Search API
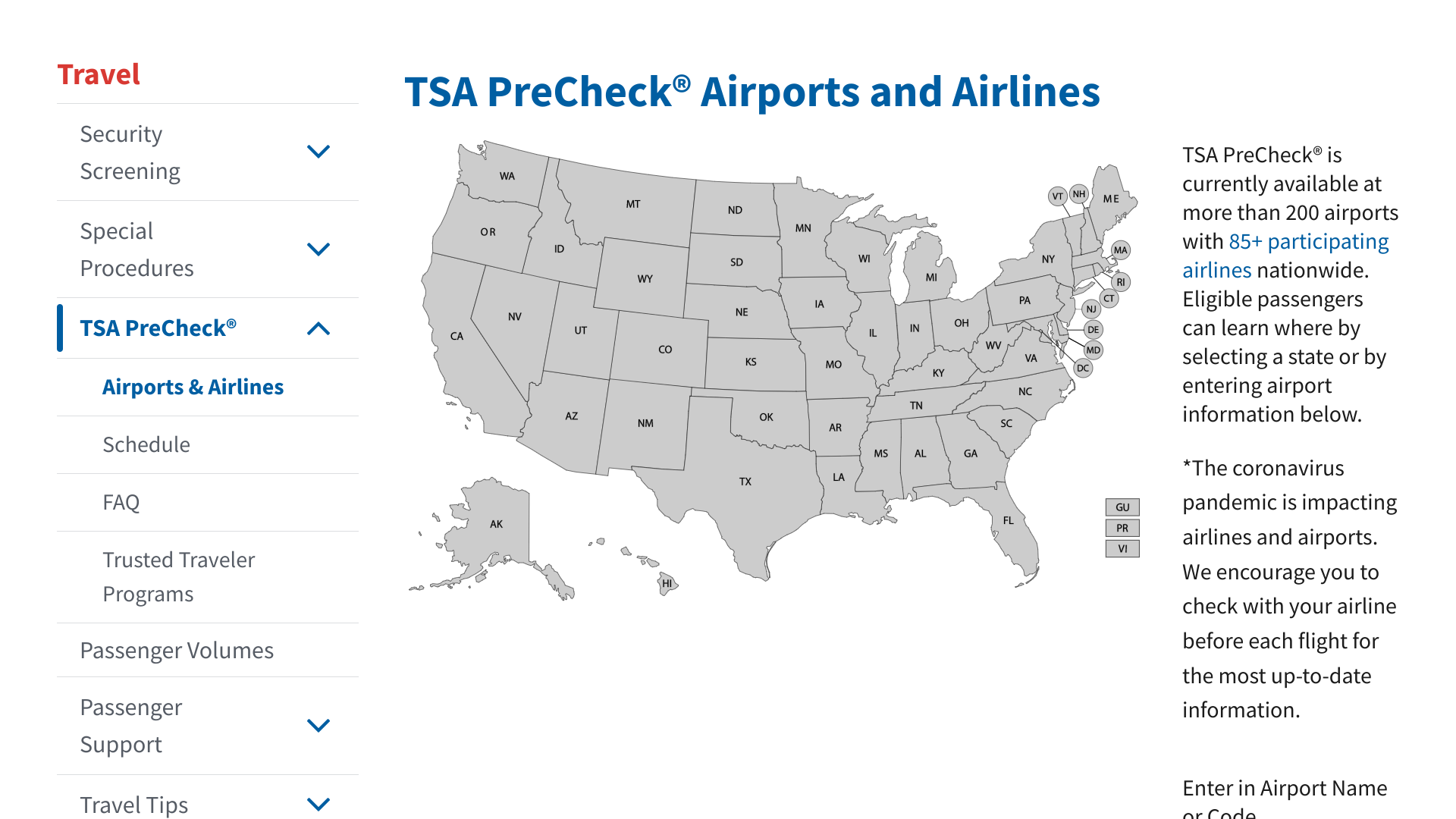
According to new data from the U.S. Transportation Security Administration (TSA), getting through airport security can be measured in minutes.
During March 2023, about 89% of TSA PreCheck® passengers waited less than 5 minutes to be processed at 200 airports in the U.S.
And the TSA makes this special service a family affair.
Children 12 and under can join a parent/guardian with TSA PreCheck® in the dedicated lanes.
Speeding through security is essential as the number of air travelers has returned to pre-pandemic levels.
As of the week ending April 27, 2023, TSA screening activity has matched 2019.
Passenger screening at the airport is part of TSA’s layered approach to security to get you safely to your destination. Search at this webpage to learn when TSA PreCheck® lanes are available at your airport.
