Search API
The U.S. Department of State recently updated its travel advisory for the Plurinational State of Bolivia in western-central South America.
On January 26, 2023, the State Department's Level 2: Exercise Increased Caution notice highlighted areas of concern.
For the Chapare region, do not travel due to civil unrest. Also called The Chapare, this rural province is in the northern part of the Cochabamba Department in central Bolivia.
Chapare is adjacent to Peru, which also reported civil unrest in 2023.
The State Department also suggested reconsidering traveling to the Yungas region, which is located further north in Bolivia.
If you visit Bolivia or are in-country, the State Department says to avoid demonstrations and crowds and enroll in STEP to receive embassy alerts and to be located in an emergency.
And, due to ongoing public demonstrations in the southern city of Santa Cruz, the U.S. Consular Agency announced on January 4, 2023, it would remain closed until further notice.
And recommended visitors should limit travel to and around Santa Cruz.
American citizens in Santa Cruz needing urgent consular assistance can contact the Consular Section at the U.S. Embassy La Paz, located at Avenida Arce 2780, La Paz, Bolivia.
At over 3,500m elevation, La Paz is positioned in the Andes' Altiplano plateau near the snow-capped Mt. Illimani, with the world's highest cable car. Or visitors can visit Lake Titicaca, the world's highest navigable lake.
Furthermore, Canada confirmed that roadblocks are common on January 26, 2023, and can lead to significant traffic and public transportation disruptions. The following areas are vulnerable to blockades: along Bolivia–Peru border and roads leading to international airports.
From a health perspective, the U.S. Centers for Disease Control and Prevention (CDC) suggests several pre-visit vaccinations, such as yellow fever.
Yellow fever vaccination is recommended for all travelers ≥9 months of age traveling to areas in elevation and east of the Andes Mountains: the entire departments of Beni, Pando, Santa Cruz, and designated areas of Chuquisaca, Cochabamba, La Paz, and Tarija departments.
In the U.S., the YF-Vax vaccine is available at certified travel clinics and pharmacies.
The article was updated on Jan. 28, 2023.

Roche and its subsidiary TIB Molbiol, recently announced they had developed a COVID-19 PCR test for researchers that detects and differentiates the latest variant of concern, XBB.1.5.
The XBB.1.5 variant is prevalent in the U.S. and is quickly spreading to other countries.
XBB.1.5 is a sub-variant of a SARS-CoV-2 beta coronavirus variant named XBB.
Differentiating emerging variants and understanding their similarities and mutations empowers experts to make predictions about treatment strategies.
The VirSNiP SARS-CoV-2 Spike F486P test is for use on the LightCycler® 480 II and cobas® z 480.
“Roche continues to develop timely diagnostic innovations related to COVID-19 by providing valuable insights - helping scientists and physicians understand this new strain, how it differs from other variants, and the impact it may have on public health,” said Matt Sause, CEO of Roche Diagnostics, in a press release on January 26, 2023.
