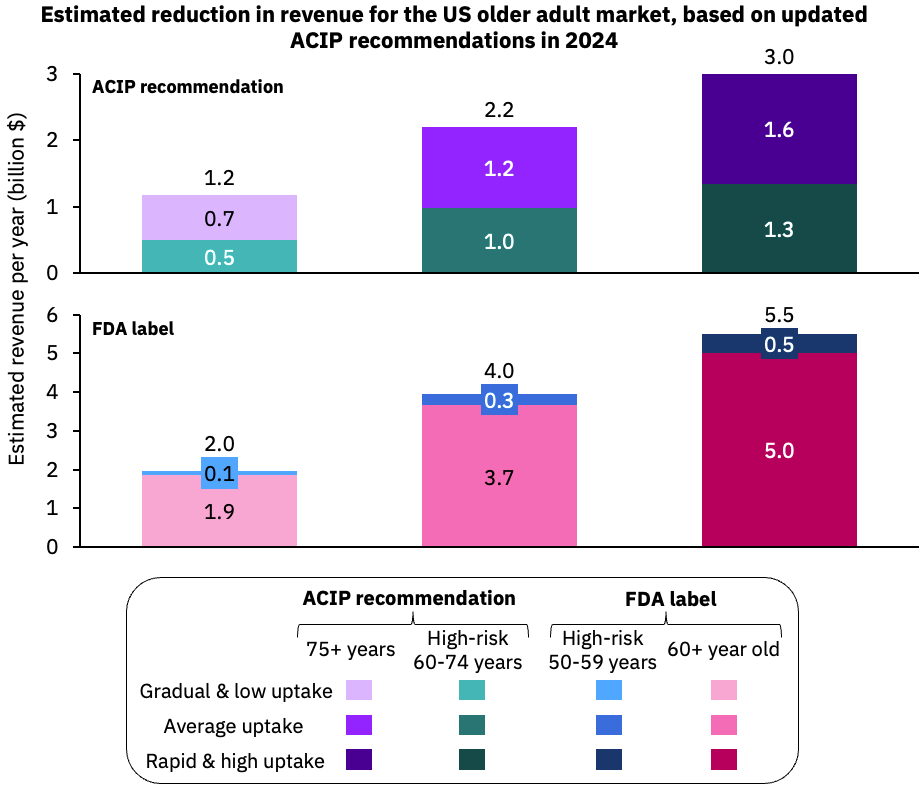
The market research firm Airfinity Limited recently lowered its sales projections for Respiratory Syncytial Virus (RSV) vaccines for older adults in the United States from $4.7 billion annually to $1.7 billion by 2030.
Airfinity’s estimate framework announced on July 23, 2024, expects 2024 revenues to reach $2.2 billion.
Which is reduced from 2023, when RSV vaccine revenues totaled about $ 2.4 billion.
This change in forecast follows new recommendations from the U.S. CDC vaccine advisory committee that RSV vaccines be offered only as a single lifetime dose for older adults. This reduces the estimated eligible population to about 46 million people.
Furthermore, the CDC has not announced a decision on potential booster doses.
However, future dosing recommendations could change as long-term efficacy and safety data emerge. Airfinity wrote that applying scenario frameworks for a booster every two or three years could increase the U.S. market's value to $6.6 billion or $5.2 billion, respectively.
Airfinity’s RSV Lead Isabella Huettner commented, “U.S. market share estimates are difficult to anticipate at this point with different scenarios being possible. Based on current data, GSK (AREXVY™) appears to be most likely to capture the majority of the market in the long term due to promising efficacy and durability."
As of July 28, 2024, three RSV vaccines are approved for use in the U.S.
Previously, Beyfortus™, the first approved extended half-life monoclonal antibody offering passive immunization to prevent lower respiratory tract infections in infants caused by RSV, is expecting a 'blockbuster' performance in the second half of 2024.
Beyfortus produced revenues of €547 million in 2023.
GlobalData plc previously issued a sales forecast indicating Beyfortus could reach global sales of $1.27 billion in 2029.
The U.S. Department of Health and Human Services recently published an amendment to a 2013 emergency declaration under the Food, Drug, and Cosmetic Act that broadens the scope of the agency's assistance in facilitating certain medical countermeasures in response to a public health emergency, such as a pandemic.
The new declaration enables the U.S. Food and Drug Administration (FDA) to extend the expiration date of certain medical products and allow HHS to issue an emergency use authorization for unapproved drugs, devices, or products, among other actions, including vaccines.
As of July 18, 2024, the amendment now applies to pandemic influenza A viruses and others with pandemic potential, such as the current H5N1 strain of avian influenza (bird flu, cow flu).
Previously, the declaration specifically covered just the H7N9 strain.
In June 2024, the U.S. Administration for Strategic Preparedness and Response announced the Pandemic Influenza Preparedness and Response Strategy. The Strategy outlines how the U.S. government will leverage existing infrastructures and capabilities to respond to the current Highly Pathogenic Avian Influenza H5N1.
In April 2024, the FDA's Dr. Peter Marks informed the media that the U.S. stockpile of avian influenza-specific vaccines would work well if deployed. Over the past few years, the U.S. has invested hundreds of millions of dollars in various bird flu vaccines.
As of July 27, 2024, FDA-approved avian influenza vaccines are not commercially available in the U.S.