Note: You do not need to get TSA PreCheck® if you already have Global Entry, NEXUS, SENTRI, or hold an active TWIC® or Commercial Driver’s License with an HME and you meet the TSA PreCheck® eligibility requirements.
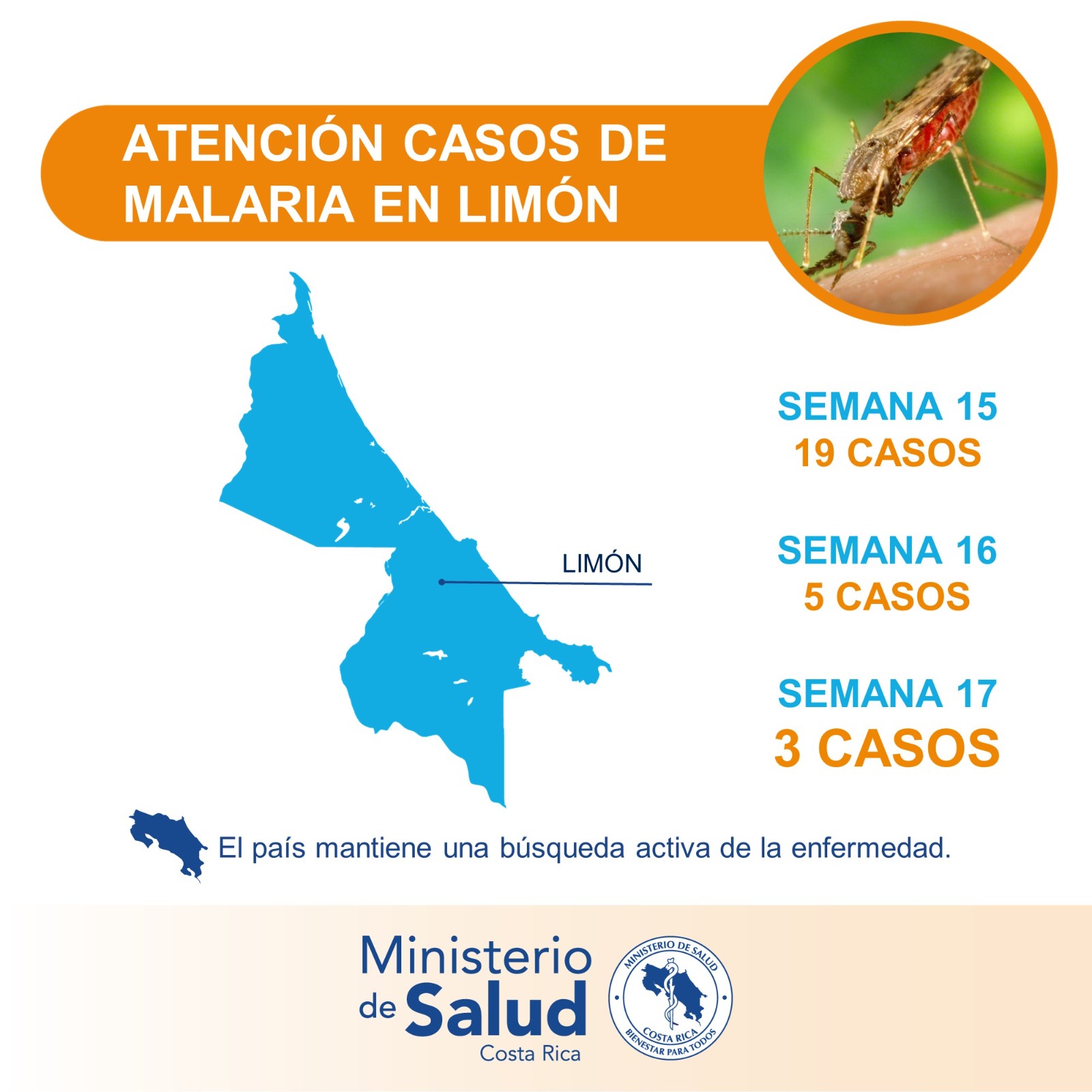
According to data recently announced from the Health Surveillance Directorate of the Costa Rican Ministry of Health, during epidemiological week #16, the Huetar Caribbean Region registered a marked decrease in malaria cases in the cantons of Limón and Pococí.
As of April 28, 2023, Limón reported 19 cases during week #15, while for week #16, 5 patients were registered, and for week #17, just 3 cases were reported, offering evidence of a significant decrease.
