Search API
The U.S. Centers for Disease Control and Prevention (CDC) has repeatedly stated that the majority of measles cases confirmed in the United States are linked to unvaccinated international travelers.
This assumption was reaffirmed in late August 2024 when the CDC reported that out of the 236 measles cases this year, 87% were in patients who were either unvaccinated or whose vaccination status was unknown, and most had contact with international travelers.
Various health agencies have recently updated their data to help determine where these measles viruses may have originated.
The Europen CDC recently reported that 30 EU/EEA Member States had reported a total of 17,949 measles cases of measles and 13 related deaths over the past year.
For example, in July 2024, Romania (382), Belgium (92), Italy (91), and Germany (84) reported the highest measles patient counts. Throughout 2024, Germany has reported 435 measles cases.
Other recent measles hot spots have been India, with about 24,000 measles cases over the past year.
As of August 2024, 12,481 measles cases had been confirmed in the Region of the Americas.
The overall measles vaccination average in the Americas is about 85% coverage for the first MMR vaccine dose, indicating millions of unprotected people in the Americas.
While MMR vaccination is not a requirement to visit the U.S., the CDC recommends that all international travelers speak with a travel vaccine provider about their options to protect themselves from this highly transmittable virus.

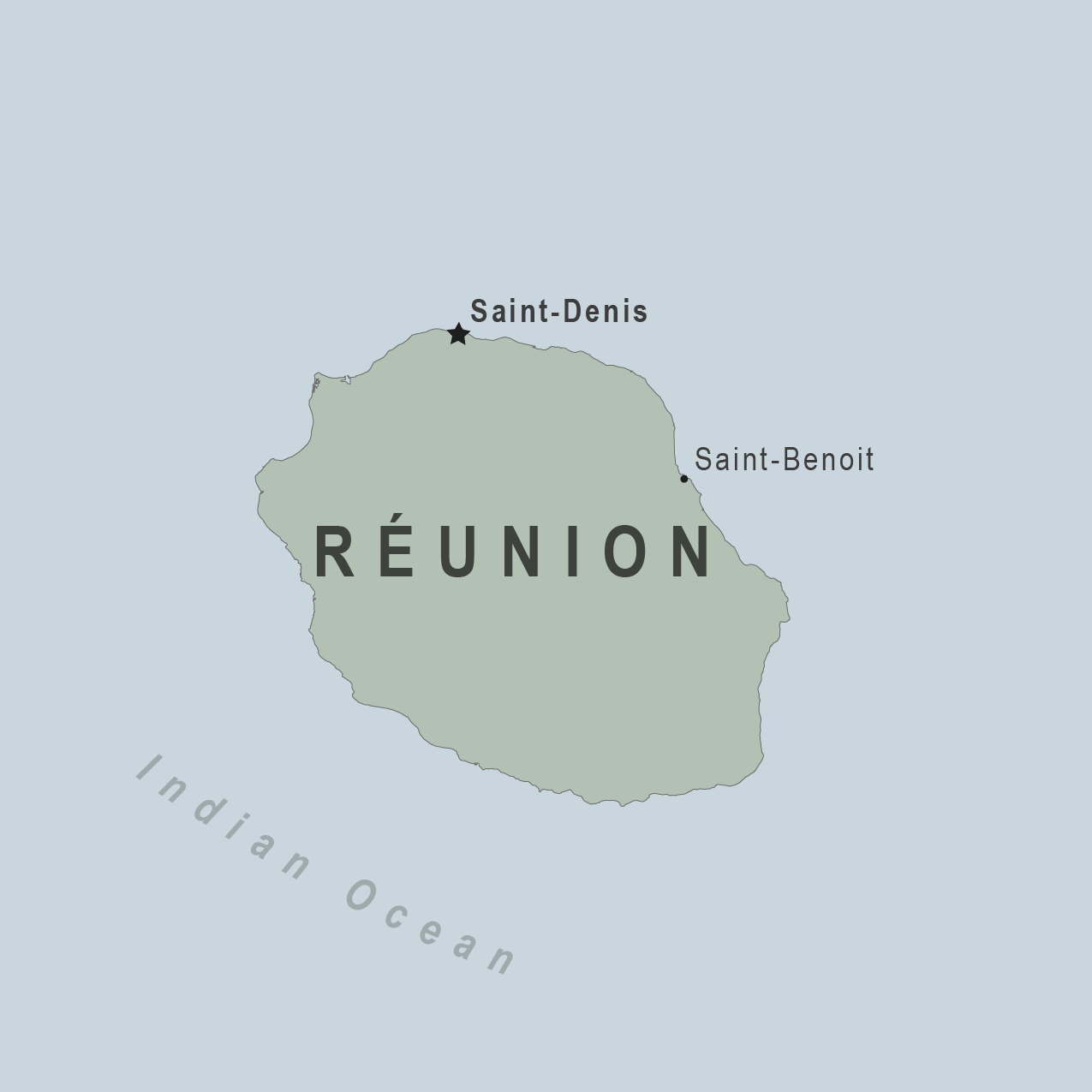
The European CDC Communicable Disease Threats Report, published on August 30, 2024, disclosed that France had reported three autochthonous (locally-acquired) chikungunya virus disease cases in the Department of La Réunion from the same neighborhood.
