Search API
The U.S. Centers for Disease Control and Prevention (CDC) today announced a meeting of the Advisory Committee on Immunization Practices (ACIP) will be held on September 12, 2023, at 10 a.m. ET.
The agenda for this ACIP meeting will include, but is not limited to, a discussion of COVID-19 vaccines and related votes.
For more information on the meeting agenda, visit https://www.cdc.gov/vaccines/acip/meetings/index.html.
The CDC's website publishes Interim Clinical Considerations for the use of COVID-19 vaccines in the U.S., which include a FAQs webpage.
As of August 18, 2023, more than 307 million doses of various COVID-19 vaccines had been administered and reported by the Federal Retail Pharmacy Program. A total of 21 retail pharmacy partners are participating in the program, with more than 41,000 locations nationwide, including long-term care pharmacies.
UPDATED on September 12, 2023 - CDC slide presentations.

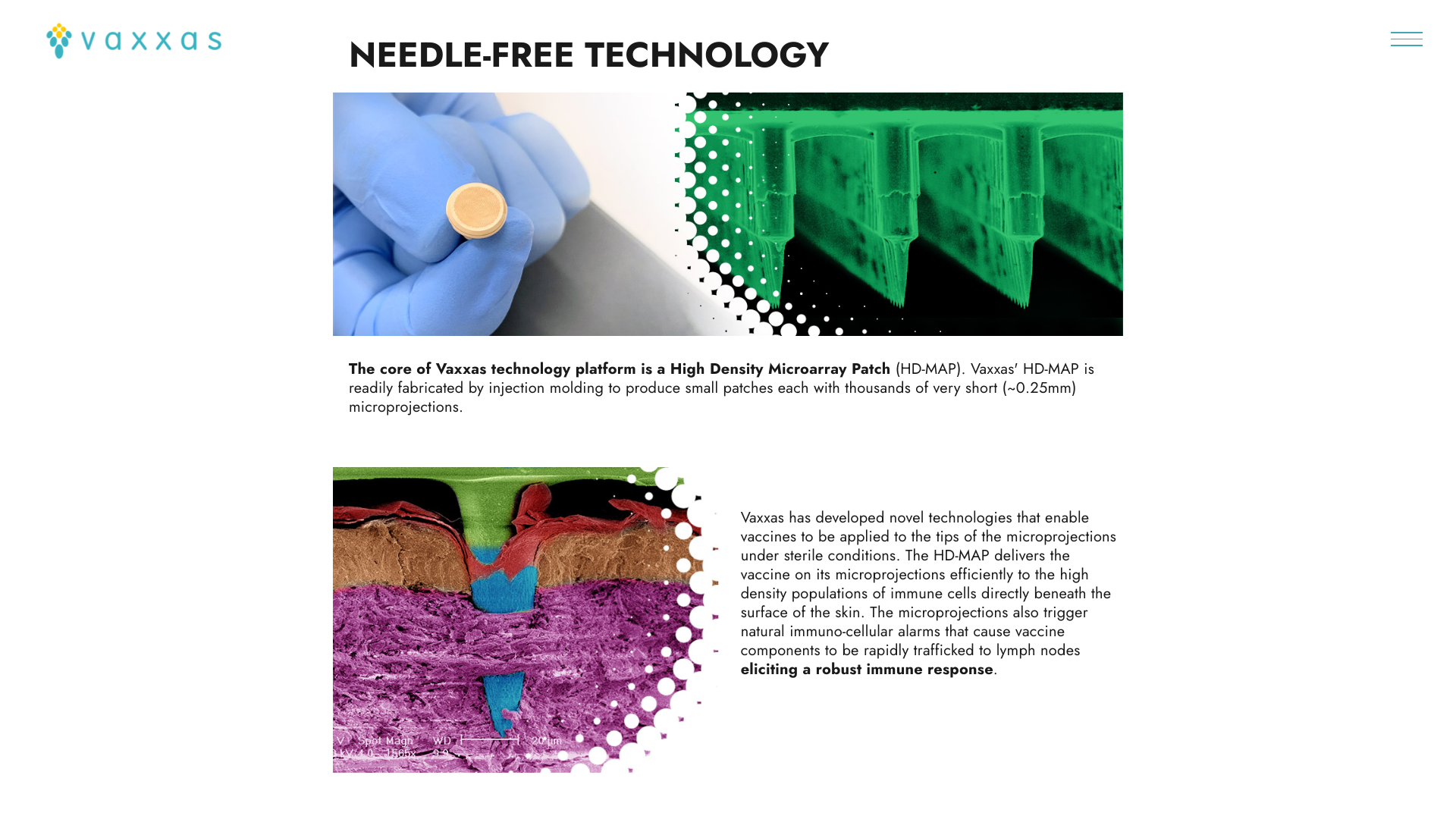
SK bioscience today announced that the company has entered into a collaboration agreement with Vaxxas to develop a second-generation typhoid conjugate vaccine.
