Search API
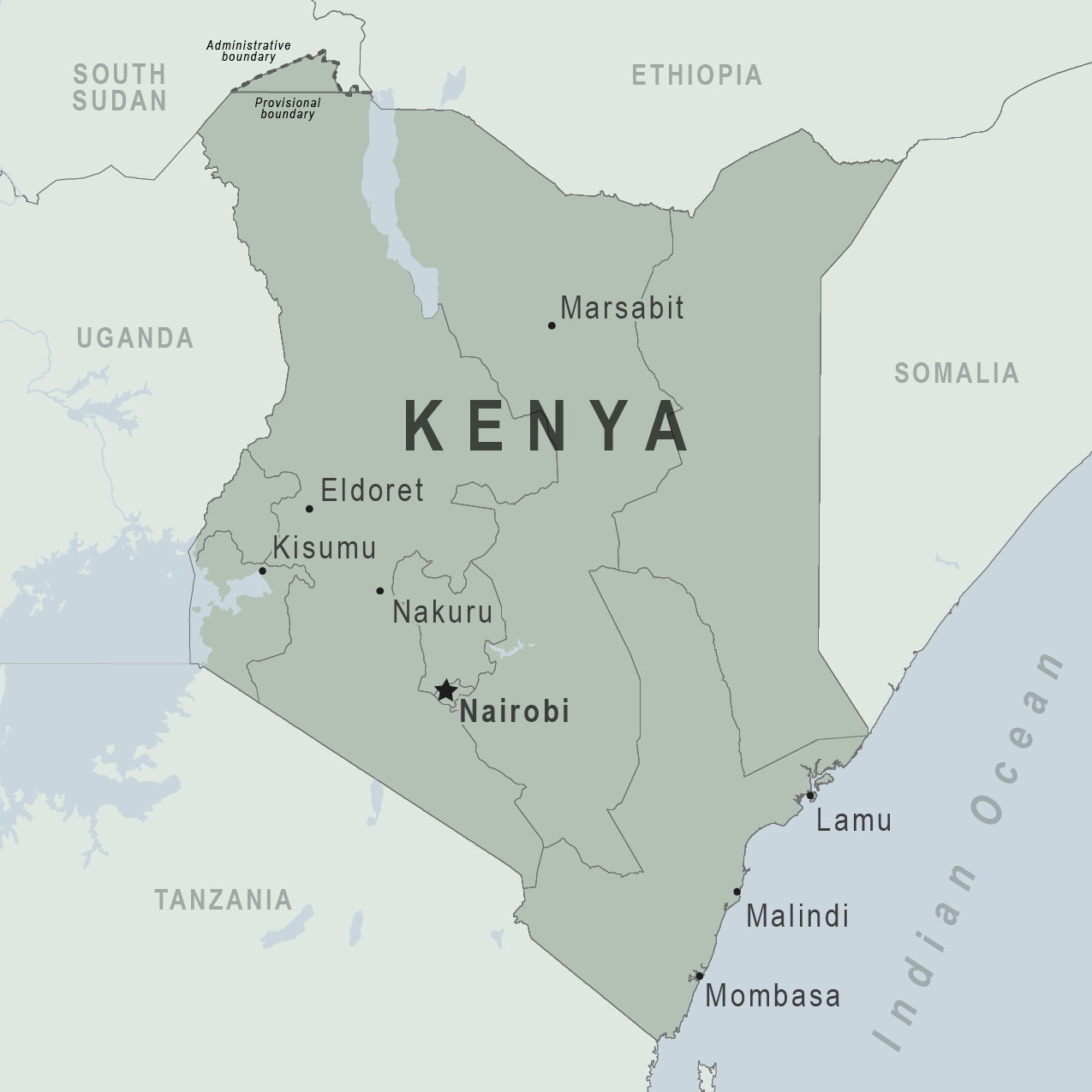
The Republic of Kenya's Ministry of Health announced in October 2024 that a nationwide polio vaccination campaign will launch following an outbreak of circulating Vaccine-Derived Poliovirus Type 2 (cVDPV2).
The vaccination campaign will target over 3.8 million children across nine high-risk counties: Nairobi, Busia, Bungoma, Turkana, Trans Nzoia, West Pokot, Kiambu, Machakos, and Kajiado, said Principal Secretary for Public Health and Professional Standards, in a press release on October 1, 2024.
