Search API
Bavarian Nordic today announced the commercial availability of Vaxchora® (CVD 103-HgR), the only single-dose oral vaccine approved in Canada to protect against cholera.
For approximately 10% of people infected with cholera, a sudden and severe intestinal infection marked by acute watery diarrhea, a severe presentation of cholera can rapidly cause dehydration and death if left untreated.
Nearly 4 million cases of cholera occur during outbreaks annually. The number of reported cholera-related deaths increased by 71% in 2023 compared to 2022.
“To help (Canadians) prevent illness while traveling internationally, we are expanding our vaccine offerings to include protection against cholera. Healthcare providers can now offer this new vaccine option to travelers who plan to visit countries where cholera is present,” said Karinne Lacombe, Canada Country Director, Bavarian Nordic, in a press release on September 16, 2024.
Cholera is endemic to approximately 50 countries and is common in Asia, Africa, and Central and South America. The Southeast Asia region, which includes Bangladesh and India, has the largest populations at risk for cholera.
While cholera is most commonly transmitted by consuming contaminated water, it may also be acquired from eating raw or undercooked food, especially fish and shellfish.
Other cholera vaccines, such as Valneva SE's oral, inactivated DUKORAL® vaccine, will be available globally in 2024. According to the World Health Organization, two doses of Dukoral® provide protection against cholera for two years.
Most cholera cases in the United States are linked to international travel, such as to Haiti.
Travel vaccine experts offer cholera vaccination advice at clinics and pharmacies in the U.S.
As the respiratory syncytial virus (RSV) activity ramps up for the 2024-2025 season, more children will have access to an innovative therapy this year.
Sanofi U.S. announced today that it is shipping BEYFORTUS™ 50mg and 100mg injection doses to private healthcare providers in the United States.
BEYFORTUS (Nirsevimab-alip) is the first and only long-acting monoclonal antibody approved for the prevention of RSV lower respiratory tract disease (LRTD) in newborns and infants born during or entering their first RSV season and for children up to 24 months of age who remain vulnerable to severe RSV disease through their second RSV season.
BEYFORTUS is not an RSV vaccine but offers passive immunization.
Bridgewater, N.J.-based Sanofi stated it anticipates having enough supply so that every eligible baby born outside of the season will have access to immunization at a regular checkup, and those born during the season will have access at birth.
Thomas Grenier, Head of Vaccines, North America, Sanofi, commented in a press release on September 16, 2024, "We're proud to offer BEYFORTUS doses to help protect every eligible baby in the U.S. this RSV season."
"This upcoming season, we look forward to BEYFORTUS offering its demonstrated real-world protection to as many infants as possible."
Sanofi also took additional measures to ensure greater readiness for this season by launching the BEYFORTUS Reservation Program, which provides critical insight into private healthcare provider demand and allows for prioritized fulfillment of requests placed through the program.
Furthermore, the Centers for Disease Control and Prevention's Vaccines for Children program will help ensure that the majority of doses are available before RSV season.
As of May 2024, about 41% of women with young infants reported that their child had received BEYFORTUS.
This announcement is essential as RSV is a highly contagious virus that can lead to serious respiratory illness in infants. Two out of three infants are infected with RSV during their first year of life, and almost all children are infected by their second birthday.

