Search API
The U.S. CDC's Tom Shimabukuro, MD, MPH, MBA Influenza Division, presented an update on the recent Highly Pathogenic Avian Influenza (HAPI) A (H5N1) outbreak in dairy herds in the United States.
The USDA has confirmed HPAI A(H5N1) in U.S. dairy herds in 324 farms across 14 states, resulting in about 26 human cases.
On October 23, 2024, Dr. Shimabukuro concluded that the overall risk to the public for HPAI A(H5N1) remains low. However, exposed individuals (Michigan, Texas) should be monitored for (pink eye) symptoms after the first exposure and for 10 days and treated with approved influenza antivirals.
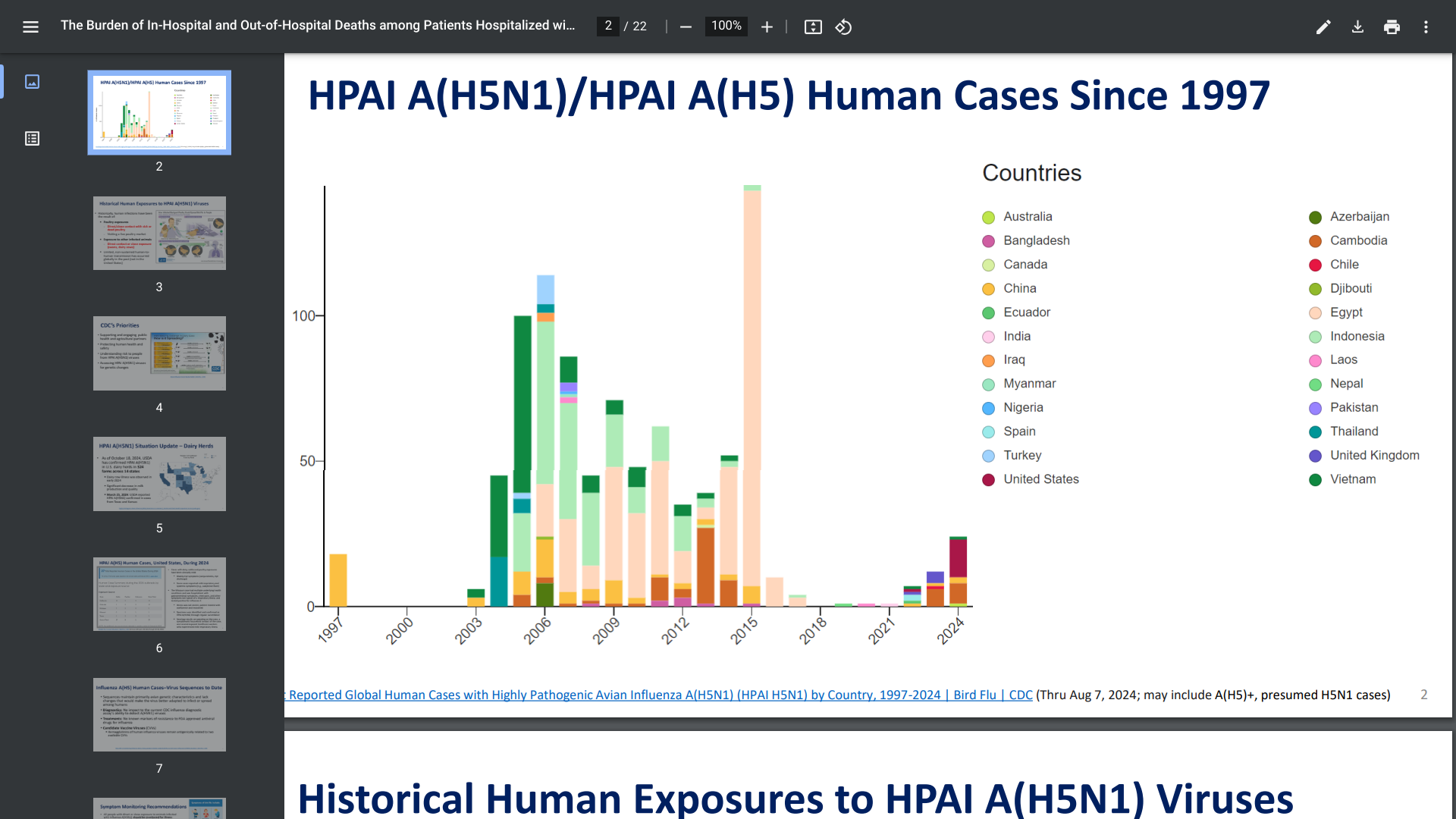
The CDC disclosed HPAI A(H5N1)/HPAI A(H5) human cases have been confirmed in numerous countries since 1997.