Five cVDPV2 cases and one positive environmental have been confirmed in Kenya in 2024.
Countries affected by poliovirus circulation are subject to temporary recommendations issued by the WHO's Emergency Committee of the International Health Regulations on Poliovirus. WHO's International Travel and Health recommends that all travelers to polio-affected areas be fully vaccinated against polio.
About 2 million people visit Kenya annually.
Kenyan residents (and visitors for more than four weeks) from infected areas should receive an additional dose of oral (nOPV2) or inactivated polio vaccine (IPV) within four to twelve months of travel.
To alert international travelers of the polio risk, the U.S. Centers for Disease Control and Prevention reissued a Global Polio ADvisory regarding polio outbreaks and poliovirus detections in 37 countries.
The IPV is offered in the U.S. and at most clinics and travel pharmacies in 2024.
Additionally, the CDC recommends yellow fever, measles, mpox, and cholera vaccination when visiting Kenya.
As the new flu season arrives in the United States, data has reaffirmed the benefits of getting an annual flu shot this year.
CSL Seqirus today announced data from a range of real-world evidence (RWE) studies, highlighting the important role influenza vaccination plays in protecting public health.
RWE complements randomized controlled trial research, assessing influenza vaccine effectiveness continuously and providing an ever-growing data set on health outcomes.
"CSL Seqirus remains committed to evaluating vaccine effectiveness through the rigorous generation of real-world evidence," said Gregg Sylvester, Chief Health Officer of CSL Seqirus, in a press release on October 1, 2024.
Among others, RWE studies being presented by CSL Seqirus at OPTIONS XII include:
Results of a study of the 2022/23 influenza season in the U.S. showed that cell-based quadrivalent (QIVc) vaccines prevented more outpatient test-confirmed influenza for those aged six months to 64 years compared with the egg-based quadrivalent (QIVe) vaccines.
Results from a study of the 2023/24 influenza season in the U.S. showed that cell culture-based inactivated influenza vaccine (ccIIV4) generated real-world effectiveness against medically attended, laboratory-confirmed influenza, compared with unvaccinated participants. Vaccine effectiveness was highest in children six months to three years of age at 88%.
Results from a review of the 2017 to 2020 influenza seasons revealed a higher relative effectiveness of QIVc over QIVe/egg-based trivalent (TIVe) vaccines across the three influenza seasons for test-confirmed and clinically diagnosed influenza.
Results from a study of the 2022/23 influenza season in the U.S. showed comparability between MF59® adjuvanted quadrivalent inactivated influenza vaccine (aQIV) and high-dose quadrivalent inactivated influenza vaccine (HD-QIV) in preventing test-confirmed influenza-related emergency department visits and/or inpatient admissions settings among adults aged 65 and over.
Various influenza vaccines for the 2024-2025 flu season are available at clinics and pharmacies in the U.S.

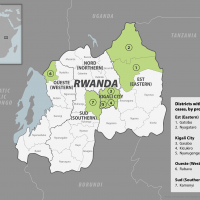
The Republic of Rwanda's health ministry reported today that the sudden Marburg virus disease (MVD) outbreak had reached 29 cases and ten related fatalities.
On September 30, 2024, the World Health Organization confirmed MVD cases from seven of the 30 districts in Rwanda.
Over 70% of the confirmed cases are healthcare workers from two health facilities in Kigali, a city with about 1.5 million residents.
On September 27, 2024, the Rwanda Ministry of Health reported its first Marburg disease case.
Additionally, the U.S. CDC published a Travel Health Advisory to alert international travelers of this expanding health risk. Marburg virus is a Filovirus that, along with Ebola, can cause severe viral hemorrhagic fever infections. MVD was initially reported in 1967 during an outbreak in Marburg a der Lahn and Frankfurt am Main, West Germany.
As of October 1, 2024, no Marburg vaccine candidate has been approved to prevent infections in people.
