Search API
Jiangsu Recbio Technology Co., Ltd. today announced it has recently completed the first batch of subject enrollment for its novel adjuvanted recombinant vaccine, REC610, that prevents shingles caused by varicella zoster virus (VZV).
The study is a randomized, observer-blinded, active-controlled phase I clinical trial to evaluate the safety and immunogenicity of REC610 in healthy adult subjects aged 40 and above.
According to research data on shingles vaccines that have been marketed worldwide, the novel adjuvanted vaccine can provide stronger cellular immunity and protective efficacy compared to live attenuated vaccines.
This announcement is essential since about 2.5 million adults in China suffer from herpes zoster yearly, and most seniors in China carry the dormant herpes zoster virus.
REC610 is equipped with a novel adjuvant BFA01 independently developed by the Company, which can promote the production of high levels of VZV glycoprotein E (gE)-specific CD4+ T cells and antibodies.
Preclinical studies have shown that REC610 has favorable immunogenicity and can induce high levels of gE-specific CD4+ T cell responses and IgG antibodies.
Its immune response is non-inferior to the controlled vaccine, Shingrix® says the Company.
Shingrix® is a non-live, adjuvanted recombinant vaccine consisting of the VZV glycoprotein E antigen and the AS01B adjuvant system, a proprietary adjuvant containing QS-21 and MPL with liposomes.
Shingrix has been commercially available in the U.S. since 2017.
Additional shingles vaccine development is posted at PrecisionVaccinations.

A United Nations (UN) team led by Beatrice Mutali, the Resident Coordinator in Zambia, recently confirmed it is supporting the Republic of Zambia's response to a cholera outbreak.
Zambia's Ministry of Health declared the cholera outbreak on January 26, 2023.
Cholera outbreaks in three districts have been reported, with 90 cases and three related fatalities confirmed.
As of February 10, 2023, the UN team is providing technical support to authorities while mobilizing funding to bridge a gap of $32 million for risk communication, water and sanitation, food, and other items.
Since December 2022, UNICEF has provided 4.5 tonnes of granular chlorine, 20,000 bottles of liquid chlorine for household water treatment, 250 containers of disinfectant, and other critical supplies to keep the water safe.
For its part, the World Health Organization (WHO) is working with Zambian authorities to strengthen assessment capacities along with surveillance, contact tracing, and lab preparations for cholera vaccination.
As of February 12, 2023, extensive cholera outbreaks have been reported in Haiti, Malawi, and Syria.
The WHO has approved various cholera vaccines for use in 2023.
Unfortunately, these cholera vaccines are not currently available in the U.S.
In addition to cholera, the U.S. CDC suggests various vaccinations before visiting Zambia.
These travel vaccines are offered in the U.S. at certified clinics and pharmacies.
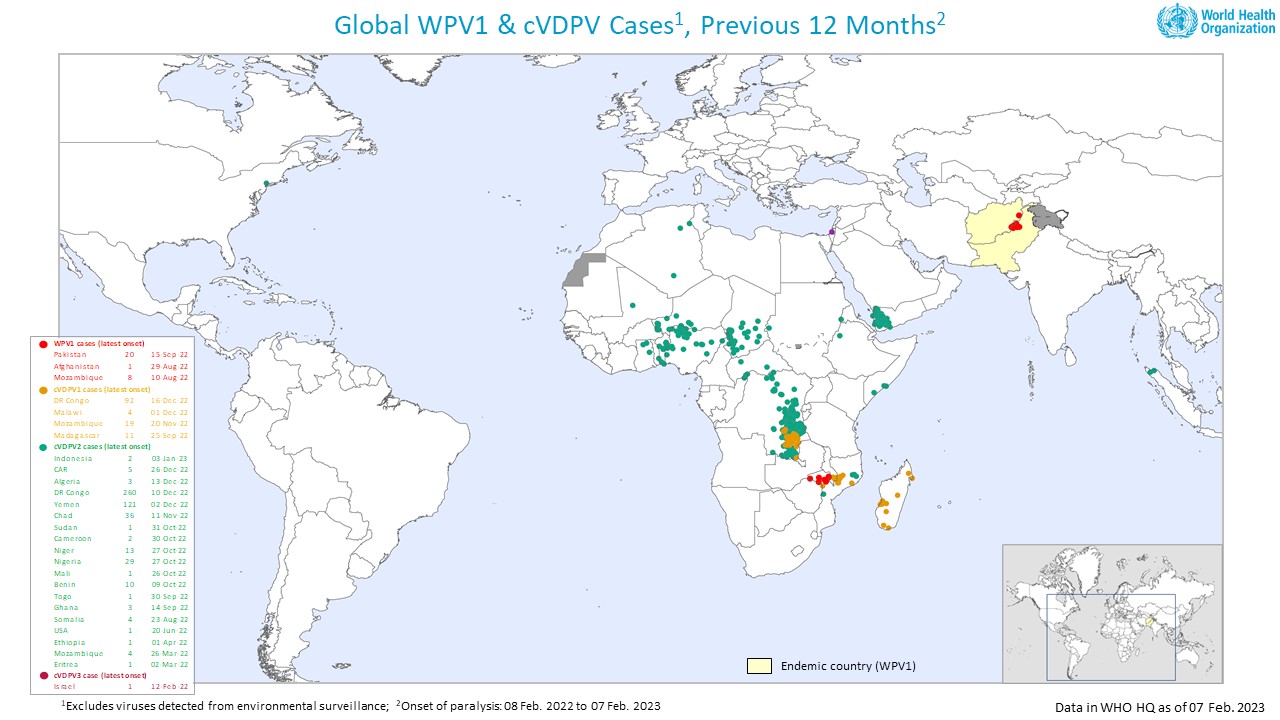
In his opening address to the Executive Board, WHO Director-General Dr. Tedros Adhanom Ghebreyesus recently noted that no wild poliovirus cases had been reported anywhere since September 2022.
