Search API
UNICEF today announced that it had issued an emergency tender for the procurement of mpox vaccines. The emergency tender is designed to secure immediate access to available mpox vaccines and expand production.
Depending on demand, manufacturers' production capacity, and funding, agreements for up to 12 million doses through 2025 can be made.
Based on recent announcements, there are four mpox vaccines available in 2024.
“Addressing the current mpox vaccine shortage and delivering vaccines to communities who need them now is of paramount importance. There is also a pressing need for a universal and transparent allocation mechanism to ensure equitable access to mpox vaccines,” said Director of UNICEF Supply Division Leila Pakkala in a press release on August 31, 2024.
UNICEF is the world’s largest single vaccine buyer, procuring more than 2 billion doses of vaccines annually for routine child immunization and outbreak response on behalf of nearly 100 countries.

The U.S. Food and Drug Administration today announced it granted emergency use authorization (EUA) for an updated version of the Novavax COVID-19 vaccine that more closely targets currently circulating SARS-CoV-2 virus variants to provide better protection against serious consequences of COVID-19, including hospitalization and death.
Novavax's updated COVID-19 vaccine targets the "parent strain" of KP.2 and KP.3, formulated to target the JN.1 variant.
The updated monovalent, protein-based vaccine is authorized for individuals 12 and older.
This FDA approval helps protect Americans and their families during the U.S.'s biggest surge in COVID-19 cases since January 2022.
The FDA has determined that the updated Novavax COVID-19 vaccine has met the statutory criteria for issuance of an EUA, including that the known and potential benefits of the vaccine outweigh its known and potential risks.
Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research, commented in a press release on August 30, 2024, “Today’s authorization provides an additional COVID-19 vaccine option that meets the FDA’s standards for safety, effectiveness and manufacturing quality needed to support emergency use authorization.”
Following the Center for Biologics Evaluation and Research's release of vaccine batches, Novacax vaccines will be available in thousands of locations, including retail and independent pharmacies and regional grocers.

Emergent BioSolutions Inc. today announced that the U.S. Food and Drug Administration (FDA) approved the supplemental Biologics License Application for the expansion of the indication for the single-dose ACAM2000® Smallpox vaccine to include the prevention of mpox disease in individuals determined to be at high risk for mpox infection.
ACAM2000® is administered percutaneously via a bifurcated needle dipped into the vaccine solution. The skin is pricked several times in the upper arm with a droplet of the vaccine.
ACAM2000 was first approved by the FDA in 2007.
Dr. Amesh A. Adalja, FIDSA FACP FACEP & health security and emerging infectious diseases expert, Johns Hopkins Center for Health Security, commented in a press release on August 29, 2024, “ACAM2000®, a direct descendant of the Jenner vaccine (humanity’s first) which was used to eradicate smallpox, and now with the broadened indication, will be an invaluable tool in this endeavor.”
The FDA's labeling for ACAM2000® contains a contraindication for individuals with severe immunodeficiency. Severe localized or systemic infection with vaccinia (progressive vaccinia) may occur in persons with weakened immune systems. Individuals with severe immunodeficiency who are not expected to benefit from the vaccine should not receive ACAM2000®.
The risk of experiencing severe vaccination complications must be weighed against the risk of experiencing a potentially severe or fatal smallpox or mpox infection.
Additionally, there are warnings and precautions for myocarditis, pericarditis, encephalitis, encephalomyelitis, encephalopathy, progressive vaccinia, generalized vaccinia, severe vaccinial skin infections, erythema multiforme major (including Stevens-Johnson Syndrome), eczema vaccinatum resulting in permanent sequelae or death, accidental eye infection (ocular vaccinia), which can cause ocular complications that may lead to blindness, and fetal death.
These side effects may occur following primary or revaccination with live vaccinia virus vaccines, including ACAM2000®. These risks are increased in certain individuals and may result in severe disability, permanent neurological sequelae, and/or death.
As of late August 2024, four mpox vaccines are in use globally.

Health officials in Minnesota today announced they are urging families to ensure they are up to date on their measles, mumps, and rubella (MMR) vaccine as the four-month measles outbreak continues to impact the Twin Cities metro area.
Since May 2024, 30 cases of measles have been reported in Minnesota. So far, the outbreak is mainly affecting unvaccinated children in the Somali community in Minnesota.
In 2023, the Minnesota Department of Health (MDH) did not report a local measles case.
MDH says most measles cases in Minnesota result from people traveling to or from countries where measles is common and who become infectious with measles after arriving in Minnesota.
“Measles is currently circulating, and infections can be severe,” said Dr. Ruth Lynfield, state epidemiologist and medical director at the MDH, in a press release on August 28, 2024.
“I urge all parents to vaccinate their children because we know that vaccination offers the best protection.”
In Minnesota, the Vaccines for Children program (MnVFC) provides Free or Low-cost Vaccines for Children who are uninsured, enrolled in a Minnesota health care program like Medical Assistance or MinnesotaCare, or American Indian or Alaska Native.
Over 750 healthcare providers, including pharmacists, are enrolled in MnVFC, and approximately half of Minnesotan children are eligible.
Additionally, children who do not have health insurance to cover vaccines can also get vaccinated through local public health departments. Check your county government website to find a local public health immunization clinic.
Minnesota is not the only state reporting a measles outbreak in 2024. On August 22, 2024, the U.S. CDC reported 227 measles cases in 29 jurisdictions, led by Chicago, Illinois.