SK bioscience’s SKYTyphoid™ vaccine will be reformulated to be ‘printed’ onto the thousands of tiny microprojections covering the Vaxxas patch to be delivered directly to the abundant immune cells just under the skin surface.
This reformulation aims to enhance access and broaden markets where traditional intramuscular delivery using needles and syringes has been employed.
Under the agreement announced on August 23, 2023, SK bioscience will supply the antigen utilized by its typhoid conjugate vaccine, SKYTyhpoid™, jointly developed by SK bioscience and the International Vaccine Institute.
Vaxxas, an Australian company, will be responsible for reformulating the SKYTyphoid antigen so that it can be applied to its proprietary HD-MAPs and then conduct preclinical studies, which, if successful, will be followed by a Phase I human clinical trial.
Jaeyong Ahn, CEO of SK bioscience said in a press release, “Typhoid fever is a dangerous disease that frequently occurs in low- and middle-income countries, but the utilization of typhoid vaccines has been limited due to the requirement for vaccines that remain stable under varying temperatures and those that can be administered without medical supervision."
This collaboration with Vaxxas provides an opportunity to overcome those challenges."
The project is expected to be completed within two years from initiation to reporting the data from the Phase I clinical trial and is supported by grant funding received from Wellcome.
According to the World Health Organization, an estimated 9 million cases of typhoid fever occur globally each year. Symptoms include prolonged high fever, fatigue, headache, nausea, abdominal pain, and constipation or diarrhea, with mortality rates up to 30%.
As of August 24, 2023, typhoid vaccines are available in the U.S. and internationally.
The U.S. Centers for Disease Control and Prevention (CDC) website reported yesterday that a new variant of SARS-CoV-2 betacoronavirus called BA.2.86 was detected in samples from people in Denmark and Israel, and at least two cases have been identified in the United States.
This variant is notable because it has multiple genetic differences from previous versions of SARS-CoV-2.
BA.2.86 may be more capable of causing infection in people who have previously had COVID-19 or who have received COVID-19 vaccines.
Scientists are evaluating the effectiveness of the forthcoming, updated COVID-19 vaccine. CDC’s current assessment is that this updated vaccine will effectively reduce severe disease and hospitalization.
Based on what the CDC knows on August 23, 2023, there is no evidence that this variant is causing more severe illness.
This CDC assessment may change as additional scientific data are developed.
The Government of Hong Kong Special Administrative Region's Centre for Health Protection (CHP) of the Department of Health today announced it is closely monitoring a human case of avian influenza A(H5N6) in the Mainland.
The CHP report on August 23, 2023, says the 27-year-old woman lives in Dazhou, Sichuan, and was admitted for treatment on July 22. This report did not disclose how or where she became infected with 'bird flu' nor the outcome.
On July 23, 2023, the Alert Response Level under the Government's Preparedness Plan for an Influenza Pandemic was activated.
From 2014 to date, 86 human cases of avian influenza A(H5N6) have been reported by Chinese health authorities. A high percentage of infections (52%) lead to death.
The CHP says that travelers to the Mainland or other affected areas must avoid visiting wet markets, live poultry markets, or farms. And they should strictly observe personal and hand hygiene when visiting any place with live poultry.
The Food and Agriculture Organization of the United Nations, the World Health Organization (WHO), and the World Organisation for Animal Health urged countries in July 2023 to work together across sectors to save as many animals as possible and to protect people.
The current outbreaks of avian influenza ("bird flu") have caused devastation in animal populations, including poultry, wild birds, and some mammals. Although primarily affecting animals, these outbreaks pose ongoing risks to humans, says the WHO.
