Search API
As the February 2026 vaccination plans are being formed, the UK Health Security Agency (UKHSA) has issued an important reminder for travelers heading to Cape Verde to take extra precautions against gastrointestinal infections when visiting the West African archipelago.
As of February 5, 2026, recent UKHSA data indicate a significant cluster of Shigella sonnei cases, a bacterial cause of severe dysentery-like illness, along with multiple clusters of non-typhoidal Salmonella infections strongly linked to recent travel to Cape Verde.
Since October 1, 2025, the UKHSA has confirmed 158 cases of Shigella sonnei in travelers returning to England, Scotland, and Wales. Of the 118 cases where international travel was reported, 112 (94.9%).
Many travelers to Cape Verde have visited the Santa Maria area on Sal Island and the Boa Vista area, popular resorts known for their beaches.
The UKHSA says this strain of Shigella sonnei shows no genomic resistance to the common antimicrobials typically used to treat traveler's diarrhea, which could facilitate treatment if an infection occurs.
Additionally, 32 cases were linked to travel to Cape Verde during the same period.
Investigations into the outbreaks are ongoing and involve international health partners. Cases have also been reported in other European countries, particularly from September to November 2025.
Shigella and Salmonella are primarily transmitted through contaminated food and water, poor hygiene practices, and person-to-person contact. The risk of infection is especially heightened in resort settings.
Symptoms typically appear within a few days, and while most people recover within a week with supportive care (with hydration being crucial), severe cases can lead to dehydration, hospitalization, or complications. This is particularly concerning for vulnerable groups such as young children, the elderly, and individuals with weakened immune systems.
The UKHSA emphasizes that while Cape Verde is a popular travel destination, increased vigilance can help prevent illness and ensure a safe holiday. Travelers who experience symptoms upon returning should seek medical advice and inform their doctor about their travel history.
From a prevention standpoint, no Shigella vaccine has received regulatory approval from any health authority as of February 2026.
However, the Shigella4V (S4V or S4V2) vaccine candidate received U.S. FDA Fast Track designation in 2024. Phase 2 studies (including in infants) are underway, but no Phase 3 completion or approval has been achieved yet.
In a recent update from the U.S. Centers for Disease Control and Prevention (CDC), details were released about a gastrointestinal illness (GI) outbreak aboard the Regent Seven Seas cruise ship, Seven Seas Mariner.
The CDC reported this incident on February 2, 2026, which affected a small percentage of passengers and crew, underscoring the ongoing challenges of maintaining health standards on luxury cruises in the travel environment.
According to the CDC's Vessel Sanitation Program (VSP), the final case counts showed that 21 of 631 passengers (3.3%) reported illness, and 6 of 458 crew members (1.3%).
The predominant symptom among those affected was diarrhea, and the specific causative agent remained unknown despite testing.
In response to the outbreak, Regent Seven Seas and the ship's crew implemented several measures as outlined in the CDC report. These included enhancing cleaning and disinfection procedures in accordance with their established outbreak prevention and response plan. Stool specimens were collected from affected individuals for laboratory testing, and ill passengers and crew members were isolated to prevent further spread.
The cruise line consulted directly with VSP officials to enhance sanitation protocols and improve case reporting.
Regent Seven Seas has consistently received high scores from the VSP, often exceeding 95 out of 100 in routine inspections. This strong track record may have helped in the swift containment of the recent outbreak.
The CDC says GI outbreaks are common in close-quarters environments, such as cruise ships, and are often linked to norovirus or other pathogens. However, the relatively low infection rate in this case indicates that the situation was effectively managed.
In 2025, there were 23 reported outbreaks, of which 17 were attributed to norovirus. This virus is the most common cause of confirmed GI outbreaks on cruise ships because it spreads easily in tight spaces.
For those planning cruise ship voyages in 2026, Vax-Before-Travel.com reports that no vaccine is currently available for norovirus.

Measles is an ongoing risk around the world, and children are at serious risk for the highly transmissible disease in most countries. Since 2023, measles incidence has been rising in many countries worldwide, including in Europe.
The Israeli Ministry of Health recently reported the 14th pediatric fatality due to measles since an outbreak began in mid-April 2025, partially from imported cases and others of unknown origin.
The Ministry's announcement on February 2, 2026, confirmed that the affected individual was a one-year-old who had not been vaccinated. The baby was brought to Hadassah Mount Scopus Hospital in Jerusalem in critical condition.