ARS Pharmaceuticals, Inc. recently announced today that the European Commission (EC) has approved EURneffy® (adrenaline nasal spray) for the emergency treatment of allergic reactions for adults and children (≥30 kg) with severe allergies.
ARS Pharma anticipates that EURneffy will be made available in certain European Union Member States in Q4 2024. It is the first novel adrenaline delivery method approved in over three decades.
The pharmacodynamics and pharmacokinetics of 2 mg EURneffy were evaluated across a range of dosing conditions, including single and repeat dosing, self-administration by patients, dosing in pediatrics, and during multiple nasal conditions that can cause congestion and rhinorrhea such as nasal allergen challenge or infectious rhinitis caused by a cold/flu.
“Adrenaline is the only first-line treatment (not a preventive vaccine) for allergic reactions including anaphylaxis, yet there is significant underutilization of adrenaline due to the limitations of currently available therapy,” said Antonella Muraro, MD PhD, Professor of Food Allergy at the University of Padua, and lead author of the European Academy of Allergology and Clinical Immunology treatment guidelines for anaphylaxis, in a press release on August 26, 2024.
Type I severe allergic reactions are serious and potentially life-threatening events that can occur within minutes of exposure to an allergen and require immediate treatment with epinephrine, the only approved medication for these reactions in the European Union.
While adrenaline autoinjectors have been shown to be highly effective, well-published limitations result in many patients and caregivers delaying or not administering treatment in an emergency situation. These limitations include fear of the needle, lack of portability, needle-related safety concerns, lack of reliability, and complexity of the devices. Even if patients or caregivers carry an autoinjector, more than half either delay or do not administer the device when needed in an emergency, says the company.
The U.S. CDC's Early Release Morbidity and Mortality Weekly Report on August 27, 2024, revealed that 21 Oropouche virus disease cases among U.S. travelers returning from Cuba have been reported this year.
At least three patients had recurrent symptoms after the initial illness, a common characteristic of Oropouche virus disease.
Most of these cases (20) were people in Florida.
The CDC recently published a Level 1 Travel Health Notice to alert travelers regarding Oropouche outbreaks in the Americas Region and Europe (19).
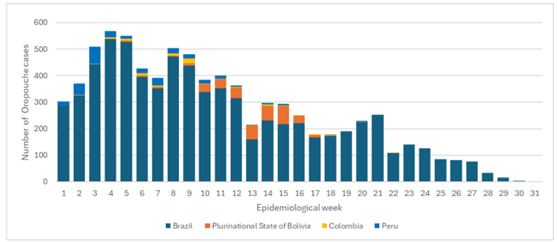
From December 2023 to June 2024, large Oropouche virus disease outbreaks were recognized in areas with known endemic diseases, and the virus emerged in new places in South America and Cuba where it had not been historically reported.
This year, cases have been reported in Bolivia, Brazil, Colombia, Cuba, and Peru.
From 2015 to 2022, only 261 cases of Oropouche fever were recorded in Brazil. However, as of August 6, 2024, Brazil confirmed 7,497 cases. However, the infectious rate has recently diminished.
The CDC says clinicians and public health jurisdictions should be aware of Oropouche virus disease in U.S. travelers and request testing for suspected cases.
In Florida, the Department of Health identified suspected Oropouche cases primarily by reviewing patients who received negative dengue test results and had visited countries such as Cuba.
Reported symptoms commenced during May–July and most commonly included fever (95%), myalgia, headache, fatigue or malaise, and arthralgia.
Travelers should prevent insect bites when traveling, and pregnant persons should consider deferring travel to areas experiencing outbreaks of Oropouche virus disease, says the CDC.
As of August 28, 2024, there are no approved vaccines to prevent Oropouche virus disease.
UCLA Health recently announced it had received a $120 million commitment from surgeon, inventor, and philanthropist Dr. Gary Michelson and his wife, Alya, to kick-start the California Institute for Immunology and Immunotherapy, an innovative public-private partnership aimed at spurring breakthrough discoveries that prevent and cure diseases and catalyze economic growth and innovation in Los Angeles.
Announced on August 27, 2024, the gift will be distributed via the Michelson Medical Research Foundation, designates $100 million to establish two research entities within the institute, each funded by $50 million,
One entity will focus on rapid vaccine development and the other on harnessing the microbiome to advance human health. The microbiome research will be conducted with the new UCLA Goodman-Luskin Microbiome Center.
In addition, the foundation, a part of the Michelson Philanthropies network of foundations, is funding a $20 million endowment to provide research grants to young scientists using novel processes to advance immunotherapy research, human immunology, and vaccine discovery.
“Immunology is the mediator of nearly all human diseases, whether we’re talking about cancer, heart disease, or Alzheimer’s,” Dr. Michelson said in a press release.
“The vision for this institute is to become a ‘field of dreams’ — the world’s leading center for the study of the immune system to develop advanced immunotherapies to prevent, treat, and cure all of the diseases that afflict people today and to end these diseases in our lifetime.
“Scientific research is the key to making possible longer and healthier lives,” Michelson added.