Search API
The U.S. CDC published a new Morbidity and Mortality Weekly Report (73(6);1–18) today that confirmed from the 2010–11 through 2022–23 influenza seasons, older adults (aged ≥65 years) consistently had the highest laboratory-confirmed influenza-associated hospitalization rates than other age groups.
The CDC wrote on October 31, 2024, that studies have demonstrated that vaccinated, hospitalized adult patients have a 26%–59% reduced risk of being admitted to the ICU and a 31% reduced risk of death compared with unvaccinated patients.
This data revealed many seniors were unprotected against influenza infection.
Adults aged ≥65 who were hospitalized with influenza consistently had the highest proportion of current-season influenza vaccine receipt (range = 40.9% during 2022–23 to 60.7% during 2018–19).
According to the CDC's Advisory Committee on Immunization Practices meeting last week, all persons should receive influenza vaccinations. Still, it is essential for those with underlying medical conditions to be vaccinated because of the elevated risk of complications from an influenza virus infection.
As of the end of October, about 100 million flu shots had been distributed to healthcare providers in the U.S. These vaccines are generally available at clinics and pharmacies.

A stay in a hospital might resolve one health challenge. Still, it may introduce another: an intractable infection with Clostridioides difficile (C difficile), wrote an Editors Summary published in the journal Science in October 2024.
This bacteria can thrive and produce toxins if antibiotics hinder the normal gut bacteria.
Alameh et al. disclosed they have been developing a multivalent mRNA–lipid nanoparticle vaccine to protect vulnerable individuals against toxigenic C. difficile. The University of Pennsylvania researchers designed the vaccine candidate to target an enzyme found in diverse strains of this bacterium that processes several surface factors required for gut colonization and virulence.
They concluded, 'Our studies demonstrate mRNA-LNP vaccine technology as a promising platform for developing novel C. difficile therapeutics with potential for limiting acute disease and promoting bacterial decolonization.'

The U.K. Health Security Agency (UKHSA) announced today that it had detected its first Clade Ib mpox human case in London, England.
As of October 30, 2024, the infected individual has been transferred to the Royal Free Hospital High Consequence Infectious Diseases unit.
