Search API
The journal Nature Communications recently published an article that concluded interim data from 2 parts of the phase 1/2 clinical trial support the continued development of mRNA-1010.
The mRNA-1010 vaccine candidate elicited either higher or comparable immune responses to a standard-dose, influenza quadrivalent inactivated vaccine.
Overall, these first-in-human safety and immunogenicity findings highlight, on June 19, 2023, the potential of the mRNA platform to improve the effectiveness of influenza vaccines.
Vaccines using mRNA technology are readily amenable to antigenic drift and shift in influenza strains, allowing for rapid deployment of vaccines. In addition, mRNA-based platforms allow for the expression of multiple antigens, raising the possibility for an increased breadth of protective responses against seasonal influenza or multiple respiratory diseases.
However, approved cell-based influenza vaccines, such as Flucelvax® Quadrivalent (QIVc), currently produce an exact antigenic match for circulating flu trains.
Further, based on findings with mRNA-1273, an mRNA-based vaccine against SARS-CoV-2, mRNA vaccines may also induce strong cellular immune responses and prolonged germinal center reactions that can improve protection in older adults, a population at particular risk for infection and severe outcomes.
While mRNA-1010 had an acceptable safety profile in this trial, transient solicited adverse reactions were more common after mRNA-1010 than with the active comparator.
Additional clinical trials are ongoing to assess further this vaccine candidate's safety, efficacy, and immunogenicity and a licensed influenza vaccine comparator (NCT05566639 and NCT05415462).
Moderna, Inc. was involved in the study design, data collection and analysis, and the writing of this manuscript. Moderna funded this study.

The Colorado Sun today reported Dr. Rachel Herlihy, an epidemiologist at the Colorado Department of Public Health and Environment (CDPHE), confirmed three mpox cases were identified in June and one in May 2023.
Dr. Herlihy told John Ingold on June 26, 2023, that some cases involved out-of-state travel, and two patients were linked.
Herlihy confirmed of the four recent Colorado cases, two were in people who had received the complete two-dose JYNNEOS® (MVA-BN) vaccine regimen, and one was in someone who was unvaccinated.
Additional mpox vaccine breakthrough cases, such as about 40 in Chicago, IL, have been confirmed.
CDPHE's press release on June 12, 2023, stated vaccination continues to be one of the most important preventive measures against mpox.
Mpox vaccination in Colorado remains limited to those currently most at risk for exposure to the virus. Go to CDPHE's website to find a vaccine provider or sign up for vaccination at a mobile clinic.
"There's always going to be the potential for re-introduction," Herlihy said. "For the time being, at least, I think we will continue to see sporadic (mpox) cases occurring."
The U.S. Centers for Disease Control and Prevention (CDC) updated the U.S. National Mpox Vaccination Strategy in 2023, continuing to recommend people exposed to the mpox virus and those more likely to contract the sexually transmitted disease should be vaccinated.
However, the CDC's Advisory Committee on Immunization Practices recently presented no recommendation for a third JYNNEOS dose, including for persons with advanced HIV or other severe immunocompromise.
Mpox vaccination and outbreak news was updated on June 26, 2023.

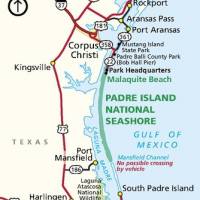
Invivyd, Inc. today announced that it has reached general alignment with the U.S. Food and Drug Administration (FDA) on a pathway to potential emergency use authorization (EUA) for VYD222 and anticipated follow-on monoclonal antibody (mAb) candidates designed to prevent symptomatic COVID-19.
The company plans to leverage the pathway, which includes the use of serum-neutralizing titers as a correlate of protection in an immunobridging approach to a pivotal clinical trial of VYD222, to rapidly generate data to support a potential VYD222 EUA for the prevention of symptomatic COVID-19.
Based on FDA feedback, the use of a correlate of protection in an immunobridging approach to a pivotal EUA-directed clinical trial may be a reasonable approach for a new mAb candidate when clinical trial data from a "prototype" mAb is available, provided that the new mAb candidate:
- Is similar to the prototype mAb such that it leverages a consistent manufacturing platform and has limited structural and functional differences, and,
- Has supportive nonclinical data, such as favorable in vitro neutralization data against currently circulating SARS-CoV-2 variants.
"We are very encouraged by the recent feedback from the FDA and appreciate their commitment to exploring alternative strategies to expedite the development of mAbs for the prevention of symptomatic COVID-19, such as the use of a correlate of protection as the primary endpoint in a pivotal clinical trial, a strategy that we are pleased to see further advance following the joint EMA-FDA workshop last December where the approach was discussed," said Dave Hering, chief executive officer of Invivyd, in a press release on June 26, 2023.