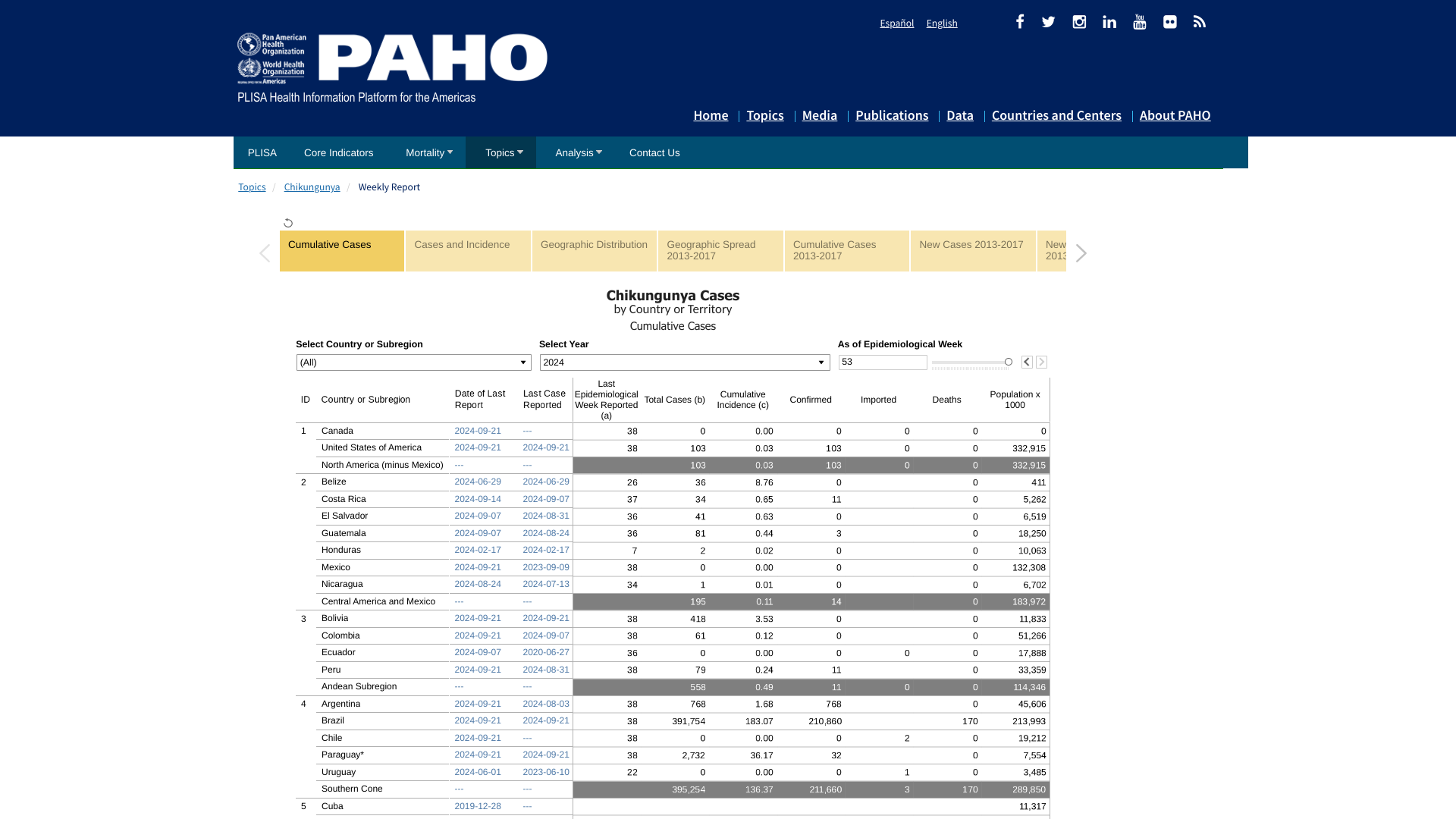
The Chikungunya Virus outbreak in the Region of the Americas has now exceeded 393,000 cases in 2024 and may soon surpass last year's record of 411,548.
Throughout 2024, the Pan American Health Organization has reported 170 Chikungunya-related fatalities in the Americas.
Before 2006, Chikungunya, a mosquito-transmitted virus, was rarely identified in U.S. travelers. However, that trend has changed,
The Centers for Disease Control and Prevention (CDC) data dashboard reported 113 chikungunya cases in 2024, an increase of 34 patients since September 2024. Over 20 states have reported Chikungunya cases this year, led by Massachusetts, New Hampshire, California, Colorado, Illinois, and New York.