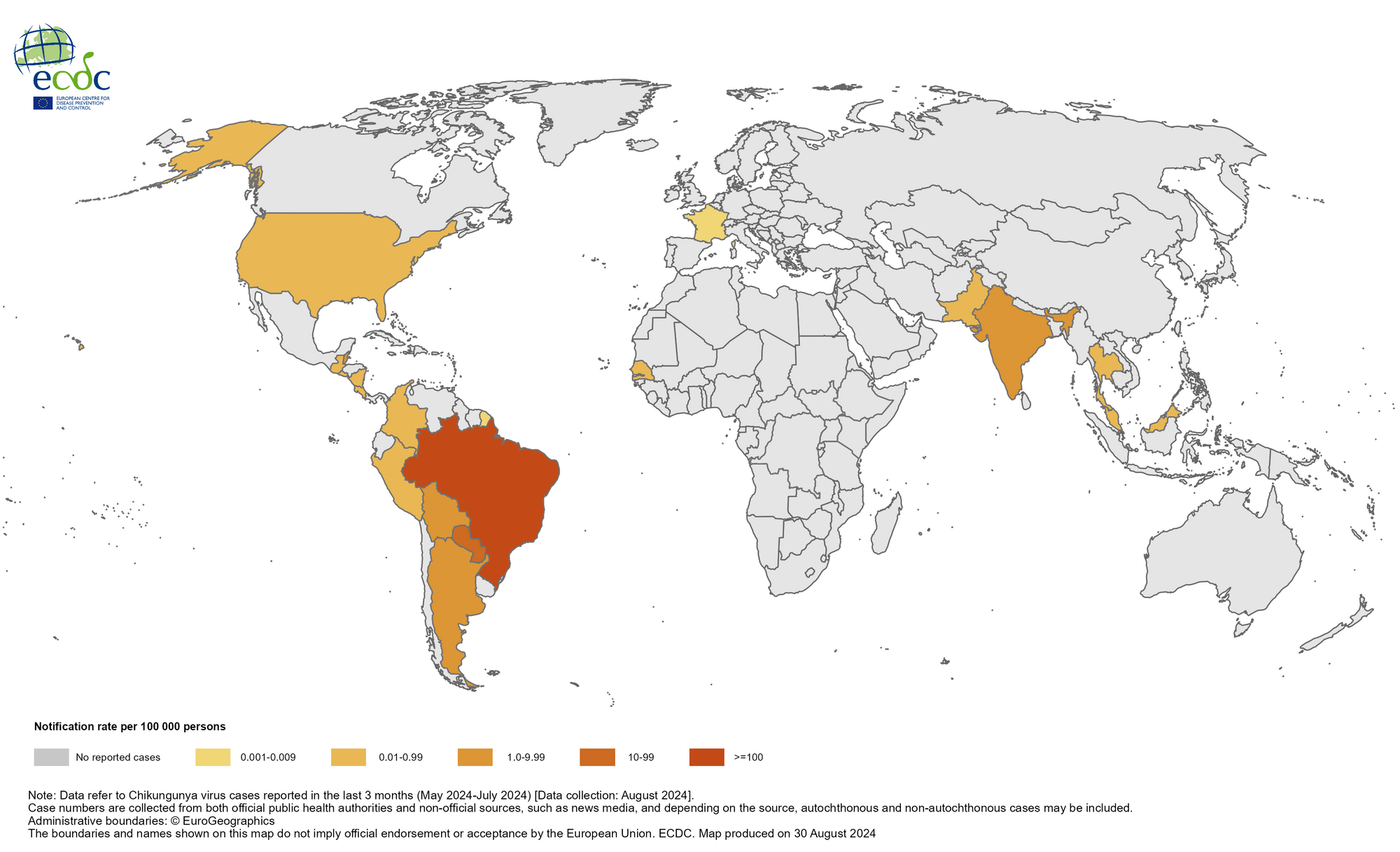
The Chikungunya virus (CHIKV) disease was first reported in the Republic of India in 1963 and has since spread to all parts of the subcontinent, becoming endemic. This health risk is seldom discussed, with over 9 million people visiting India, including about 1.5 million people from the United States.
As of early September 2024, India's health department had reported 69,395 cases of CHIKV this year. At this rate, India may exceed last year's unfortunate outbreak record of 200,064 CHIKV cases, which was the most significant number reported in Asia.
For example, the Times of India reported on September 14, 2024, that chikungunya cases had spiked in Maharashtra. By September 7, 2024, there were 2,643 cases, surpassing the 1,702 recorded in 2023, representing a 55% increase in just nine months.
This year, Pakistan (1,302), The Maldives (389), and Thailand (280) have also reported increases in CHIKV cases in the Asia/Pacific region.
As of early August 2024, approximately 450,000 CHIKV cases and over 160 deaths have been reported worldwide.
To alert international travelers, the U.S. CDC recently stated that the Chikungunya vaccination (Valneva SE's IXCHIQ®) may be considered for certain visitors and long-term residents in 2024.
The CDC also recommended long-term visitors to India consider Japanese encephalitis vaccination (IXIARO® JESPECT®).
These vaccines are generally available at travel clinics and pharmacies in the U.S. and should be administered at least one month before traveling abroad.