"Given our previous work developing adintrevimab and our platform-based approach to rapid mAb discovery, we believe we are one of few companies positioned to rapidly and serially generate data for potential EUA submission for next-generation mAb candidates for the prevention of symptomatic COVID-19."
"This potential pathway supports the company's vision and strategy of establishing a platform and stream of optimized anti-SARS-CoV-2 mAb candidates that can be deployed to keep pace with viral evolution and protect the vulnerable."
COVID-19 antibody and antiviral news was updated on June 26, 2023.
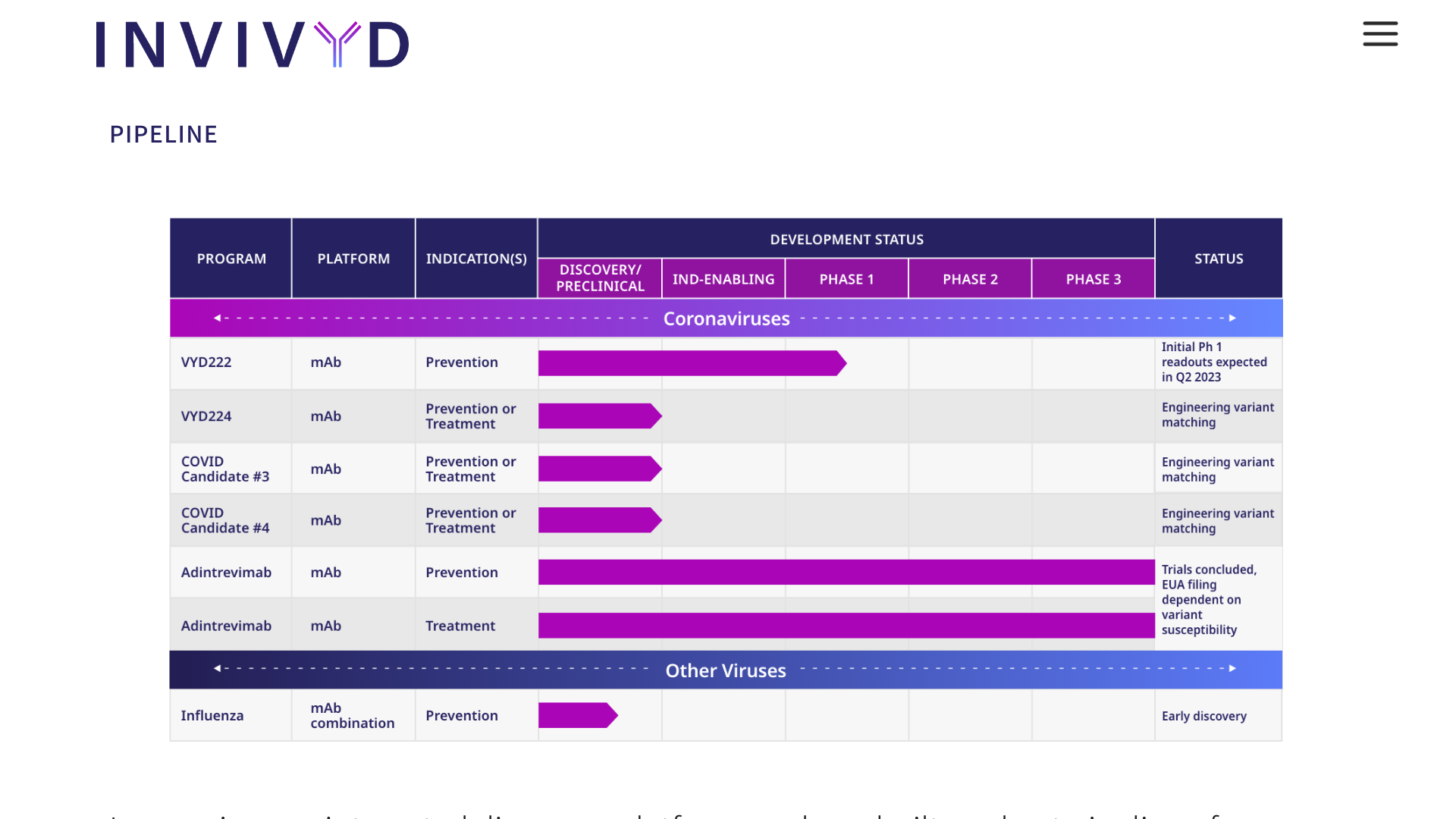
Sorrento Therapeutics, Inc. today announced the full enrollment in a pivotal Phase 3 study of the oral Mpro inhibitor, Ovydso (STI-1558), in mild or moderate symptomatic adults infected with SARS-CoV-2, or COVID-19.