Furthermore, no malaria cases were identified in the canton of Talamanca, which corresponds to the South Caribbean, despite active inter-institutional searches in the border area with Panama and passive pursuits.
During the last weeks, the containment actions established by the Ministry of Health in conjunction with the Costa Rican Social Security Fund have been maintained, such as exhaustive tracking of all people in contact with confirmed cases.
Malaria is a disease caused by a parasite that spreads to humans through the bite of an infected mosquito.
Previously, the U.S. CDC issued an Alert - Level 2, Practice Enhanced Precautions confirming an outbreak of Plasmodium falciparum malaria in Limón and Alajuela Province.
Malaria can be prevented by taking a prescription antimalarial drug to kill the parasites, and in some countries, other than the U.S., malaria vaccines are available as of April 30, 2023.
The U.S. Centers for Disease Control and Prevention (CDC) today announced two additional influenza-associated pediatric fatalities were reported last week.
A total of 145 pediatric flu fatalities have been reported during the 2022-2023 season as of April 28, 2023.
The previous peak in pediatric fatalities was 199 during the 2019-2022 flu season before the COVID-19 pandemic closed society.
And based on National Center for Health Statistics Mortality Surveillance data available on April 27, 2023, about 7.5% of the fatalities during the week ending April 22, 2023 (week #16) were due to pneumonia, influenza, and/or COVID-19.
Most influenza viruses tested are in the same genetic subclade and antigenically similar to the influenza viruses included in this season’s influenza vaccine.
These flu shots remain available at most community pharmacies in the U.S.

The U.S. Food and Drug Administration (FDA) issued a letter today updating Pfizer Inc.'s COVID-18 vaccine authorization.
Addressed to Pfizer's Leslie Sands on April 28, 203, the new authorization for the Pfizer-BioNTech COVID-19 Vaccine, Bivalent for individuals six months through 4 years of age with certain types of immunocompromised, who have previously received three 0.2 mL doses (Pfizer-BioNTech COVID-19 Vaccine or Pfizer-BioNTech COVID-19 Vaccine, Bivalent):
- a fourth dose administered at least one month following the most recent dose;
- additional doses may be administered at the healthcare provider's discretion, considering the individual's clinical circumstances.
This issue was discussed during the U.S. CDC's Advisory Committee on Immunization Practices meeting on April 19, 2023.

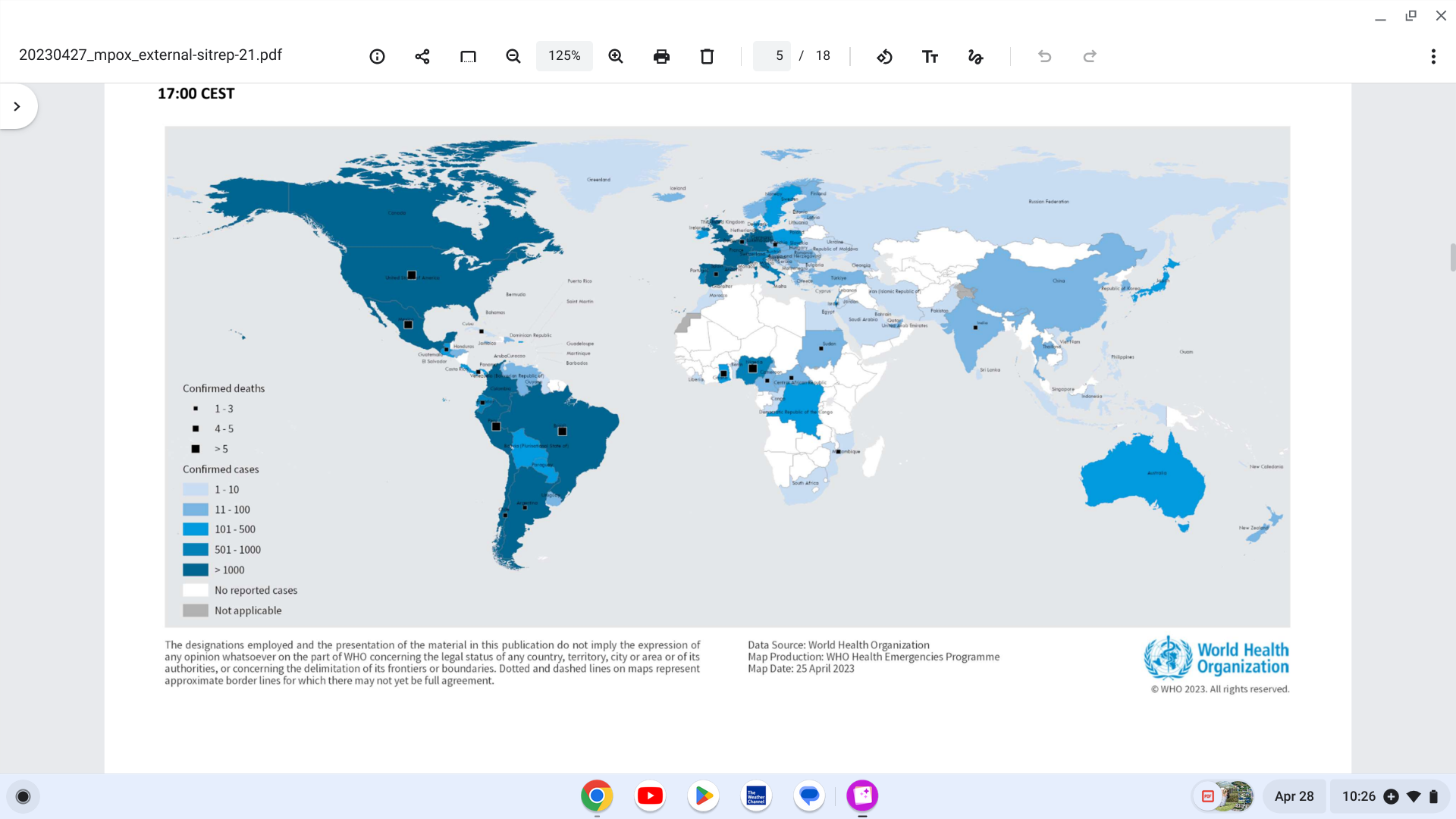
The World Health Organization (WHO) published the 21st situation report for the multi-country outbreak of mpox. Issued on April 27, 2023, the report offers an update from the WHO Eastern Mediterranean region.
Since April 13, 2023, 183 new Mpox cases (0.2% increase in total cases) and 14 new related fatalities have been reported to WHO.