Similar to previous cases, most of the deceased were healthy infants without any underlying conditions and had also not received vaccinations.
The Ministry emphasizes that measles is a preventable disease and advises that all children should receive their first vaccine dose at the age of one. In areas affected by the outbreak in Israel, the Health Ministry recommends administering the second dose of the measles vaccine at 18 months.
Additionally, an extra vaccine dose is recommended for infants aged 6 to 11 months in outbreak areas and for those traveling to outbreak areas.
As of February 5, 2026, the areas in Israel that have been classified as experiencing a measles outbreak are Jerusalem, Beit Shemesh, Bnei Brak, Harish, Modi'in Illit, Nof HaGalil, Kiryat Gat, Ashdod, Safed, Netivot, Haifa, Tiberias, the Mateh Binyamin Regional Council, and the settlement of Tekoa.
To alert international travelers to their health risk, the U.S. CDC has maintained a Level 1, Travel Health Notice, that identifies measles outbreaks in numerious countries. The CDC advises all travelers to speak with a local travel vaccine expert about measles immunization options and other vaccine-preventable diseases.
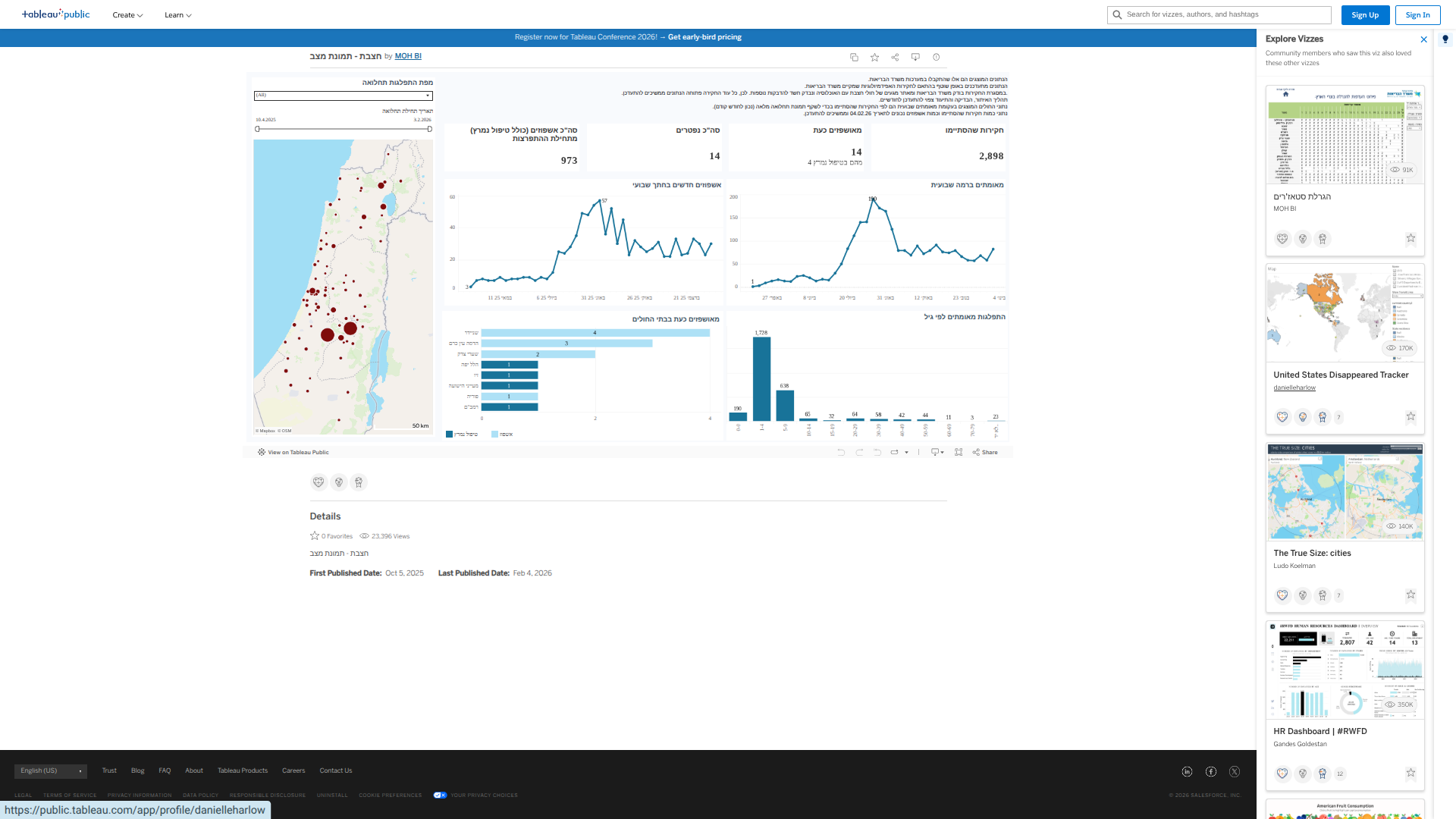
The Florida Department of Health (DOH) today announced updated surveillance data on chikungunya fever cases in the state.