Since the annual flu shot is not designed to be effective against bird flu viruses, the U.S. government has invested in various avian influenza vaccines.
In June 2023, the U.S. Centers for Disease Control and Prevention confirmed about 20 million H5N1 and 12 million H7N9 vaccines were available in the U.S. National Strategic Stockpile.

The U.S. Administration for Strategic Preparedness and Response's Biomedical Advanced Research and Development Authority (BARDA) today awarded $10 million to Johnson & Johnson Innovation for a competition through project Blue Knight™.
"As the virus continues to evolve, we need new tools that keep pace with those changes," said Assistant Secretary for Preparedness and Response Dawn O'Connell in a press release on August 22, 2023.
This BARDA award is in alignment with 'Project NextGen' which focuses on advancing solutions aimed at addressing health security threats and improving preparedness,
Announced in May 2023, the U.S. Department of Health and Human Services Project NextGen is a $5 billion initiative led by BARDA in partnership with the National Institute of Allergy and Infectious Diseases, coordinates activities across the federal government and the private sector to advance innovative vaccines and therapeutics into clinical trials, regulatory review, and potential commercial availability for the American people.
Announced on May 11, 2023, the Blue Knight challenge offers current and alumni Blue Knight residents and their collaborators the opportunity to apply for the chance to receive funding to help them reach their critical developmental milestones.
Learn more about Blue Knight and hear from current companies at this Johnson & Johnson Innovation LLC link.

Regeneron Pharmaceuticals, Inc. today announced that the U.S. Biomedical Advanced Research and Development Authority (BARDA) entered into an agreement with the Company to support the clinical development, clinical manufacturing, and regulatory licensure process of a next-generation COVID-19 monoclonal antibody (mAb) therapy for the prevention of SARS-CoV-2 infections, which cause COVID-19 in people.
The agreement is part of the U.S. Department of Health and Human Services (HHS) 'Project NextGen' initiative to advance innovative vaccines and therapeutics for COVID-19.
Regeneron's most advanced next-generation antibody candidate under this agreement is expected to enter clinical trials in 2023.
For the new COVID-19 program announced on August 22, 2023, HHS will fund up to 70% of Regeneron's costs for certain clinical development activities for a next-generation mAb therapy.
The new BARDA contract has an estimated value of up to approximately $326 million of government funding.
Regeneron's first COVID-19 mAb cocktail, REGEN-COV, was granted Emergency Use Authorization in November 2020, with nearly 3 million doses delivered to the U.S. Government between 2020 and 2022.
"We're pleased to expand our longstanding BARDA relationship, which is predicated on Regeneron's decades of investment in deep scientific research and enabling technologies," said Leonard S. Schleifer, M.D., Ph.D., Board Co-Chair, President and Chief Executive Officer of Regeneron, in a press release.
"Although COVID-19 has moved to an endemic stage, many people – including those with immunocompromising conditions – continue to face exposure that impacts their everyday life and could cause serious health consequences."
Previously, the U.S. CDC wrote some immunocompromised people benefit from mAb therapy instead of COVID-19 vaccination.
Under the NextGen project structure, Regeneron independently invents and proposes an antibody candidate, which BARDA and Regeneron will evaluate and agree upon for further development, manufacturing, and regulatory activities.
BARDA and Regeneron have previously worked together to deliver novel medicines for Ebola.
The new program announced today falls under Regeneron and BARDA's ongoing Other Transactions Agreement initiated in 2017 to develop a portfolio of antibodies targeting up to ten pathogens that pose significant risks to public health.