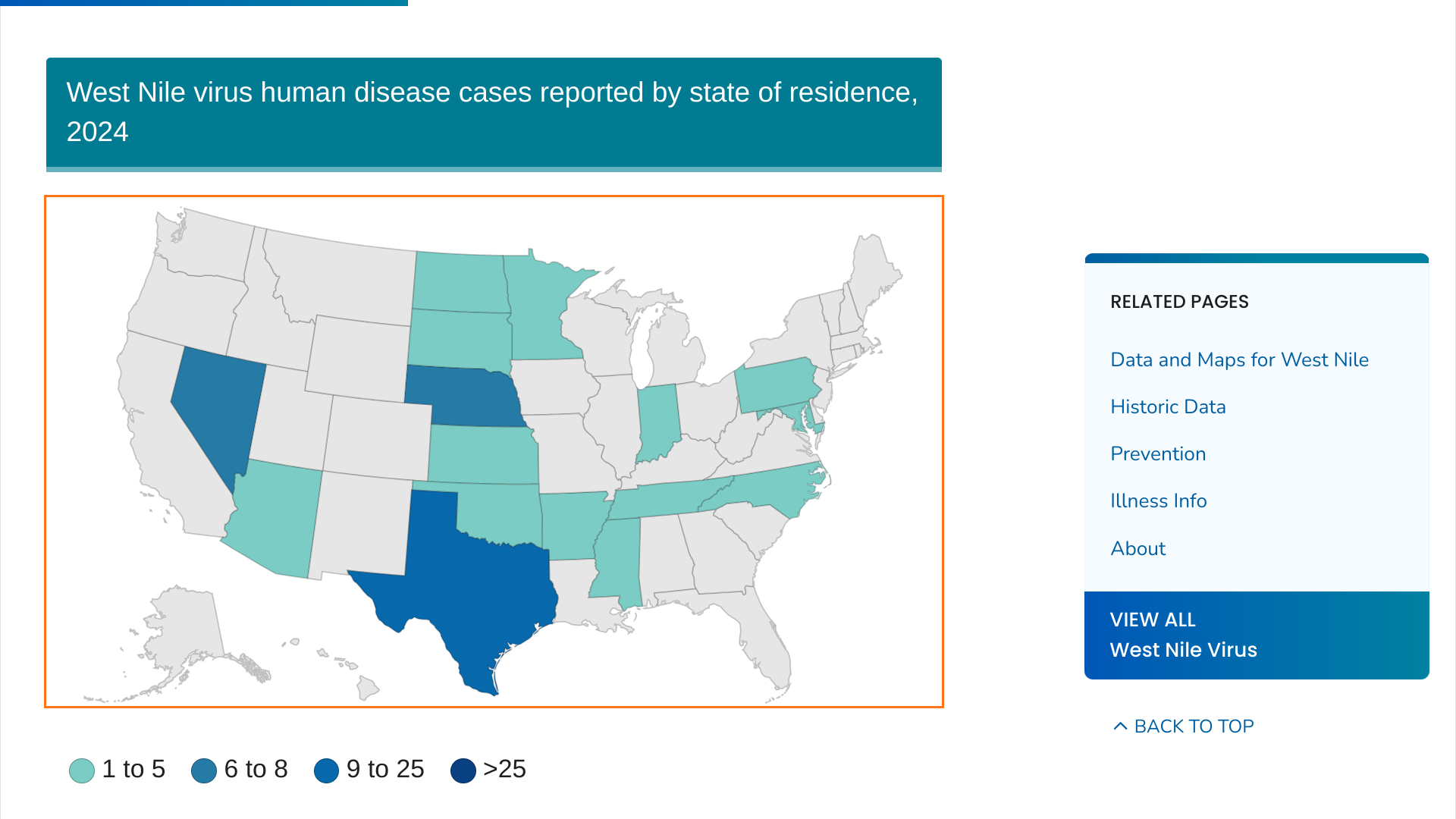
Harris County Public Health’s (HCPH) Mosquito and Vector Control Division today reported a significant increase in West Nile virus (WNV), which is the leading cause of mosquito-borne disease in the continental United States.
On July 26, 2024, seven human cases of WNV were reported to HCPH in unincorporated Harris County (outside the City of Houston).
Additionally, 520 positive mosquito samples were identified across 168 of its 268 operational areas in Harris County, which has a population of about 4.9 million.
HCPH urges residents to protect themselves and their loved ones against this mosquito-transmitted illness.
As of July 23, 2024, the U.S. CDC reported 45 WBV disease cases and 24 WNV neuroinvasive disease cases from 19 states this year.
Furthermore, the CDC says there are no vaccines or medicines to prevent WNV disease.
Several vaccine candidates, including live attenuated chimeric, DNA (first and second generation), recombinant subunit, and inactivated whole-virus vaccines have been the subject of human clinical studies.
The Minnesota Department of Health (MDH) recently confirmed three additional measles cases in unvaccinated children in Anoka, Hennepin, and Ramsey counties.
MDH stated that based on current information, these cases are not directly linked and have not traveled, so there is concern for the possible spread of measles in the community.
As of July 26, 2024, Minnesota has confirmed 15 measles cases in 2024, and is an increase compared to other years. All the cases have occurred among unvaccinated children.
Minnesota isn’t the only state to have seen an increase in measles cases. Just to the south, Chicago, Illinois, reported a significant outbreak (64 cases) this year.
MDH is working with local health departments and other locations to notify people who may have been exposed directly. However, health officials note that anyone not vaccinated against measles could be at risk and should watch for symptoms of measles.
“Measles spreads easily, and it finds those who are vulnerable,” said Jessica Hancock-Allen, infectious disease division director at MDH, in a press release.
“That is why families need to ensure their children are up to date on their immunizations to protect them from this potentially serious disease.”
"The best way to prevent measles is through immunization."
Measles vaccines are generally available at clinics and pharmacies throughout the U.S.