As of February 4, 2026, the data emphasize the ongoing risks associated with travel to chikungunya-endemic areas, particularly Cuba, as well as the rare but noteworthy occurrence of local transmission in Florida.
As of the latest reporting period (#4) in 2026, nine cases have been confirmed among individuals with travel history to chikungunya-endemic areas within two weeks before onset, all linked to travel to Cuba.
And one case of locally acquired chikungunya fever was reported in Miami-Dade County, with symptom onset in December 2025.
In 2025, a total of 350 chikungunya cases were reported among individuals who had recently traveled to chikungunya-endemic areas. These cases were distributed across multiple Florida counties, with Miami-Dade County reporting the highest number at 229.
Chikungunya fever is a viral illness primarily transmitted by Aedes mosquitoes. Symptoms include high fever, severe joint pain, muscle pain, headache, nausea, fatigue, and rash. Most individuals recover completely; however, joint pain can be debilitating and may persist for months in some cases. There is no specific antiviral treatment, but supportive care can help relieve symptoms.
The DOH and the U.S. CDC advise travelers to endemic areas to consult local healthcare clinics about preventive strategies, including vaccination options. In Florida and most states, U.S. FDA-approved chikungunya vaccines are available in 2026.
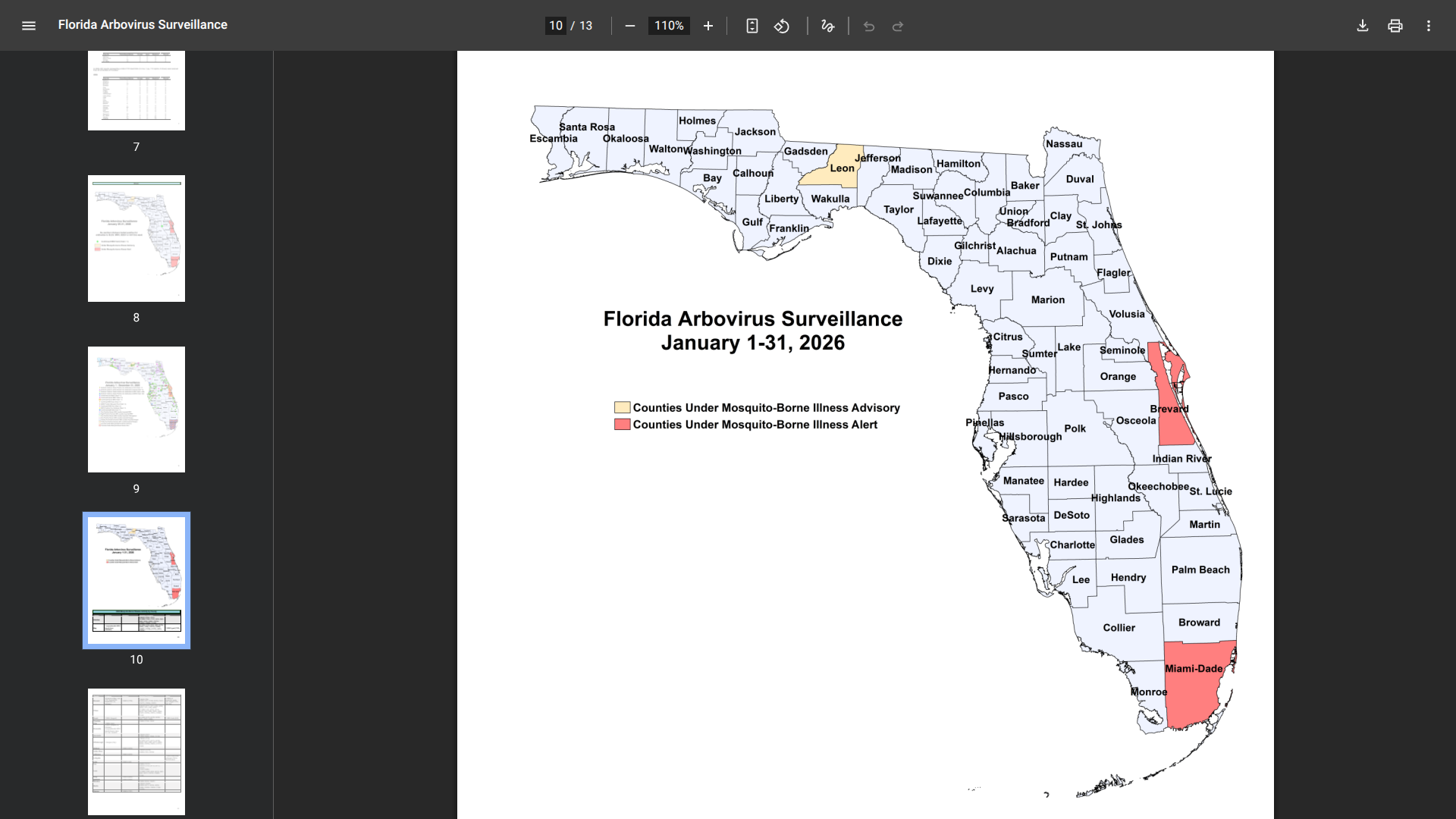
In the past year, the World Health Organization (WHO) reported over 600,000 cases of cholera or acute watery diarrhea and nearly 7,600 deaths across 33 countries.
However, these figures are likely underreported, as cholera cases often go unrecorded.
Today, Gavi, the Vaccine Alliance, UNICEF, and the WHO announced that the global supply of oral cholera vaccine (OCV) has increased sufficiently to resume life-saving preventive vaccination campaigns for the first time in over three years.
A first allocation of 20 million doses is being deployed for preventive campaigns.
Of these, 3.6 million doses were delivered to Mozambique; 6.1 million to the Democratic Republic of the Congo, which is also experiencing significant outbreaks; and 10.3 million doses are planned for delivery to Bangladesh.