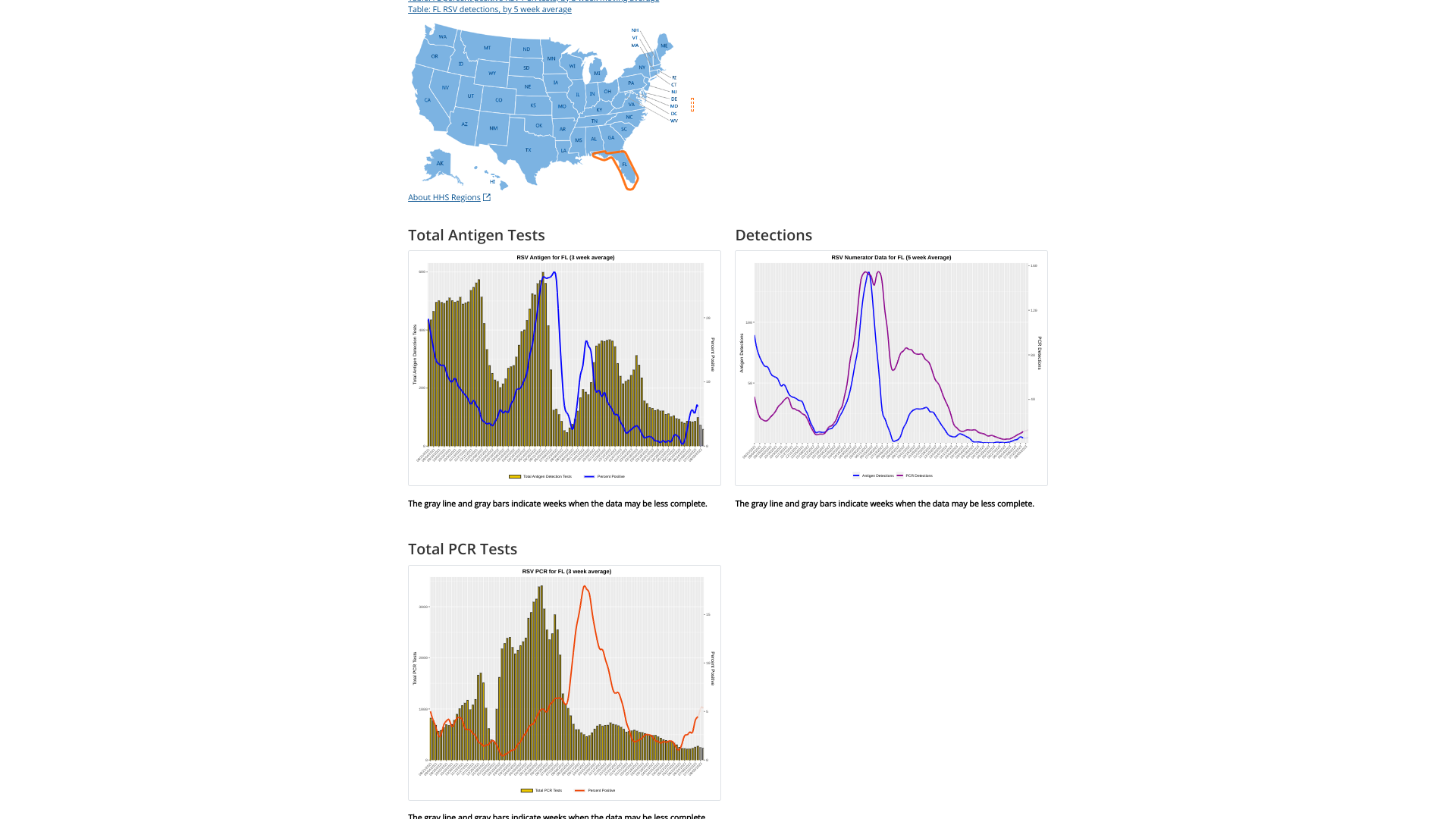
In the United States, the timing of seasonal respiratory syncytial virus (RSV) outbreaks throughout the country is generally reported between October - April most years.
The 2022–23 season it started later than in 2021–2022 but earlier than prepandemic seasons, suggesting a return toward normal seasonality.
Furthermore, the state of Florida is often a bell-weather state for RSV detections, as it is in 2023.
Florida’s RSV season is longer than the rest of the nation and has distinct regional patterns. For this reason, the state is segmented into five RSV regions, each with its own season.
The Florida Department of Health recently announced RSV activity during week #33 had increasing detection and admission rates.
As of August 19, 2023, two RSV outbreaks were confirmed in Martin County, located north of Jupiter. Last week, one RSV outbreak occurred in Jacksonville, Florida.
In the U.S., RSV infections cause thousands of hospitalizations among young children and adults aged ≥65. The U.S. Centers for Disease Control and Prevention (CDC) RSV detection 5-week moving average graphs for each state are displayed at this link.
This RSV season, the U.S. government has approved RSV vaccines and monoclonal antibody therapeutics available in specific clinics and pharmacies as of August 22, 2023.
The CDC says people should speak with a healthcare provider if these new therapeutics are appropriate for their health.