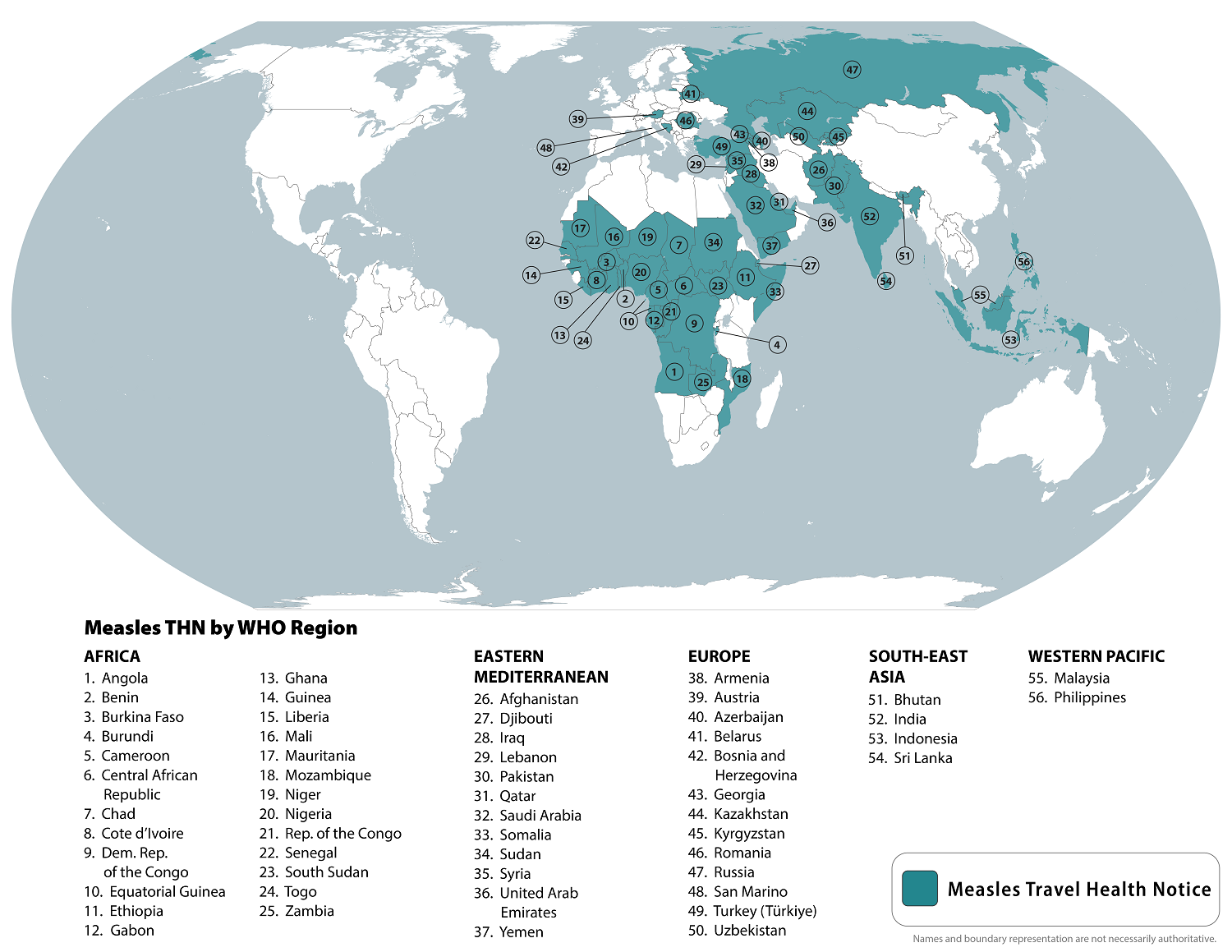
As the global measles outbreak continues in 2024, a 32nd U.S. state confirmed its initial case.
The Tennessee Department of Health (TDH) recently confirmed that a Tennessee resident who recently traveled internationally is recovering from a measles infection.
As of September 14, 2024, TDH has not identified additional measles cases in Tennessee. The last year in which TDH reported positive measles cases in Tennessee was 2019.
On September 5, 2024, the U.S. CDC reported 251 measles cases in 31 jurisdictions this year. So far this year, Chicago (61), Minneapolis (49), and Portland (31) have reported an unusual number of cases.
The measles virus can spread through the air when an infected person speaks, coughs, or sneezes. It can live for up to two hours in the air or on a surface.
Symptoms include fever, headache, and general unwellness, followed by fever, rash, cough, red eyes, or congestion, says the TDH.
To notify international travelers of the measles outbreaks, the CDC reissued a Level Travel Health Advisory in August 2024, identifying 56 countries reporting measles cases to alert international travelers of the ongoing health risk.
The CDC recommends that international travelers speak with a healthcare provider at least one month before traveling abroad about measles vaccination options.