"We are in a much better situation now than we were previously. But the last mile is the hardest."
"There can be no room for complacency. Now is actually the moment to double down on our efforts. So let's continue to push on."
Experts noted the unique window of opportunity to succeed in 2023, the target year for stopping all remaining poliovirus transmission globally.
The WHO also provided guidance to develop a new vision for the polio transition that will go beyond 2023, supported by tailored regional action plans to drive country progress.
AN updated list of At-Risk Countries was posted.
Following that WHO meeting, the Global Polio Eradication Initiative reported on February 8, 2023, a summary of new polioviruses detections and polio cases this week:
- Afghanistan: two WPV1-positive environmental samples
- Algeria: one cVDPV2 case
- Chad: two cVDPV2 cases
- DRC: eight cVDPV1 cases, 22 cVDPV2 cases and one cVDPV2 positive environmental sample
- Mali: one cVDPV2 case
- Nigeria: two cVDPV2-positive environmental samples
To alert international travelers of their potential health risk, the U.S. CDC updated its Alert - Level 2, Practice Enhanced Precautions notice regarding polio outbreaks in 2023.
Other polio outbreak news is posted at Vax-Before-Travel.
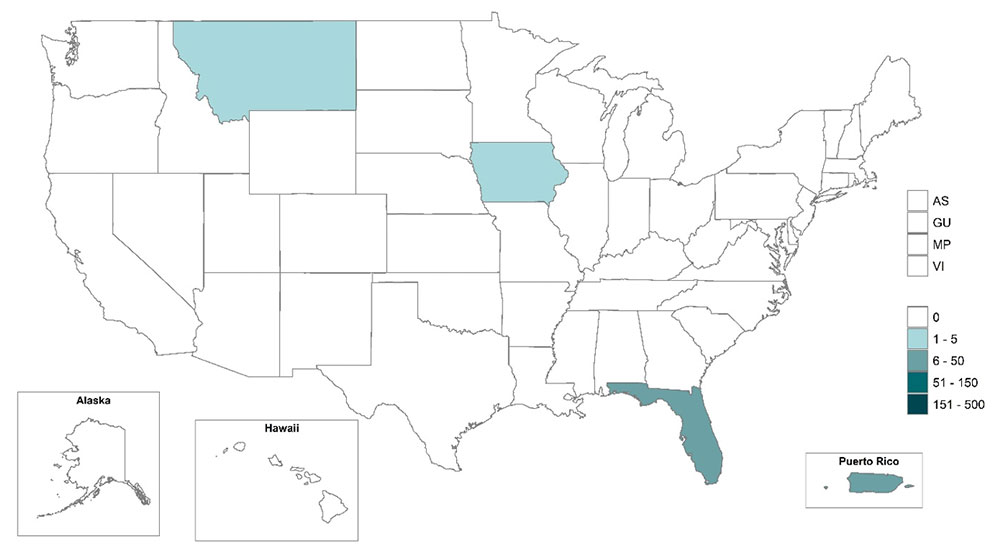
The Florida Health Department reported on February 4, 2023, sixteen travel-associated and one locally acquired dengue cases have been confirmed this year.
In 2022, Florida reported 869 travel-associated and 68 locally-acquired dengue cases.
To alert residents of their potential health risks, the Florida Department of Health in Miami-Dade County issued a mosquito-borne illness advisory in mid-2022.
Additionally, Broward County, which is located north of Miami, issued its mosquito-borne illness alert in 2022.