The last major chikungunya outbreak in La Réunion was from 2005 to 2006.
The ECDC says the risk of chikungunya infection for residents and travelers to La Réunion is currently low. This mosquito-transmitted disease activity has been reduced as it's winter in La Réunion.
However, further cases cannot be excluded, says the ECDC.
La Réunion is an island in the Indian Ocean east of Madagascar and Africa that welcomes over 350,000 visitors annually.
As of September 2, 2024, the U.S. CDC had not issued a Travel Healht Advisory regarding La Réunion's chikungunya outbreak.
From a disease prevention option, Valneva SE's IXCHIQ® single-dose, live-attenuated chikungunya vaccine has been approved by the U.S. FDA and throughout Europe. Travel vaccination services are offered throughout the United States at certain pharmacies and Passport Health USA.
OVAX Inc. is now emerging from stealth mode with an initial $10 million seed funding backed by its team, concerned parents, and mission-aligned investors. According to Pitchbook reporting, no enterprise valuation was disclosed.
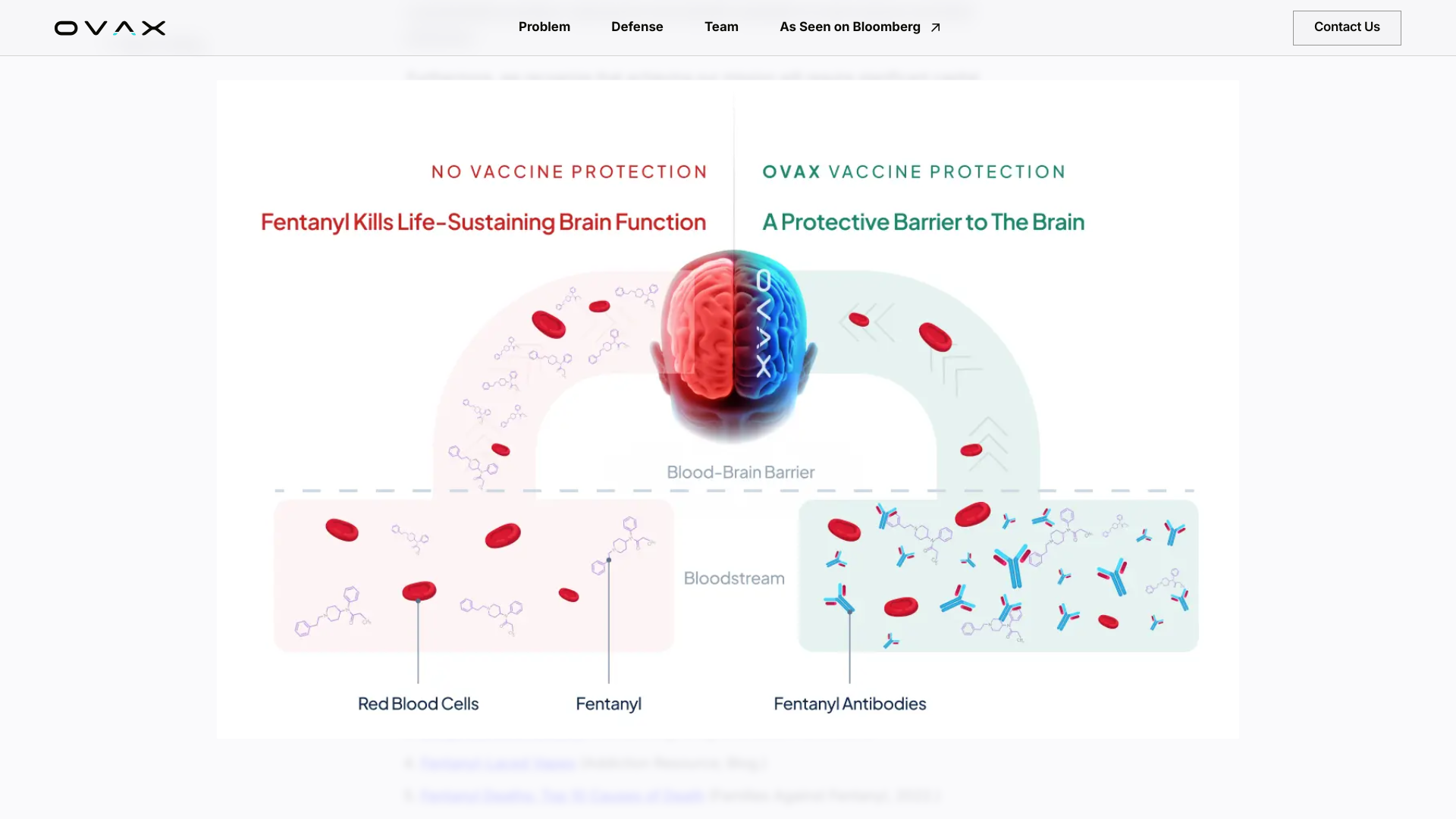
OVAX is developing Fentanyl Armour nasal spray vaccine technology designed to stop fentanyl overdoses. Illicit fentanyl kills approximately 200 Americans daily.
Fentanyl is a synthetic opioid that is often added to illegal drugs, says the U.S. CDC.
The company offers a defense system that immediately neutralizes fentanyl upon exposure and blocks the euphoric effects, enabling the healthcare industry to prevent overdoses leading to death.
