Search API
Health experts recently warned of additional measles outbreaks occurring in 2023 after measles cases increased by about 80% worldwide last year.
Since measles is a very contagious virus, the threat to the United States continues to be closely monitored.
Recently, the Illinois Department of Public Health (IDPH) reported a suspected case of measles was confirmed by Cook County Department of Public Health to IDPH.
This unvaccinated individual was exposed to the measles virus internationally, and the infectious period would be between October 5 through October 13, 2023. IDPH says people can spread measles up to four days before and four days after a rash appears.
Before this case, the last measles case in Illinois was identified in 2019, when 9 cases were confirmed.
"This first reported case of measles in Illinois since 2019 is a reminder that this disease can be prevented with up-to-date vaccination," said IDPH Director Dr. Sameer Vohra in a press release.
"Two doses of measles vaccine are 97% effective in preventing measles. However, as we saw this week, it still can affect unvaccinated people."
"I urge everyone to make sure they and their family members are up-to-date on measles/mumps/rubella (MMR) vaccine and all other age-appropriate immunizations."
Measles is easily spread through the air when someone coughs or sneezes. People can also get sick when they come in contact with mucus or saliva from an infected person. Measles can cause serious complications such as pneumonia and encephalitis.
In the U.S., the Centers for Disease Control and Prevention (CDC) reported 29 measles cases in 16 jurisdictions.
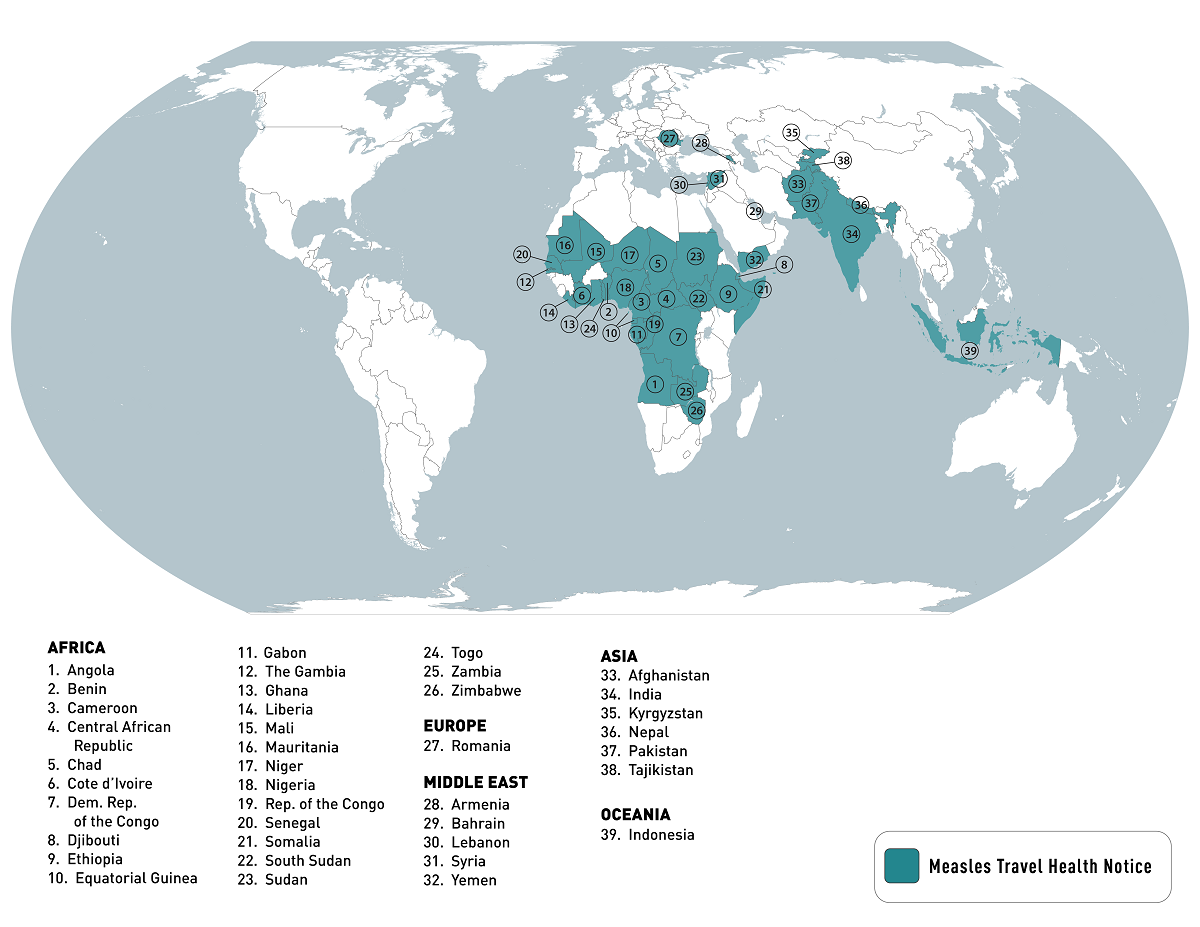
Internationally, the CDC listed the Top 10 Countries with Global Measles Outbreaks as of October 19, 2023. India, with 36,154 cases, and Yemen, with 28,271, lead this list.