In the Region of the Americas, 46 countries and territories reported dengue cases in 2022.
Four different viruses cause dengue fever, and a person can be infected with each serotype during their lifetime.
Dengue viruses are spread to people through the bite of an infected Aedes mosquito.
Most symptoms of dengue are mild and typically last 2–7 days. But, infections can turn severe.
The U.S. CDC says no specific treatment for dengue/severe dengue exists.
However, one approved vaccine (Dengvaxia) is available in the U.S. on a limited basis.
And the U.K., Europe, and Indonesia have recently authorized the newer, less restrictive Qdenga® vaccine.
Since no U.S. Food and Drug Administration (FDA) vaccines are available to prevent Alzheimer's Disease (AD), can other vaccines reduce the risk of this old-age disease?
The Conversation published an article written by neurodegenerative disease experts on January 18, 2023, highlighting the potential value of existing FDA-approved vaccines to reduce AD aggravating factors from viral infections.
In previous studies, researchers identified that infections by varicella zoster (chickenpox and shingles) and influenza viruses could lead to a higher risk of AD.
However, researchers have been unable to consistently detect these suspect viruses in the brains of people who died of Alzheimer's.
In other words, when researchers analyze AD patient brains, detectable viral components may have disappeared, and causation is difficult to establish.
The full unedited article published by The Conversation is posted at this link.
As of February 12, 2023, the FDA has not authorized any of the current Alzheimer's vaccine candidates.

An outbreak of an undefined disease that has killed several people in Equatorial Guinea was reported on February 8, 2023, according to a Health Alert issued by the U.S. Embassy in Malabo.
The Government of Equatorial Guinea (EG) announced that over the past month, there had been an epidemiological outbreak of an unidentified disease in the Nsok Nsomo district of the Kie-Ntem province in the mainland region.
In a statement, EG said that travel restrictions had been imposed in "view of the high risk of importation of this disease.
According to preliminary investigations, these fatalities were linked to a funeral ceremony.
In reaction, the neighboring country of Cameroon has restricted movement along its border with Equatorial Guinea.
This area of Africa has been involved with numerous Ebolavirus outbreaks over the past decade.
The Republic of EG is located on Africa's west coast, consisting of Río Muni and five islands.
The local U.S. Embassy website has not posted an alert regarding this situation as of February 11, 2023, but does say in case of emergency, American citizens have access to an after-hours help line: (+240) 555-516 008.
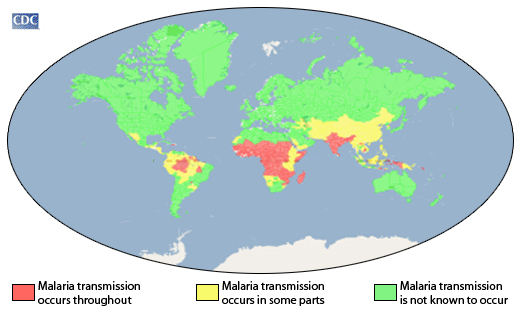
According to research published in The Lancet Infectious Diseases by researchers at the University of Maryland School of Medicine (UMSOM), a monoclonal antibody (mAbs) treatment conferred protection in people against P falciparum (malaria) at low doses
Published on January 25, 2023, this phase 1 clinical study found the mAbs CIS43LS protected 18 (82%) of 22 participants who received a dose. In addition, no participants developed parasitemia following dosing at 5 mg/kg intravenously or subcutaneously or at 10 mg/kg intravenously or subcutaneously.
All six control and four of seven participants dosed at 1 mg/kg intravenously developed parasitemia after controlled human malaria infection.
"The study demonstrates the feasibility of using mAbs therapies to help prevent malarial infection and holds promise for deployment to places where the disease is endemic," said Kirsten Lyke, MD, at UMSOM, in a related press release.
"This may allow us to revisit malaria eradication efforts."
According to the World Health Organization (WHO), malaria is a vaccine-preventable disease caused by a parasite.
Vaccines like Mosquirix™ (RTS,S), and R21/Matrix-M™ have been reported to be effective at preventing disease in Africa and India.
As of February 11, 2023, the U.S. Food and Drug Administration (FDA) had not approved a malaria vaccine.
According to the 2021 World Malaria Report, about half the world's population lives in areas at risk of malaria transmission.
There were 241 million malaria cases and 627,000 deaths reported worldwide in 2020 alone, a 12% increase from 2019.
However, outbreaks of locally transmitted malaria cases in the U.S. have been limited and relatively isolated. The FDA has approved Artesunate to treat severe malaria in adult and pediatric patients.
Malaria outbreak news is posted at Vax-Before-Travel.com/MalariaOutbreasks.