On June 24, 2024, the company's media statement confirmed, 'Our vaccines (candidates) are designed to be taken a few times per year with several options in development, including a convenient nasal spray designed to be administered in the comfort of one's home, eliminating the requirement for a needle injection.'
The company licensed intellectual property from the University of Houston, Texas, which completed pre-clinical evaluations.
According to Ovax executives' comments, they plan to launch human clinical trials of Fentanyl Armour in 2025.
'We recognize that achieving our mission will require significant capital investment and a dedicated team. Additionally, OVAX will work alongside key collaborators at non-profits, governments, and communities dedicated to solving the fentanyl crisis,' wrote the company.
As of September 2024, there are no approved vaccines targeting fentanyl overdosing.
The U.S. Centers for Disease Control and Prevention (CDC) recently updated its annual vaccination recommendations for the 2024-25 flu season, noting two fundamental changes.
On August 29, 2024, the CDC stated (73(5);1–25) the primary updates include the following two topics: the composition of 2024–25 seasonal influenza vaccines with the return to trivalent vaccines and updated recommendations for vaccination of adult solid organ transplant recipients.
The CDC recommends that everyone six months and older, with rare exceptions, receive an annual flu shot beginning in the Fall of 2024—decisions about timing need to consider the unpredictability of the flu season.
As of September 2, 2024, various flu shots are available at most pharmacies in the United States. Last flu season, over 157 million influenza vaccines were distributed in the U.S.