The CDC did confirm some good news today; these confirmed cases are travel-related.
Locally acquired Chikungunya cases have not been reported from U.S. states or territories since 2019.
Furthermore, the CDC says all travelers to countries with a risk of Chikungunya should avoid mosquito bites and discuss the need for vaccination with their healthcare provider.
As of October 2024, the Valneva SE's IXCHIQ® single-dose Chikungunya vaccine is readily available at travel clinics and pharmacies in the U.S.
Moderna, Inc. today announced that the first participant in the U.S. has been dosed in the Nova 301 Trial, a pivotal Phase 3 randomized clinical trial evaluating the efficacy, safety, and immunogenicity of an investigational norovirus vaccine, mRNA-1403.
As of October 1, 2024, no U.S. FDA-approved norovirus vaccines are available.
"Norovirus is a significant public health concern that affects millions of people worldwide each year, leading to severe symptoms and, in some cases, hospitalization," said Stéphane Bancel, CEO of Moderna, in a press release.
Enteric viruses, including norovirus, are a leading cause of diarrheal diseases, resulting in significant morbidity and mortality worldwide, particularly among young children and older adults.
Norovirus is highly contagious and a leading cause of diarrheal disease globally, associated with 18% of all AGE, resulting in approximately 200,000 deaths per year and substantial healthcare costs.
The Nova 301 Phase 3 trial is a randomized, observer-blind, placebo-controlled trial evaluating the efficacy, safety, and immunogenicity of mRNA-1403. The trial aims to enroll approximately 25,000 participants 18 years of age and older globally, including in countries in the Northern Hemisphere (U.S., Canada, UK, Japan), the equatorial region, and the Southern Hemisphere (Australia and countries in South America).
Approximately 20,000 participants 60 years of age and older and 5,000 participants between 18 and 59 years of age will be enrolled to assess the investigational vaccine's ability to protect against moderate to severe norovirus acute gastroenteritis (AGE) in adults, with a focus on the older age group that is at the highest risk of severe outcomes, including hospitalization.

For the first time in the United States, 55.8% of infants were protected by two approved immunization methods last year against respiratory syncytial virus (RSV) infections.
According to survey results published in a Morbidity and Mortality Weekly Report (73(38);837–843) on September 26, 2024, 33% of eligible pregnant women reported receiving an RSV vaccination. Most vaccinated women (54.1%) reported receiving the vaccine at an obstetrician or gynecologist's office.
Among women with an infant, 45% reported that their infant received the passive immunization Beyfortus™ (nirsevimab). However, the availability of Beyfortus was limited during the 2023–24 RSV season, creating access challenges and clinical confusion.
Overall, 14.2% of infants were protected by both products.
Furthermore, about 50% of pregnant women did not report receiving a provider recommendation for maternal RSV vaccination or nirsevimab, indicating missed opportunities to protect infants from RSV disease.
These researchers wrote this survey's findings 'underscore the importance of a strong provider recommendation for immunization during the 2024-2025 RSV season.'
As of September 27, 2024, the U.S. CDC's latest RSV data indicates the current RSV season has started slowly but is anticipated to peak in December/January 2025.
The CDC encourages at-risk groups to protect themselves against RSV this winter.