Furthermore, the CDC clarified that seasonal flu shots are not designed to protect people from 'Bird or Cow Flu' exposure.
If you plan to travel, you may need to be vaccinated against some diseases in other parts of the world. Some vaccines must be given well in advance to allow your body to develop immunity.
For some vaccines to become protective, several doses spread over several weeks or months.
According to the U.K. travel office on October 22, 2024, prospective international travelers should visit a G.P. or a private travel clinic at least 6 to 8 weeks before traveling bread in 2024.
In the U.K., the NHS routine immunization schedule lists vaccines that protect people against multiple diseases but does not cover all infectious diseases found overseas.
Furthermore, some countries require proof of vaccination (meningitis, polio, or yellow fever), which must be documented on an International Certificate of Vaccination or Prophylaxis (ICVP) before entering or leaving a country.
Even if an ICVP is not required, the U.K. recommends keeping a record of the vaccinations you have had with you, such as chikungunya, dengue, and measles.
In the United States, the CDC's travel advice website offers similar advice.

The U.S. CDC's Advisory Committee on Immunization Practices (ACIP) is scheduled to review scientific data and vote on vaccine recommendations on October 23-24, 2024.
Dr. Keipp Talbot, ACIP Chair, will lead this ACIP meeting from Atlanta, GA.
The final agenda includes presentations on chikungunya, HPV, influenza, pneumococcal, and RSV vaccines. These discussions are open to the public and available online via live webcast.
Several committee votes are planned during this meeting. The vote language shown at this link is considered draft language.
Furthermore, ACIP recommendations become official CDC policy once adopted by the CDC's Director.
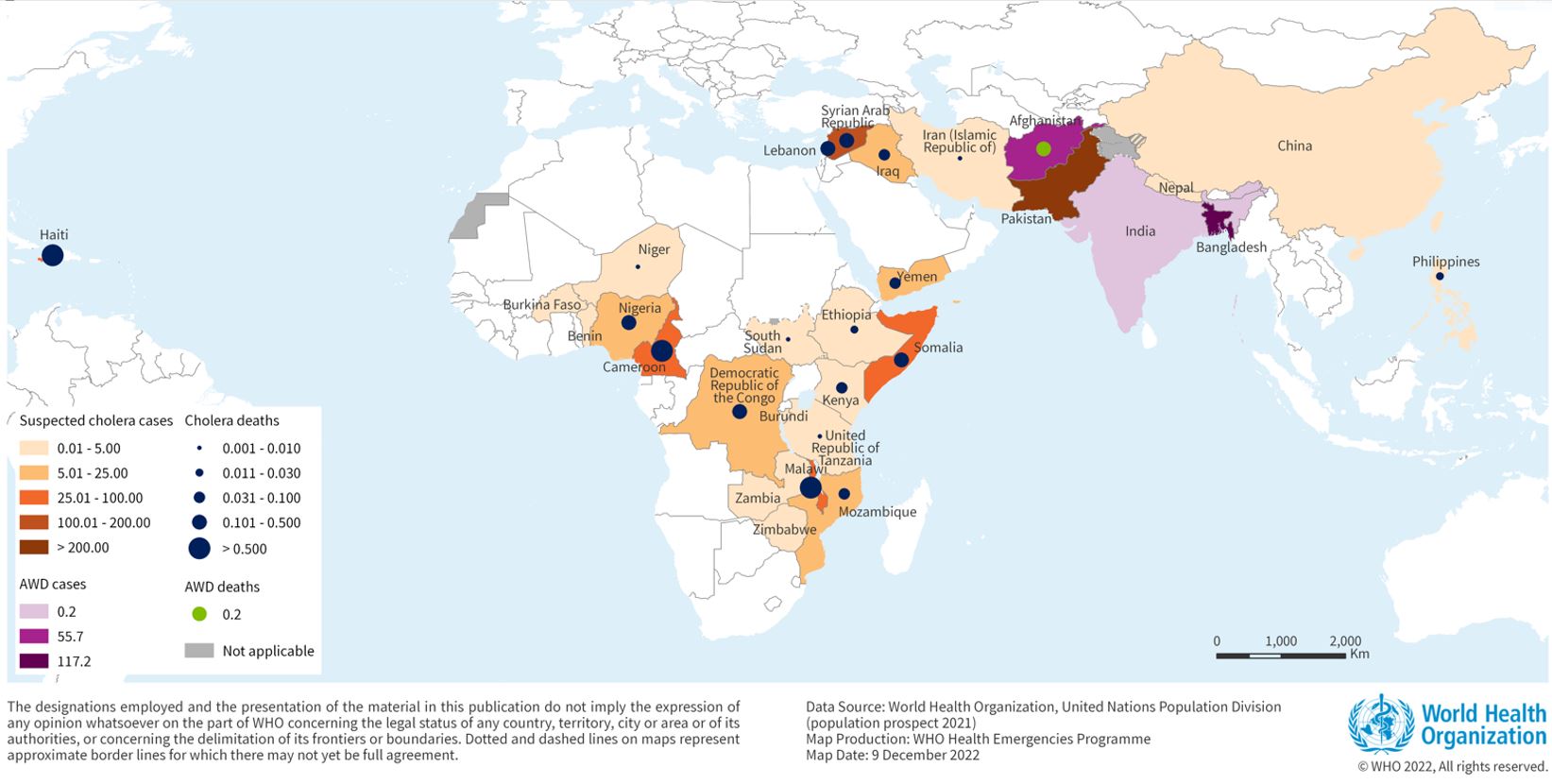
Since the last World Health Organization (WHO) report, new cholera outbreaks have been reported in Niger and Thailand, pushing the total number of affected countries in 2024 to 30.