"Global vaccine shortages forced us into a cycle of reacting to cholera outbreaks instead of preventing them. We are now in a stronger position to break that cycle. I thank EUBiologics, currently the only manufacturer producing cholera vaccines at the scale needed for mass vaccination campaigns, for its efforts, and urge others to enter this vital space. These vaccines will save lives," said Dr Tedros Adhanom Ghebreyesus, WHO Director-General, in a press release on February 4, 2026.
Over the last few years, the annual global supply of OCV has doubled from 35 million doses in 2022 to nearly 70 million doses in 2025. The doses are being financed by Gavi and procured and delivered to countries by UNICEF.
"The multi-year surge in cholera cases and resulting unprecedented demand for vaccines were stark reminders that sustainable, accessible vaccine supply is a global public good – and the world cannot afford complacency," added Dr Sania Nishtar, CEO of Gavi, the Vaccine Alliance.
"For the first time in years, this increase in vaccines will allow us to prevent large-scale cholera emergencies better," said Catherine Russell, UNICEF Executive Director. "Resuming preventive cholera vaccination will protect children and help stop this highly contagious disease in its tracks. But it must go hand in hand with other efforts, including better access to safe water and basic sanitation."
While global vaccine supply steadily improves, the one-dose strategy will remain the standard for outbreak responses, with the use of two doses considered on a case-by-case basis.
Cholera spreads through contaminated food and water, causing severe diarrhoea and dehydration. It can lead to death if it is not treated quickly. It is found in places without safe water and sanitation, mainly in localities affected by conflict and poverty.
The WHO says vaccination is only one aspect of cholera prevention and response. Long-term investments in safe water, sanitation, and hygiene infrastructure, alongside disease surveillance, rapid treatment, and community engagement, remain essential to prevent outbreaks from starting and spreading, and to reduce deaths in the long term.
In the United States, OCVs are offered at travel vaccine clinics, located in every state.