From January 2022 through April 24, 2023, a cumulative total of 87,113 laboratory-confirmed cases of mpox, including 130 fatalities, have been reported from 111 countries/territories/areas.
For example, the Nation recently reported Pakistan had confirmed its fifth Mpox case related to international travelers.
According to sources, on April 27, 2023, three passengers arrived in Karachi on two flights from UAE on April 26 and were having symptoms of Mpox.
Previously, two Mpox cases were detected in Islamabad, originating from travelers from Saudi Arabia.
In response, the Director General of Health Services Sindh issued directives to all government hospitals to establish isolation areas for Mpox patients.
Mpox is a vaccine-preventable disease, according to the U.S. CDC. Throughout the Mpox outbreak, Bavarian Nordic's JYNNEOS® (MVA-BN) vaccine has been offered in various countries, including the U.S.
In the United States, there remains a considerable burden of disease attributed to serotypes not included in currently approved pneumococcal conjugate vaccines.
To address this need, Pfizer Inc. today announced that the U.S. Food and Drug Administration (FDA) had approved PREVNAR 20® for the prevention of invasive pneumococcal disease (IPD) caused by the 20 Streptococcus pneumoniae serotypes contained in the vaccine in infants and children six weeks through 17 years of age.
And for preventing otitis media in infants six weeks through five years of age caused by the original seven serotypes contained in PREVNAR®.
"Today's FDA approval of our vaccine, PREVNAR 20, now offers parents the ability to help protect their children against 20 pneumococcal serotypes in circulation, which represent the majority of pneumococcal disease in U.S. infants and children," said Annaliesa Anderson, Ph.D., Senior Vice President and Chief Scientific Officer, Vaccine Research and Development, Pfizer, in a press release on April 27, 2023.
"This important PREVNAR 20 approval builds on more than 20 years of real-world impact with PREVNAR and PREVNAR 13, safety data, and effectiveness, highlighting Pfizer's leadership in developing groundbreaking pneumococcal conjugate vaccines to help protect infants and their families from life-threatening infections."
Pneumococcal vaccine news is posted by Precision Vaccinations.

A National Academies committee recently assessed the value of community-level wastewater surveillance in controlling infectious diseases beyond Covid-19, wrote a Perspective published by the NEJM on April 20, 2023.
Nearly 80% of U.S. households are connected to municipal wastewater collection systems that contain biological waste, including discharged pathogens, such as poliovirus, in New York.
To better understand this innovative, ubiquitous method of detecting the prevalence of infectious diseases, listen to an interview with Profs. Michelle Mello and Guy Palmer focused on wastewater surveillance.
The 14:19 audio interview is accessible at this link.