CSL and self-amplifying mRNA (sa-mRNA) pioneer Arcturus Therapeutics today announced that Japan's Ministry of Health, Labor and Welfare (MHLW) granted approval and authorization for their updated sa-mRNA COVID-19 vaccine, KOSTAIVE®.
According to the September 13, 2024, press release, KOSTAIVE is the world's first commercially available sa-mRNA COVID-19 vaccine for adults 18 and older.
Unlike conventional mRNA vaccines, sa-mRNA vaccines instruct the body to make more mRNA and protein to boost the immune response.
"We believe KOSTAIVE® has the potential to change the paradigm for COVID-19 vaccines in Japan," commented Jonathan Edelman, M.D., Senior Vice President, Vaccines Innovation Unit, CSL.
"Today's approval further demonstrates CSL's promise to pursue, develop, and deliver new innovative treatment options to protect public health."
Meiji Seika Pharma, CSL's exclusive partner in Japan, will begin vaccine distribution next month.
Following a peak in July 2024, MHLW data indicates COVID-19 cases in Japan have rapidly declined as of early September.
To avoid diseases such as Japanese encephalitis (IXIARO®), measles, and rubella, the U.S. CDC recommends speaking with a travel vaccine expert at least one month before visiting Japan.
CSL, including three businesses: CSL Behring, CSL Seqirus, and CSL Vifor, provides lifesaving products to patients in more than 100 countries and employs 32,000 people.
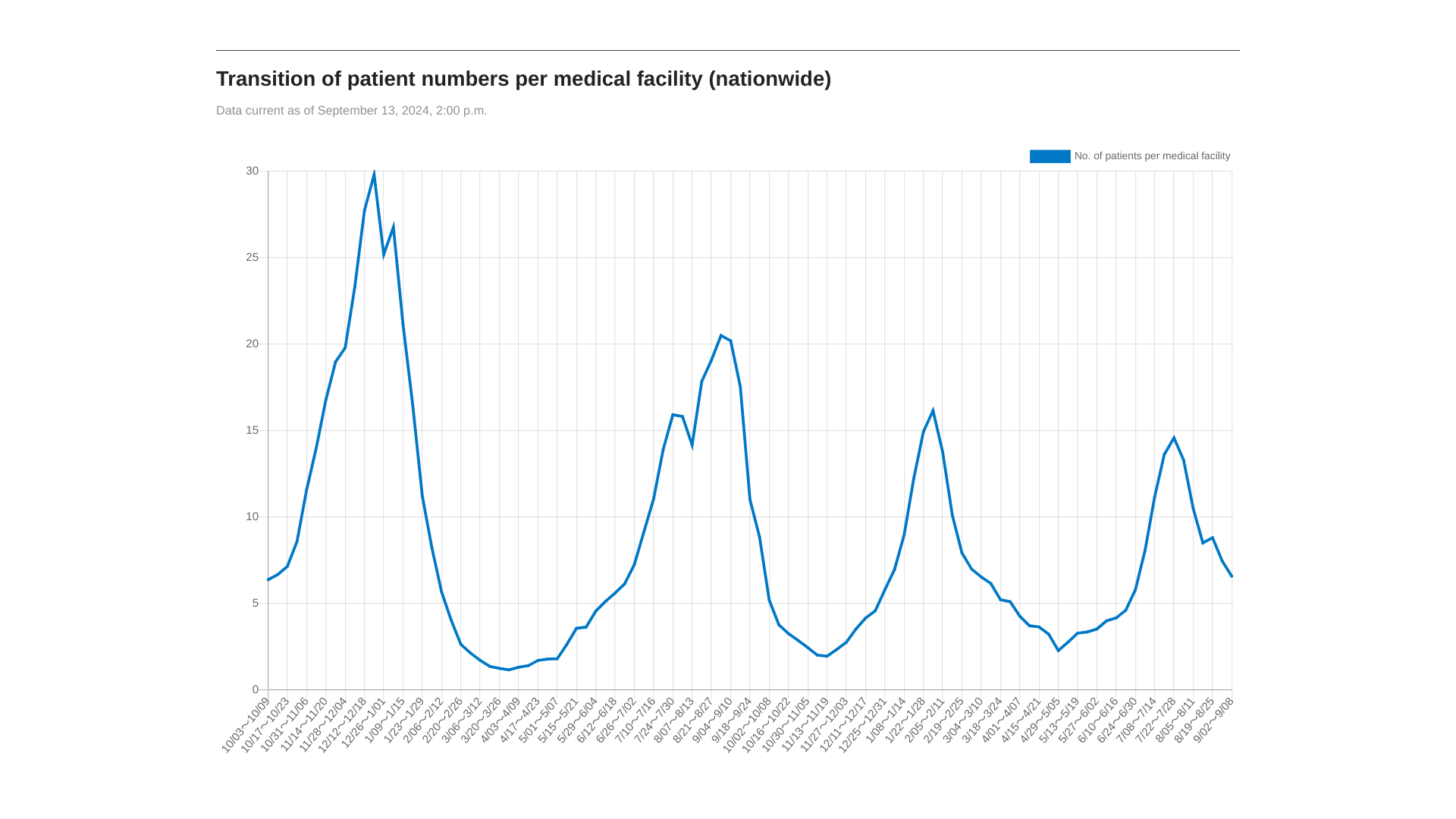
About two years ago, the World Health Organization (WHO) published a report highlighting the first-ever list of fungal priority pathogens, which included 19 fungi representing the greatest threat to public health.