The Republic of Paraguay's Ministry of Public Health and Social Welfare announced today that the Dengue vaccination strategy, initially targeted at children in priority municipalities, is now being expanded to include individuals aged 39 and younger.
Announced on February 3, 2026, this decision is based on an epidemiological analysis conducted over recent years, which indicates a significant burden of Dengue not only among children but also among adolescents and young adults.
Between 2019 and 2024, the 20 to 39-year-old age group accounted for more than 108,000 Dengue cases in this South American country, reflecting one of the highest incidence rates recorded.
These findings support the extension of the second-generation, two-dose, Qdenga (TAK-003) vaccine, which protects against all four serotypes of the Dengue virus.
Paraguay says vaccination is a critical component of a comprehensive strategy that includes epidemiological surveillance and preventive measures to mitigate the impact of Dengue in the country.
Paraguay's neighbor, Brazil, recently announced it would focus on a newly approved third-generation, single-dose Butantan-DV vaccine for its residents.
Currently, Dengue vaccines are unavailable in the continental United States.
As of February 2026, the U.S. Food and Drug Administration (FDA) has not yet approved any vaccine for the herpes simplex virus (HSV). However, several candidates are currently undergoing clinical trials.
These efforts aim to tackle the global impact of HSV-1 and HSV-2 infections, which cause oral and genital herpes in millions of people annually.
Unfortunately, the FDA states that there is currently no cure for HSV infections.
However, antiviral medications can help shorten the duration of herpes outbreaks. A significant advancement in HSV treatment is currently attracting attention.
AiCuris Anti-infective Cures AG is set to present detailed results from its pivotal Phase 3 clinical trial of pritelivir as a late-breaking oral presentation at the Tandem Meetings 2026, held February 4-7 in Salt Lake City, Utah.
Pritelivir, a novel oral helicase-primase inhibitor, targets both HSV-1 and HSV-2 by blocking viral DNA synthesis through a mechanism distinct from traditional nucleoside analogs like acyclovir or valacyclovir.
This unique mode of action makes it effective against strains resistant to current standard therapies, addressing a critical unmet need—particularly in immunocompromised patients, where resistant or refractory HSV infections can lead to severe complications and limited treatment options.
The PRIOH-1 trial focused on immunocompromised individuals with acyclovir-refractory (and sometimes foscarnet-resistant) mucocutaneous HSV infections. In October 2025, AiCuris announced that pritelivir met its primary endpoint, demonstrating statistically significant superiority in lesion healing compared to the investigator's choice of standard-of-care treatments (such as foscarnet, cidofovir, or topical options).
Superiority was shown for treatment up to 28 days (p=0.0047), with even stronger results extending to 42 days (p<0.0001).
Earlier Phase 2 data supported a favorable safety profile, and pritelivir has held an FDA Breakthrough Therapy designation for this indication since 2020. AiCuris expects to file for FDA marketing authorization in 2026, potentially offering the first new HSV therapy in decades that is active against resistant strains.
Full abstracts will be released on February 4, 2026, and will be available via the conference website here.
This development highlights progress in managing HSV complications in high-risk groups, even as preventive vaccines remain further on the horizon. Ongoing research and presentations like this at major conferences continue to drive hope for improved options against this widespread virus.

Three months after Hurricane Melissa made landfall in Jamaica on October 28, 2025, as the strongest Category 5 storm ever to hit the Caribbean island, the country's health system is still in the process of recovery.
The aftermath has resulted in widespread damage and ongoing public health risks.
In this interview, published on February 2, 2026, Ian Stein, the Pan American Health Organization/World Health Organization country representative in Jamaica, shares insights into the realities of recovery, lessons learned, and what success looks like in a complex emergency.
As of 2026, the U.S. CDC website states that, in the wake of Hurricane Melissa, travel by land may be dangerous in flood zones, and the healthcare infrastructure has been damaged in these areas.
There may be an increased risk of waterborne (including leptospirosis), vector-borne, and fungal diseases. And avoid contact with floodwater.
Furthermore, before visiting Jamaica in 2026, check the list of recommended vaccines and medications and see your travel health provider at least a month before your trip to get any you may need.
From a security perspective, the U.S. Department of State lowered its advisory for Jamaica to Level 2: Exercise Increased Caution on January 17, 2026, citing crime, health, and natural-disaster risks.
The State Department advisory clarifies that some areas have an increased risk. The U.S. advises against traveling to these areas for any reason.