This person had recently traveled to countries in Africa that are reporting Clade Ib mpox outbreaks.
The UKHSA and NHS stated that the risk to the U.K. population remains low and will not disclose further details about this case. Any contacts will be offered testing and vaccination as needed and advised on any necessary further care if they have symptoms or test positive.
This is the first detection of this Clade of mpox in the U.K. It differs from mpox Clade II, which has circulated at low levels in the U.K. since 2022.
U.K. Health and Social Care Secretary Wes Streeting commented in a press release, "The overall risk to the U.K. population currently remains low... (We) are securing vaccines and equipping healthcare professionals with the guidance and tools to respond to cases safely."
According to a recent World Health Organization analysis, Clade 1 viruses have been detected in Central Africa for decades.
In the United States, the CDC reported on October 19, 2024, that 2,230 Clade II cases were confirmed in 2024, compared with 1,096 reported in 2023.
In the U.S., Bavarian Nordic's JYNNEOS® (MVA-BN®, IMVAMUNE®, IMVANEX®) two-dose mpox/smallpox vaccine is commercially available at clinics and pharmacies. Vaccine efficacy data against Clade Ib is pending.

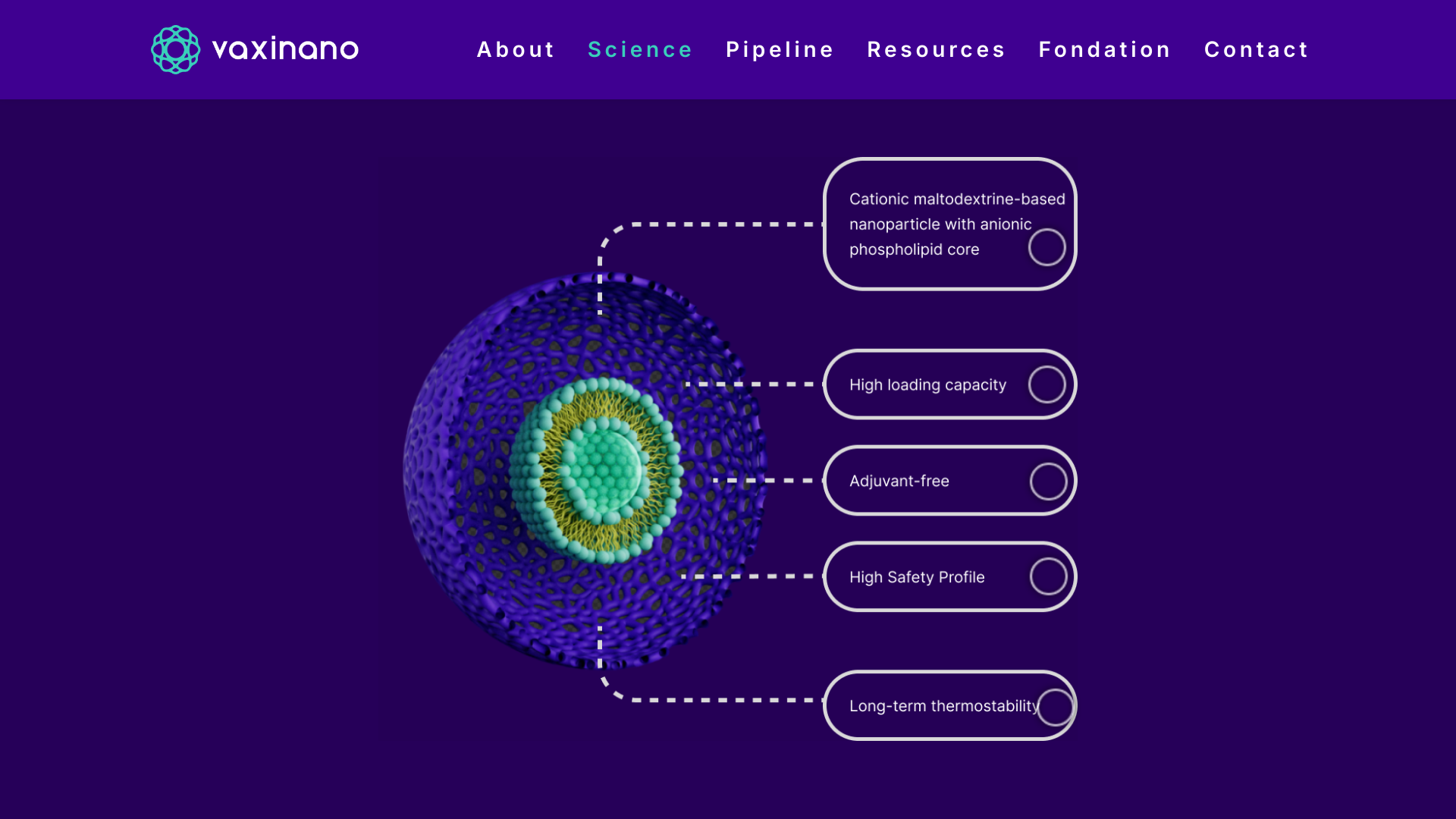
Vaxinano, a biotech company specializing in developing prophylactic and therapeutic nasal vaccines, has successfully raised €6 million in a funding round.
Thanks to its Stellar-NP nanoparticle technology, Vaxinano is developing a new generation of highly stable, adjuvant-free nasal vaccines designed to deliver effective and long-lasting responses to the most challenging pathogens, including parasites, viruses, and bacteria.
The platform has demonstrated preventive and therapeutic efficacy across multiple species and various infectious diseases, such as leishmaniasis, toxoplasmosis, and colibacillosis.
Didier Betbeder, founder and CSO of Vaxinano, stated in a press release on October 29, 2024, "The Stellar-NP technology has the potential to revolutionize the vaccine landscape by providing more effective and cost-efficient solutions in a context of emerging pandemics."
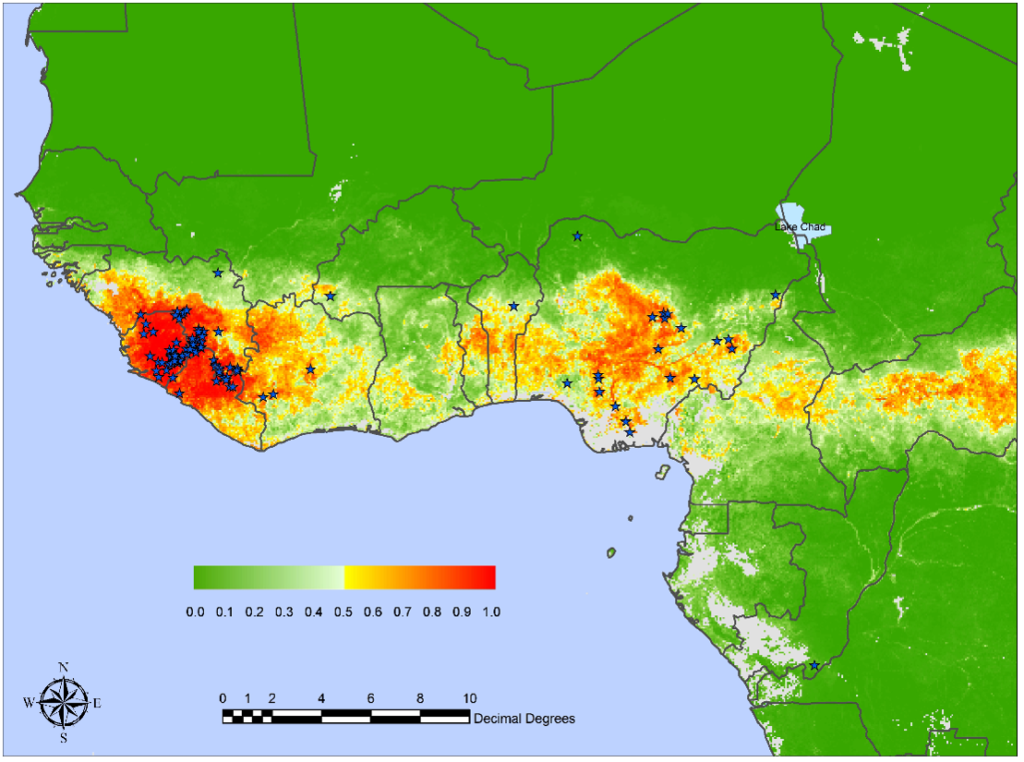
Malaria vaccines have been in clinical development since the 1960s, with substantial progress over the past few years. In October 2021, the first of two malaria vaccines was approved to prevent Plasmodium falciparum malaria in children living in regions with moderate to high transmission.