Since then, the WHO has not recommended a fungal vaccine but continues to encourage the development of innovative therapies.
To address this unmet need, F2G Ltd announced a $100 million financing round that will enable the company to complete late-stage development, seek regulatory approval, and prepare for commercialization in the U.S. of olorofim, a novel oral antifungal therapy to treat invasive aspergillosis (IA) and other invasive fungal infections.
It is anticipated that this treatment will be used to treat patients with a serious invasive, rare fungal disease for which existing treatments are inappropriate or no longer effective.
Olorofim is the first orotomide antifungal, an entirely novel class of antifungal agents. It is the only antifungal medication awarded a Breakthrough Therapy Designation for multiple indications by the U.S. Food and Drug Administration.
Olorofim has a novel mechanism of action, different from that of existing classes of antifungals. It exerts fungicidal activity by inhibiting the pyrimidine synthesis pathway.
Henry Skinner, Ph.D., Chief Executive Officer of AMR Action Fund, commented in a press release on September 12, 2024, “Fungal infections are a growing threat to patients worldwide and have a disproportionate impact on vulnerable populations, yet there has been a paucity of innovation in the field of antifungals."
"For decades, clinicians have relied on a handful of antifungal classes, with few mechanisms of action and significant limitations due to spectrum of activity, drug toxicities, or drug-drug interactions. These therapies are increasingly failing in patients."
The U.S. NIH says some fungal infections are more common in people with weakened immune systems or hospitalized individuals, while others can infect anyone, including otherwise healthy people.
As of 2023, fungal vaccine candidates were segmented into broad categories based upon their composition, ranging from multiple to single antigens: whole organism vaccines (live-attenuated or killed fungal cells), crude extracts (fractions derived from cells and medium of fungal cultures), purified subunit vaccines (proteins, peptides), and nucleic acids (RNA and DNA) encoding the antigen(s) of interest.
No fungal vaccine has been approved as of 2024.
The U.S. Centers for Disease Control and Prevention (CDC) today published updated recommendations from the Advisory Committee on Immunization Practices (ACIP) regarding the use of 21-Valent Pneumococcal Conjugate Vaccine Among U.S. Adults.
On September 12, 2024, the CDC's MMWR confirmed the U.S. Food and Drug Administration approved 21-valent pneumococcal conjugate vaccine (PCV) (PCV21; CAPVAXIVE; Merck Sharp & Dohme, LLC) for adults aged ≥18 years.
PCV21 does not contain certain serotypes that are included in other licensed pneumococcal vaccines but adds eight new serotypes. It is recommended for all adults aged ≥65 years and adults aged 19–64 years with certain risk conditions for pneumococcal disease if they have not received a PCV or whose vaccination history is unknown.
Previously, options included either 20-valent PCV (PCV20; Prevnar20; Wyeth Pharmaceuticals, Inc.) alone or a 15-valent PCV (PCV15; VAXNEUVANCE; Merck Sharp & Dohme, LLC) in series with 23-valent pneumococcal polysaccharide vaccine (PPSV23; Pneumovax23; Merck Sharp & Dohme, LLC).
Additional recommendations for the use of PCV20 exist for adults who started their pneumococcal vaccination series with 13-valent PCV (PCV13; Prevnar13; Wyeth Pharmaceuticals, Inc.).
On June 27, 2024, ACIP recommended a single dose of PCV21 for adults aged ≥19 for whom PCV is currently recommended. Indications for PCV have not changed from previous recommendations.
This report summarizes the evidence considered for these recommendations and provides clinical guidance for using PCV21.
In the U.S., various healthcare providers offer pneumococcal vaccines.