As of the end of September 2024, a cumulative total of 439,724 cholera cases and 3,432 deaths were reported across five WHO regions.
Although the number of cases is lower than last year, the 126% spike in deaths is deeply concerning, says the WHO.
Unfortunately, the WHO has reported that as of October 14, 2024, the global stockpile of Oral Cholera Vaccine (OCV) is depleted, with no remaining doses available.
However, in the United States, travel vaccine clinics and pharmacies have confirmed supplies of OCVs.
With the global expansion of the chikungunya, dengue, and zika viruses in 2024, many publishers are challenged to present outbreak data in a balanced voice and tone in the media.
The lessons learned from the recent pandemic indicate that people want to read the data as soon as possible, whether published as a pre-print or peer-reviewed. With new information, people can privately confer with their healthcare providers about personal health actions such as vaccination.
A recent Editorial published by The Lancet Infectious Diseases on October 15, 2024, encapsulated these concerns regarding the sudden Oropouche virus outbreak in the Region of the Americas, such as in Brazil and Cuba.
These researchers wrote, "While such early data are key to understanding and tackling outbreaks, they come with considerable uncertainty and raise questions about how to report and publish them responsibly."
"However, we believe knowledge will inform public health responses, focus resources, and garner urgency in studying the virus further, which is the first step to prevention and treatment."
The full, unedited Editorial is posted at this link.

The steady progress towards a polio-free world has been successful for over three and a half decades. According to a recent announcement, the achievement of this goal has been delayed.
The Global Polio Eradication Initiative (GPEI) announced on October 17, 2024, that billions of children have been protected with lifesaving immunizations. More than 20 million people are walking who would otherwise have been paralyzed by this dreadful disease.
The polio program is now concentrated in some of the most complicated and fragile settings to deliver essential healthcare.
Recognizing these challenges, the GPEI’s Polio Oversight Board decided in July 2024 to extend the timelines needed to achieve polio eradication to the end of 2027 for wild poliovirus.
And the end of 2029 for type 2 variant poliovirus.
The Board's decision was informed by critical analysis and expert consultations, and extended timelines require additional financial resources to support ongoing eradication activities.
These shifts are being driven by partners' expertise and a program-wide commitment to double down on the toughest but most critical challenges standing between us and a future without polio, wrote the GPEI.
On October 17, 2024, the U.S. Centers for Disease Control and Prevention (CDC) reported poliovirus outbreaks that had caused 672 confirmed polio cases over the past year and a half.
The CDC also reissued a Global Travel Health Notice in August 2024 to alert international travelers, confirming 37 countries report poliovirus detections. People are encouraged to speak with a travel vaccine expert at least one month before visiting any of these countries about immunization options.

Blue Lake Biotechnology, Inc. and its affiliate CyanVac LLC today announced the presentation of positive interim pediatric clinical data on BLB201, Blue Lake’s vaccine candidate against respiratory syncytial virus (RSV).
Hong Jin, Ph.D., Chief Scientific Officer of Blue Lake and CyanVac, presented a talk that included results from an interim analysis of RSV-seropositive children participating in the company’s ongoing Phase 1/2a pediatric study.
The data demonstrated the safety and immunogenicity of BLB201 in this population, including RSV F-specific systemic and mucosal antibody responses and cell-mediated immune responses in CD4+ and CD8+ T cells.
“Our vaccine approach is designed to stimulate potent immune responses from the humoral, cellular, and mucosal pillars of the immune system,” said Biao He, Ph.D., CEO of Blue Lake and CyanVac, in a press release on October 21, 2024.
“Many currently available vaccine technologies such as mRNA and protein-based vaccines are ineffective in inducing mucosal immunity because they are given intramuscularly."
"By robustly stimulating all three pillars of immunity, our intranasal vaccine may prevent disease transmission and generate longer-lasting immunity than other types of vaccines.”