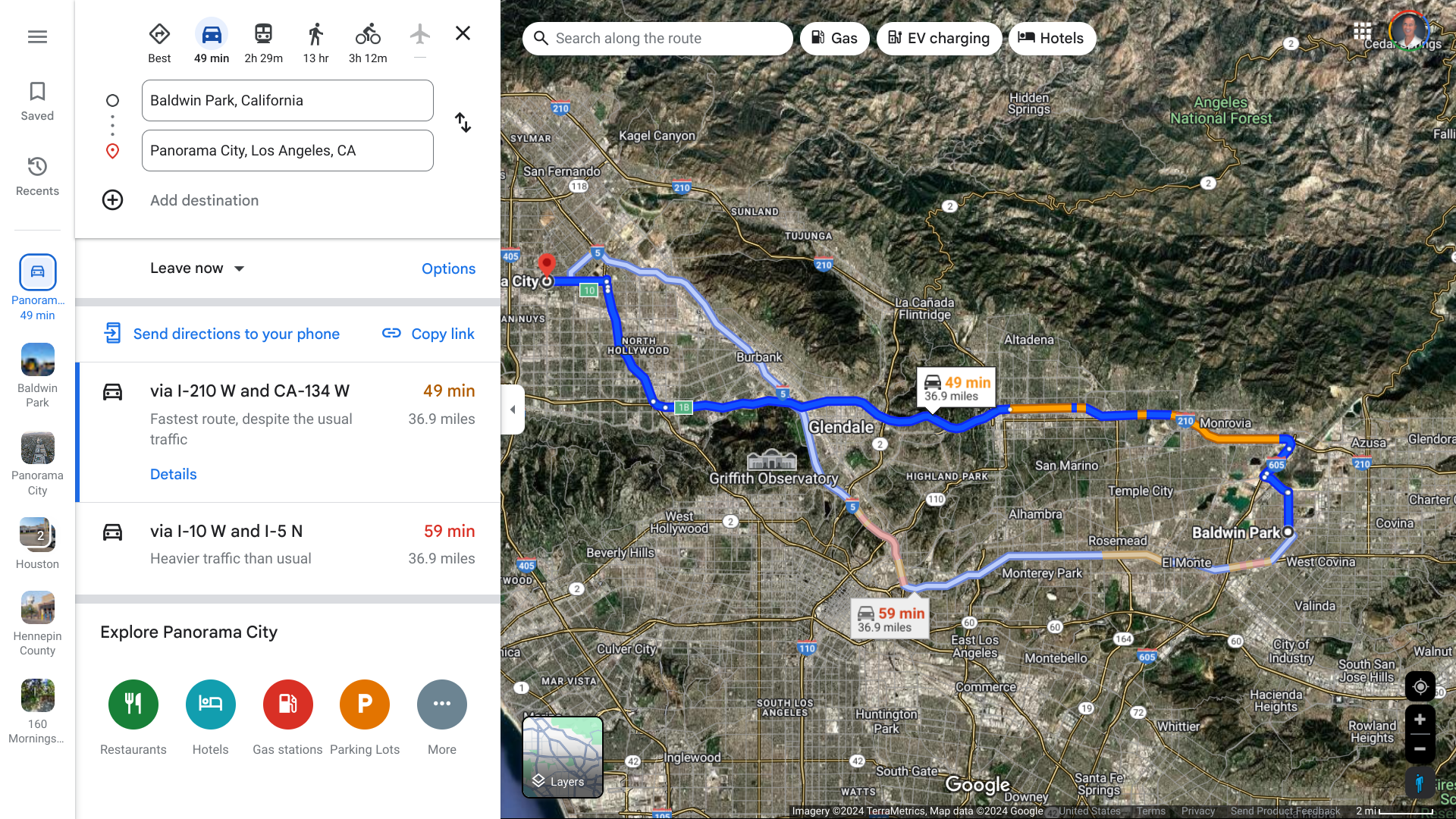
The Los Angeles County Department of Public Health is investigating another case of locally acquired Dengue in a Panorama City resident. The person had no history of travel to areas where Dengue is endemic.
This case of local transmission is not related to the cluster of 3 cases in the City of Baldwin Park on September 18, 2024.
About 55 miles are separating these cities.
Long Beach and Pasadena previously confirmed cases of locally acquired Dengue in fall 2023.
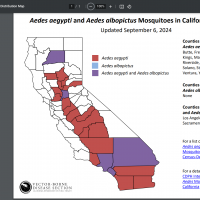
Although Aedes mosquitoes are common in LA County and can spread the dengue virus, almost all previously reported dengue cases in LA County have been associated with travel to a country where Dengue is endemic.
“We are seeing the local dengue transmission unprecedented in Los Angeles County. Preventing mosquito bites and mosquito breeding is the best way to stop the ongoing local transmission of Dengue,” said Muntu Davis, MD, MPH, Los Angeles County Health Officer, in a press release.
As of September 30, 2024, Public Health has reported four cases of locally acquired Dengue. These local transmission cases are extremely rare in LA County, and residents are urged to take proactive steps to prevent mosquito breeding and bites.
The U.S. CDC reported that 50 jurisdictions, led by Florida, New Jersey, New York, and Puerto Rico, had reported over 5,300 dengue cases this year.