“Since the start of this global health crisis, Roche has been committed to bringing effective diagnostic solutions to address unmet needs to healthcare communities worldwide.”
The research-use-only test, VirSNiP SARS-CoV-2 Spike F486P, adds to the broad suite of COVID-19 test kits developed by Roche and TIB Molbiol.
In the U.S., the Food and Drug Administration has authorized numerous COVID-19-related tests as of January 27, 2023.

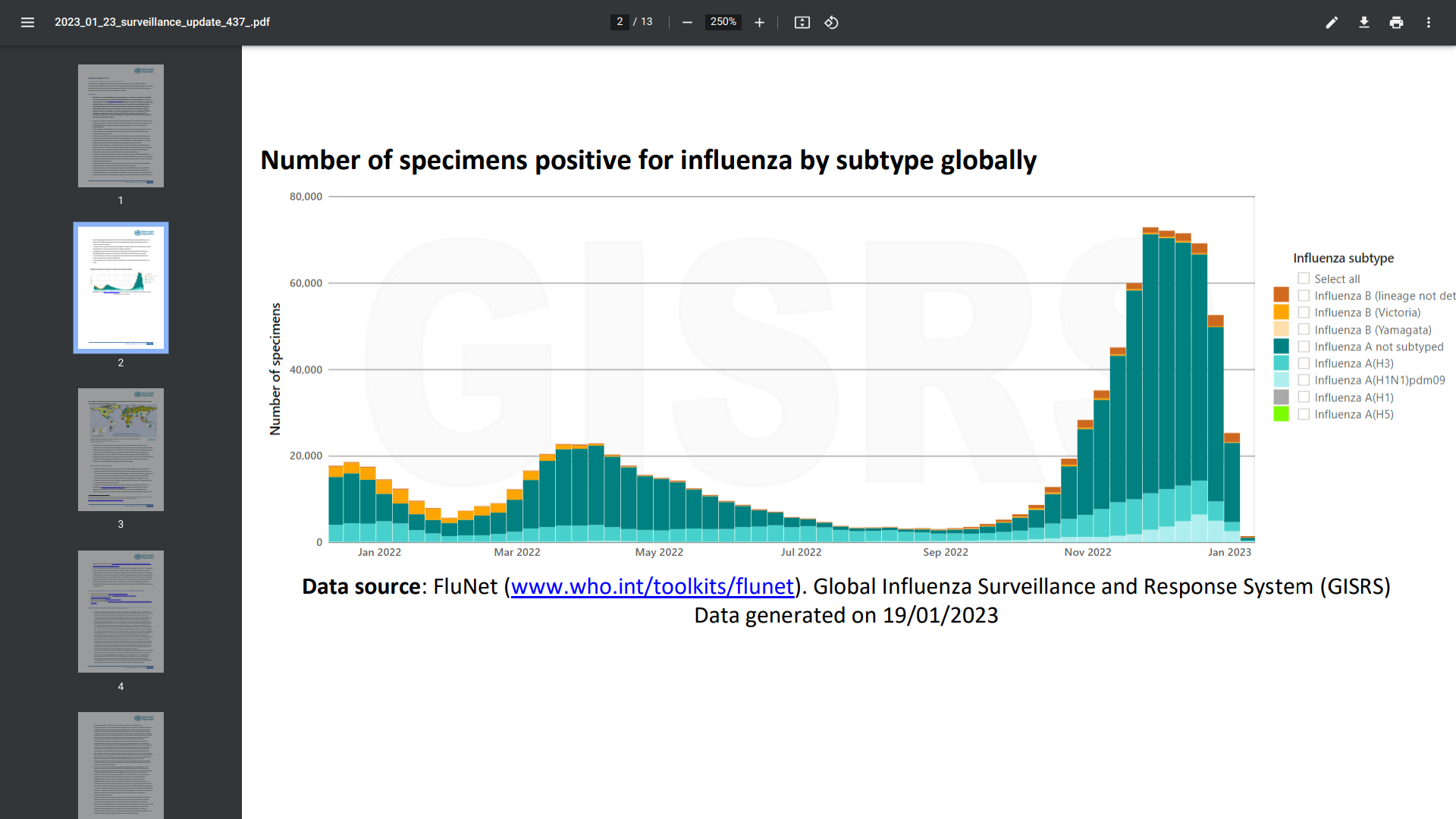
The World Health Organization (WHO) Influenza Update N° 437 report recently confirmed influenza activity decreased globally but remained somewhat elevated due to activity in the northern hemisphere.
In the countries of North America, most indicators of influenza activity decreased to levels similar to or below levels typically observed this time of year.
Influenza A(H3N2) was the predominant virus detected.
In Europe, overall influenza activity decreased.
Many European countries reported high or very high intensity, and more than half reported widespread activity. And various indicators of influenza activity decreased in many countries while others reported increases.
Overall, influenza A viruses predominated with similar proportions of A(H1N1)pdm09 and A(H3N2) influenza viruses detected in primary care sentinel sites but with regional differences.
The full, unedited WHO January 23, 2023 report is posted at this WHO link.
On January 20, 2023, the U.S. CDC FluView week #2 report confirmed seasonal influenza activity was declining in most areas.
Three regions in the U.S. were below their outpatient respiratory illness baselines for the first time since October 2022.
In the U.S., over 170 million influenza vaccines have been distributed during the 2022-2023 flu season. These flu shots remain generally available at most clinics and pharmacies in the U.S.
The U.S. Centers for Disease Control and Prevention (CDC) Travelers’ Health today announced the expansion of the Traveler Genomic Surveillance (TGS) program to two additional U.S. airports, Los Angeles and Seattle.
This expansion is helping the CDC to detect new SARS-CoV-2 coronavirus variants among international air travelers.
U.S. airports are visited by more than 1 billion travelers annually and can serve as the front line for public health officials to detect the virus that causes COVID-19.
Recently, the TGS program was among the first globally to identify BQ.1.1 and contributed towards its designation as a sub-lineage.
TGS also has been among the first to identify and report BA.2.75.2, XBB, and CH.1.1 in the U.S.
In total, the TGS program currently has sites in seven major U.S. international airports:
Hartsfield-Jackson International Airport in Atlanta
John F. Kennedy International Airport in New York City
Los Angeles International Airport
Newark Liberty International Airport
San Francisco International Airport
Seattle-Tacoma International Airport
Washington Dulles International Airport
The next time you travel through one of these airports, stop by the TGS booth, volunteer to participate, and receive a free COVID-19 at-home test kit for your time contributing to public health, stated the CDC on January 26, 2023.

