Search API
Uvax Bio, LLC today announced the interim analysis results from its first Phase 1 clinical trial evaluating the Company’s HIV-1 vaccine candidates, UVAX-1107 and UVAX-1197.
In the first stage of this trial, the subjects received either UVAX-1107 adjuvanted with CpG 1018® and aluminum hydroxide or placebo.
UVAX-1107 was immunogenic and generated robust IgG responses to the vaccine antigen derived from an HIV-1 strain known as BG505. 100% of subjects in the vaccine group demonstrated antibody responses after two priming vaccinations with UVAX-1107.
Antibody response titers increased >200-fold 14 days after the 2nd dose compared to the same period following the first dose.
“We are pleased that our first Phase 1 trial is progressing smoothly, and we have preliminary confirmation that UVAX-1107 was well tolerated at all doses by the study participants, and no vaccine-related serious adverse events were reported,” said Pedro Garbes, M.D., Vice President and Global Clinical Lead of Uvax Bio, in a press release on November 19, 2024.
“As per the time of this 1st interim analysis, no participant was withdrawn from the study due to local/systemic reactogenicity; local and systemic adverse events were mild to moderate, transient, and resolved on average within two days. These preliminary safety results are aligned with expectations for an adjuvanted protein-based vaccine.”
UVAX-1107 utilizes Uvax Bio’s 1c-SApNP® vaccine development platform to generate virus-like particles that closely resemble the target virus in size, shape, and multivalent antigen display; in this case, 20 copies of the native-like, prefusion-stabilized trimeric HIV-1 antigen.

The World Health Organization (WHO) today announced it has granted Emergency Use Listing (EUL) for the LC16m8 smallpox / mpox vaccine, making it the second mpox vaccine to be supported by WHO.
LC16m8 is a vaccine developed and manufactured by KM Biologics in Japan.
It is recommended by the WHO for use in individuals over one year of age as a single-dose vaccine via a multiple puncture technique using a bifurcated needle.
“WHO emergency use listing of the LC16m8 vaccine against mpox marks a significant step in our response to the current emergency, providing a new option to protect all populations, including children,” commented Dr. Yukiko Nakatani, WHO Assistant Director-General for Access to Medicines and Health Products, in a press release on November 19, 2024.
“Vaccines are one of the important tools to help contain the outbreak as part of a comprehensive response strategy that also includes improved testing and diagnosis, treatment and care, infection prevention control, and engagement and education within affected communities.”
This decision is expected to facilitate increased and timely vaccine access in communities where mpox outbreaks are surging.
Today’s move is particularly relevant as the Government of Japan has announced that it will donate 3.05 million doses of the LC16m8 vaccine and specialized inoculation needles to the Democratic Republic of the Congo. This is the most extensive vaccine donation package announced to date in response to the current mpox emergency.
Previously, the WHO has LIsted Bavarian Nordic A/S JYNNEOS® (MVA-BN®) smallpox / mpox vaccine.

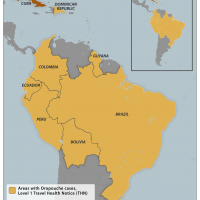
When searching for a winter holiday destination in 2024, Bermuda may top the healthy destination list, as no vaccination requirements are currently in effect.
This good news helped boost Bermuda's tourism industry in 2023 when arrivals (air, cruise, and yacht) increased by 29.1% year over year.
As of November 18, 2024, this British Overseas Territory, home to about 63,000 people and in the middle of the Atlantic Ocean, has the lowest travel advisory status with the U.S. and U.K. governments.
The U.K. Travel Health Pro suggests visitors be up to date with routine vaccination courses and boosters, as recommended in the U.K. These vaccinations include the measles-mumps-rubella (MMR) vaccine.
And the U.S. CDC's Travel Health Advisory recommends most travelers, especially those staying with friends or relatives or visiting smaller cities or rural areas, are up to date with typhoid vaccination.
Furthermore, from a security perspective, the U.S. Department of State reissued Bermuda's Level 1 travel advisory status on November 12, 2024. Visitors should exercise standard precautions and enroll in the Smart Traveler Enrollment Program to receive Alerts, making locating you in an emergency while in Bermuda easier.
The U.S. Consulate General Hamilton is located at 16 Middle Road, Devonshire, DV 03, and can assist U.S. citizens in Bermuda.

The California Department of Public Health (CDPH) today announced it had identified the first known case of clade I mpox in the United States. This case was confirmed in an individual who recently traveled from Africa and is related to the ongoing outbreak of clade I mpox in Central and Eastern Africa. Historically, clade I has caused more severe illness than clade II.
The clade II outbreak began worldwide in May 2022, reaching Boston in late May.
As of November 9, 2024, 2,410 clade II cases have been confirmed in the U.S. this year. This is a significant increase compared with last year, when 1,267 mpox cases were reported.
The CDPH stated on November 16, 2024, the affected individual received health care in San Mateo County. The individual is isolating at home and recovering. Public health workers are contacting people who had close contact with this individual.
According to the U.S. CDC, there is no concern or evidence that mpox clade I is spreading between individuals in California or other states.
However, the World Health Organization declared the increasing number of mpox cases reported in the African Region, notably the Democratic Republic of the Congo, a Public Health Emergency of International Concern on August 14, 2024.
Bavarian Nordic's JYNNEOS® (MVA-BN®) mpox/smallpox vaccine has been effective against clade II and is commercially available at clinics and pharmacies in the U.S.

Hookworms infect about half a billion people worldwide and cause more than two billion disability-adjusted life years to be lost. In a recent review, researchers wrote that mass drug administration is the most popular preventive approach but does not prevent reinfection.
The U.S. CDC says people get hookworm infection by walking barefoot on or consuming contaminated soil and passing the parasitic worm's eggs in their feces. And zoonotic hookworms are parasites generally found in animals like dogs or cats. Furthermore, they can also spread to humans through the skin.
This review, published by Talor and Francis online, highlights recent human studies in which vaccination with irradiated larvae and repeated rounds of infection treatment have induced partial protection.
On November 15, 2024, these researchers stated, 'An effective vaccine would be a major public health tool in hookworm-endemic areas.'
'These studies have emphasized the importance of targeting the infective larvae to generate immunity and prevent adult worms from maturing in the gut.'
In summary, these researchers wrote 'that significant recent advances in human challenge vaccine studies should accelerate the process.'

Bavarian Nordic A/S (OMX: BAVA) announced today it's interim financial results and business progress for the third quarter of 2024, highlighting mpox vaccine sales.
Bavarian Nordic stated on November 15, 2024, that it retains sufficient capacity to fulfill the requirement as stated by Africa CDC for at least 10 million doses of MVA-BN® (JYNNEOS®) by the end of 2025 and could even further scale up capacity for future demand if needed.
For 2025, the Company has so far secured revenue from mpox/smallpox vaccine orders of approximately DKK 2,400 million.
Paul Chaplin, President & Chief Executive Officer of Bavarian Nordic, said in a press release, “We have had a busy but highly purposeful and rewarding third quarter, where we have worked intensively to support the efforts to curb the ongoing mpox outbreak in Africa."
In the United States, the government recommends two doses of JYNNEOS to provide more robust protection. However, routine immunization against mpox is not recommended for the general public. The U.S. CDC does not endorse booster doses (3rd).