A new study from researchers at Wake Forest University School of Medicine sheds light on how the U.S. news media recently portrayed scientific evidence and the uncertainty surrounding unproven therapeutics.
The research team analyzed news reports on how scientific evidence, evidence details and limitations, safety, efficacy, and sources of authority were portrayed to the public.
“We found that 67% of news reports included scientific evidence, but only 24% mentioned scientific publications or journals,” said the study’s corresponding author in a press release on August 29, 2024.
Zubin Master, Ph. D., associate professor of social sciences and health policy at Wake Forest University School of Medicine, commented, “This period of time (the recent pandemic) was when medical specialists and the general public were anxiously scrambling to learn as much as possible about prevention and treatments because there were yet no proven therapeutics or vaccines."
"This makes for an ideal case study to examine how the news media portrays scientific evidence.”
According to the American Press Institute, only 40% of the public read news articles beyond headlines or lead paragraphs.
“It’s crucial, especially with controversial science topics, that the evidence and uncertainty are featured more prominently,” Master said.
The study authors also noted that science can be strengthened by acknowledging limitations and by portraying science as a process that is constantly changing and being corrected as additional knowledge is gained.
These findings appear online in the Journal of Medical Internet Research Infodemiology.

With more than 150 countries and territories reporting rabies cases, accessing one of the four authorized vaccines is essential to reducing this viral disease, says the World Health Organization.
Bavarian Nordic recently announced it is in agreement with the U.K.'s Medicines and Healthcare products Regulatory Agency (MHRA) regarding reports of rubber particles after reconstitution of the Rabipur rabies vaccine.
Bavarian Nordic has recently received an unexpected number of product quality complaints about visual particles in the vaccine solution.
Recommendations, issued on August 14, 2024, have been provided to minimize health risks. The analysis revealed that these particles consisted of rubber transferred from the rubber stopper of the vaccine vials during (coring) reconstitution.
The MHRA's letter to healthcare providers recommends that the reconstituted Rabipur vaccine be carefully inspected visually and not administered if visible particles are present. Suspected adverse drug reactions can be reported to the MHRA through the Yellow Card scheme.
While most of the world traces rabies infections in people to bites from an infected dog, in the United States, rabid bats are the leading source of infection.

The recently announced Africa Regional Polio Eradication Action Plan 2024-2025 outlines a new strategy for responding to polio outbreaks.
Released on August 19, 2024, the pre-publication version introduces a different approach by addressing all instances of poliomyelitis (polio) transmission, including outbreaks of circulating variant poliovirus, as if they were cases of wild poliovirus (WPV).
This action plan advocates for a more proactive strategy instead of following the previous two-round campaign response. It suggests that countries affected by polio should conduct between three and five vaccination campaign rounds based on their specific risk and population immunity.
The plan's authors wrote, 'By pursuing a new course of action defined by these priorities, the WHO African Region will end outbreaks and build the resilience required to achieve and maintain a polio-free Africa.'
Furthermore, this plan offers hope for the countries currently reporting poliovirus detections. On August 28, 2024, the Global Polio Eradication Initiative reported the following summary:
Afghanistan: four WPV1 cases,
Pakistan: two WPV1 cases and 16 positive environmental samples,
Chad: two cVDPV2 cases and two positive environmental samples,
Côte d’Ivoire: two cVDPV2 positive environmental samples,
DR Congo: one cVDPV2 case,
Niger: one cVDPV2 positive environmental sample,
Nigeria: three cVDPV2 cases and one positive environmental sample,
Palestinian territory: one cVDPV2 case,
South Sudan: one cVDPV2 case.
To alert international travelers to their potential polio risk, the U.S. CDC reconfirmed in August 2024 that before any of 37 countries, ensure you are up to date on your polio vaccines.
The GPEI previously launched its Polio Eradication Strategy 2022–2026. The GPEI strategy called for a rigorous review of its plan in 2023.
As of late August 2024, the type 2 novel oral polio (nOPV2) vaccine, produced by PT Biofarma, has been administered over one billion times in various countries. This vaccine was designed to improve phenotypic stability and make the poliovirus strains less prone to reversion to virulence.