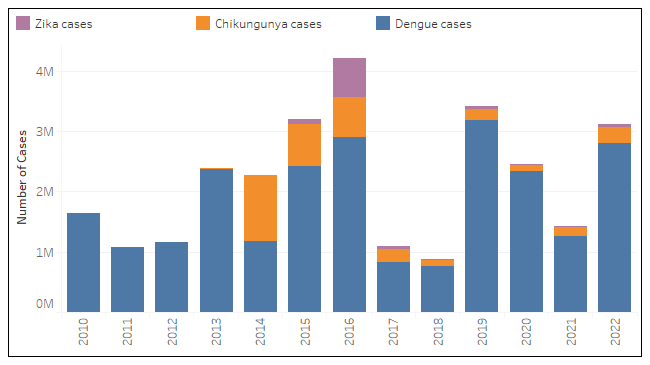
According to new data published today by the Pan American Health Organization (PAHO), the Region of the Americas reported 3,113,022 arboviral disease cases in 2022.
Of those cases, 90.1 % were dengue, 8.7 % were chikungunya, and 1.2 % were Zika virus.
Regarding related fatalities, dengue infections produced 1,289 deaths, chikungunya was 87, and Zika produced 2 deaths last year.
It is worth noting that in 2022 dengue peaked at week #17, whereas chikungunya peaked in week #18, and the Zika virus circulation was more robust in the first semester.
Country-specific data on case counts and completeness can be found at https://www.paho.org/plisa.
While dengue has two approved preventive vaccines available in certain countries, chikungunya and Zika candidates remain in various stages of human clinical trials as of January 26, 2023.
AC Immune SA today announced the first interim safety, tolerability, and immunogenicity findings from the Phase 1b/2 ABATE trial of its anti-amyloid-beta (Abeta) vaccine ACI-24.060 in patients with prodromal Alzheimer's disease (AD).
Early results from the first cohort of AD patients in ABATE showed that low dose ACI-24.060 could elicit an anti-Abeta antibody response as soon as week 6 (2 weeks after the second injection).
The data show that ACI-24.060 vaccination has been safe and well tolerated to date.
As a result, dosing in ABATE's second, higher-dose AD cohort has begun, and the trial is cleared to start screening specific individuals for part 2 of the study.
Dr. Andrea Pfeifer, CEO of AC Immune SA, commented in a press release on January 26, 2023, "We are delighted with the encouraging initial safety, and immunogenicity findings for ACI-24.060 in ABATE reported today."
"We believe ACI-24.060's successful development could provide patients with a novel therapeutic option offering numerous potential advantages in treatment, maintenance, and prevention settings."
"These early findings from ABATE represent an important step towards this goal, and we look forward to reporting more detailed data at a future conference."
ACI-24.060, derived from AC Immune's SupraAntigen® platform, has been shown in preclinical studies to induce a strong polyclonal antibody response that matures and is maintained against oligomeric and pyroglutamate-Abeta species, essential pathological forms of Abeta believed to drive Abeta plaque formation and disease progression.
Targeting Abeta using antibodies has recently been validated with U.S. FDA approvals of new monoclonal antibody treatments for patients with AD.
By eliciting polyclonal anti-Abeta antibodies, the ACI-24.060 anti-Abeta vaccine development program aims to ultimately deliver significant benefits to patients, their caregivers, and healthcare systems regarding potential safety and tolerability, low-frequency dosing, low overall costs, and durable responses.

TG Therapeutics, Inc. today announced the commercial launch of BRIUMVI™, for treating relapsing forms of multiple sclerosis (RMS), including clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
BRIUMVI is the first and only anti-CD20 monoclonal antibody (mAbs) approved for patients with RMS that can be administered in a one-hour infusion following the starting dose.
BRIUMVI targets a unique epitope on CD20-expressing B-cells and was designed to lack specific sugar molecules normally expressed on the antibody.
Removing these sugar molecules, a process called glycoengineering allows for efficient B-cell depletion at low doses.
BRIUMVI was approved by the U.S. Food & Drug Administration based on data from Phase 3 trials, which demonstrated superiority over teriflunomide in significantly reducing the annualized relapse rate (the primary endpoint), the number of T1 Gd-enhancing lesions and the number of new or enlarging T2 lesions.
Michael S. Weiss, the Company's Chairman and CEO stated in a press release on January 26, 2023, "Our team has been working hard to make BRIUMVI available as quickly as possible, and we are pleased to announce that BRIUMVI is now available to healthcare providers and patients."
"We continue to be highly focused on ensuring patients who benefit from BRIUMVI can easily access treatment."
RMS is a chronic demyelinating disease of the central nervous system. It includes people with relapsing-remitting multiple sclerosis and people with secondary progressive multiple sclerosis who continue to experience relapses.
The results from the ULTIMATE I & II trials were published on August 25, 2022, in The New England Journal of Medicine.
The launch of BRIUMVI includes an extensive patient support program designed to support patients through their treatment journey. More information about the BRIUMVI Patient Support program can be accessed at www.briumvi.com.
TG Therapeutics was founded with one goal in mind—to leverage scientific advances in B-cell biology to develop novel treatments for patients.