However, recent studies have found maternal antibodies passed to infants can interfere with the response to the malaria vaccine.
The lower antibody titers in infants were attributed to either co-administration with routine vaccines included in the WHO Expanded Programme on Immunization, maternal anti-CSP antibodies, immune status regarding previous exposure, the infant's immature immune system, or a combination of these factors.
Published on October 23, 2024, this observational study conducted in six African countries, researchers concluded that interference between passive immunity and vaccine response is clinically significant and might affect the implementation of next-generation CSP-based vaccines for young infants and mothers and passive immunization with human monoclonal antibodies.
To validate this conclusion, additional clinical studies are being conducted.
As of late October 2024, malaria vaccines are offered in Africa, not the United States.
Bavarian Nordic A/S today announced the initiation of a clinical study of the MVA-BN® (JYNNEOS) mpox/smallpox vaccine in children 2 to 11 years of age, partially funded with $6.5 million from the Coalition for Epidemic Preparedness Innovations (CEPI).
The phase 2 study is currently enrolling children in the Democratic Republic of Congo, with plans to include sites in Uganda. Results from this study could support an extension of the current approval of MVA-BN to include young children.
Last month, the WHO prequalified MVA-BN for adolescents 12 to 17 years of age, adopting the recent approval from the European Medicines Agency (EMA) for this age group.
While this study represents the first investigation of MVA-BN as a mpox/smallpox vaccine for younger children, a recombinant version of MVA-BN (Mvabea®) was approved by EMA in 2020 as part of a prime-boost vaccine regimen or the prevention of disease caused by Zaire Ebolavirus.
Paul Chaplin, President and CEO of Bavarian Nordic, said in a press release, “Following the recent approval of MVA-BN for adolescents, we are pleased to initiate this study, which could provide additional data to extend the indication to include children. We thank CEPI and our partners in Africa for their support of this important work.”
From an access perspective, Bavarian Nordic announced an agreement with UNICEF on September 26, 2024, to supply 1 million doses of the MVA-BN® mpox vaccine for African countries impacted by the ongoing mpox outbreak.
As of late October 2024, there have been about 2,230 clade 2 mpox cases reported to the U.S. CDC this year.
The JYNNEOS vaccine is commercially available in the United States at various clinics and pharmacies.

Sandra Levy, senior editor of Drug Store News, conducted a digital conversation with Tim Ducharme, vice president of Immunization Growth and Strategy at CVS Health®.
This discussion, posted on YouTube on October 28, 2024, centered around the company’s vaccination business and its offerings for the respiratory virus season, and how it is working with patients to help them schedule any immunizations ahead of time.
Ducharme highlighted three areas of focus: appointment scheduling, vaccine availability, patient access issues and opportunities.
He also discusses how CVS is working to educate patients about the 2024-2025 respiratory season, which includes RSV and influenza. Various vaccines are available for these diseases.
Product delivery, available through the more than 9,000 CVS Pharmacy locations across the U.S., is a core component of the company’s digital offerings and efforts to meet consumers’ unique health and wellness needs.
The Iowa Department of Health and Human Services (IDHHS) today announced the death of an Iowa resident from Lassa fever, a rare, often fatal, viral hemorrhagic fever.
There have been eight travel-associated cases of Lassa fever in the U.S. in the past 55 years.
If confirmed, the Iowa case would be the ninth known occurrence of travelers returning to the U.S. since 1969.
This individual had recently returned from travel to West Africa, where ISHHS believes the person contracted the virus. About 5,000 Lassa fever-related deaths occur in West Africa each year.
Dr. Robert Kruse, State Medical Director of the IDHHS, stated in a press release on October 28, 2024, "I want to assure Iowans that the risk of transmission is incredibly low in our state. We continue investigating and monitoring this situation and are implementing the necessary public health protocols."
Lassa fever is carried by rodents and is transmitted to humans who may come in contact with the urine or feces of the infected rodents. Approximately 80% of Lassa fever patients have mild or no symptoms.
The U.S. Centers for Disease Control and Prevention posted a statement emphasizing that the overall risk to the public from this case is very low.
Lassa is included in the World Health Organization's R&D Blueprint of priority pathogens, for which there is an urgent need for accelerated research, vaccine development, and countermeasures.
As of October 2024, several Lassa fever vaccine candidates are conducting research, but the U.S. FDA has approved none.