Sorrento anticipates that top-line data from the study will be available in the third quarter of 2023.
Once the data is finalized, Sorrento plans to open discussions with regulatory authorities worldwide to discuss the path required for each particular authority for full approval of Ovydso.
If the trial meets its endpoints, the company has agreements with the China Health Authority and the National Medical Products Administration (NMPA) for an application review.
“We are pleased to see that Ovydso has enrolled quickly for successful completion of enrollment for the phase 3 pivotal trial in China. We look forward to seeing the final data and to working closely with the NMPA during the review to evaluate this as a potential stand-alone treatment for COVID-19 patients as rapidly as possible,” stated Henry Ji, Ph.D., Chairman and CEO of Sorrento, in a press release on June 26, 2023.
STI-1558 is a prodrug, and its active form AC1115 binds to Cys-145 of the catalytic domain of Mpro, which is 100% conserved in all SARS-CoV-2 variants and achieves a broad-spectrum anti-SARS-CoV-2 activity.
STI-1558 is also a Cathepsin L inhibitor, which may block effective viral entry into host cells without accelerating viral mutations.
Sorrento is a clinical and commercial stage biopharmaceutical company based in California.
Pfizer Inc. recently announced that the U.S. Centers for Disease Control and Prevention’s (CDC) Advisory Committee on Immunization Practices (ACIP) unanimously voted to recommend PREVNAR 20® (20-valent Pneumococcal Conjugate Vaccine) for routine use to help protect infants and children from invasive pneumococcal disease (IPD) caused by the 20 Streptococcus pneumoniae serotypes covered by the vaccine and for the prevention of otitis media in infants six weeks through five years of age caused by the original seven serotypes contained in PREVNAR®.
These provisional recommendations will be reviewed and finalized by the director of the CDC and the Department of Health and Human Services.
“We are thrilled with today’s ACIP decision as it recognizes the increased level of protection that PREVNAR 20 will provide to millions of infants and children against pneumococcal disease,” said Luis Jodar, Ph.D., Chief Medical Affairs Officer, Vaccines/Antivirals and Evidence Generation, Pfizer, in a press release on June 22, 2023.
Previously, the European Medicines Agency approved the brand name Apexxnar in February 2022.

The Florida Health Department recently published an updated Mosquito-Borne Disease Surveillance that revealed additional dengue and malaria patients in Florida.
As of June 17, 2023, the Weekly Arbovirus Report confirmed:
- Four cases of dengue were reported in persons with international travel. In 2023, 88 travel-associated dengue cases were reported. And two cases of locally acquired dengue have been reported.
- Another case of locally acquired malaria was reported in Sarasota County. In 2023, two cases of locally acquired malaria have been reported.
In response to these mosquito-transmitted diseases, Florida has issued alerts for Sarasota and Manatee counties regarding malaria.
And in Miami-Dade County, a dengue alert was issued in April 2023.
The Florida Department of Health is working with local partners and county mosquito control in these areas to mitigate the risk of further mosquito transmission.
Florida continues statewide surveillance for mosquito-borne illnesses, including West Nile virus, Eastern equine encephalitis, St. Louis encephalitis, malaria, chikungunya, and dengue.
From a vaccination perspective, one dengue vaccine is approved in the U.S., and malaria vaccines are currently available in Africa as of June 23, 2023.

The Florida Department of Health (DOH) in Sarasota and Manatee Counties recently confirmed a second locally-acquired malaria case.
As of June 19, 2023, this patient was being treated for this mosquito-cause infection.
This case has been identified as the P. vivax malaria species, which is not as fatal as other species.
In response, a mosquito-borne illness alert was issued for Sarasota and Manatee counties.
On May 26, 2023, DOH released information on the first confirmed local malaria case, who was treated and recovered.
Effective treatment is readily available through Florida hospitals and other healthcare providers.
The Florida Department of Health confirmed aerial and ground mosquito spraying is being conducted in these counties to mitigate the risk of further transmission.
Malaria is not transmitted from person to person.
Only infected Anopheles mosquitoes can transmit malaria to humans.
Malaria infects approximately 219 million people each year, with an estimated 660,000 deaths, mostly children in Africa.
The U.S. Centers for Disease Control and Prevention (CDC) recently stated the risk of locally acquired malaria in the U.S. is extremely low.
About 2,000 malaria cases are diagnosed annually in the U.S., most in international travelers.
However, Florida has had malaria outbreaks in the past.
In 2003, eight locally acquired P. vivax malaria cases were reported in Palm Beach County, FL.
From a prevention perspective, malaria vaccines have already been approved.
The World Health Organization recommended the Mosquirix™ malaria vaccine in 2021, and the R21/Matrix-M™ vaccine was approved for use in Africa in 2023.