To alert international travelers, the CDC published an updated list of measles outbreak countries, travel vaccine recommendations, and issued a Level 1 - Practice Usual Precautions, Travel Health Advisory in September 2023.
Pfizer Inc. recently announced that the U.S. Food and Drug Administration (FDA) approved PENBRAYA™, the first and only pentavalent vaccine that provides coverage against the most common serogroups causing meningococcal disease in adolescents and young adults 10 through 25 years of age.
PENBRAYA combines the components from two meningococcal vaccines, Trumenba® (meningococcal group B vaccine) and Nimenrix® (meningococcal groups A, C, W-135, and Y conjugate vaccine), to help protect against the five most common meningococcal serogroups that cause the majority of invasive meningococcal disease (IMD).
The FDA's decision is based on the positive results from the Phase 2 and Phase 3 trials, including a randomized, active-controlled, and observer-blinded Phase 3 trial assessing the safety, tolerability, and immunogenicity of the pentavalent vaccine candidate compared to currently U.S. licensed meningococcal vaccines, to determine immunologic noninferiority.
"Nearly 9 out of 10 adolescents have incomplete protection against IMD caused by the leading serogroups," said Jana Shaw, MD, Pediatrics Infectious Disease Specialist, Upstate Golisano Children's Hospital in Syracuse, NY, in a Pfizer press release on October 20, 2023.
"For the first time, we have a single vaccine that helps protect against the five most common serogroups and has the potential to improve coverage and increase protection among adolescents and young adults."
Meningococcal disease is an uncommon but serious illness that can lead to death within 24 hours and, for survivors, can result in life-altering, significant long-term disabilities.
The U.S. CDC Advisory Committee on Immunization Practices is scheduled to meet on October 25, 2023, to discuss recommendations for the appropriate use of PENBRAYA in adolescents and young adults and other vaccines.
Various meningococcal vaccines are approved by the CDC in 2023.

The Janssen Pharmaceutical Companies today confirmed at the American Society of Tropical Medicine & Hygiene Annual Meeting positive results from a Phase 2a human challenge study evaluating JNJ-1802, a first-in-class oral antiviral in development for the prevention of dengue.
Announced on October 20, 2023, the data showed that the JNJ-1802 induced antiviral activity against dengue (DENV-3) in humans, compared to placebo, and is safe and well-tolerated.
JNJ-1802 is the first antiviral to show such activity in humans during a clinical trial.
The compound is not a vaccine and has advanced to a community-based field study to establish efficacy against circulating dengue serotypes in a real-world setting. The new research is being conducted in 30+ sites in 10 countries.
Marnix Van Loock, Ph.D., Lead for Emerging Pathogens, Global Public Health R&D at Janssen Pharmaceutica NV, stated in a press release, “Dengue requires global action, and we are proud to collaborate alongside partners around the world in advancing the development of this compound to its next phase.”
According to the U.S. CDC, dengue viruses are spread to people through the bite of an infected Aedes species. Annually, up to 400 million people get infected with dengue, and 40,000 die from severe dengue.
This new study follows data published in the journal Nature in March 2023, which showed that JNJ-1802 provides strong protection against dengue in non-human primates and mice.
And a Phase 1 first-in-human clinical study showed that the antiviral was safe and well-tolerated.
Globally, dengue outbreaks are active in numerous countries during 2023.
As of October 21, 2023, two dengue vaccines are approved for use in various countries.

Without U.S. FDA-approved vaccines, a common antibiotic may become a "morning-after" pill to reduce the spread of chlamydia, syphilis, and gonorrhea among populations at higher risk for contracting sexually transmitted diseases (STDs).
On October 11, 2023, Dr. Connie Celum, a professor of global health and medicine at the University of Washington School of Medicine, stated in a press release, "Every year for the last six or seven years, there's been an increase in bacterial STDs. So the current approach of testing and treating is just not enough."
"Preventive doxycycline is "the best new intervention identified for STD prevention for quite a long time."
The U.S. Centers for Disease Control and Prevention (CDC) recently issued a preliminary recommendation for doxycycline to be prescribed to trans women and men who have sex with men, who also have experienced at least one sexually transmitted infection in the previous year, and who are at ongoing risk to acquire an STD.
Dr. Celum co-led one research study whose findings motivated the CDC's recommendation for "doxy-PEP."
In 2022, a study ended early because a single dose of 200 milligrams of doxycycline, taken within three days of unprotected sex, was highly effective at preventing the abovementioned STDs.
The most significant reduction was in chlamydia and early syphilis infections, added Celum.
As of October 20, 2023, there are approved vaccines that prevent certain STDs, such as HPV, and various vaccine candidates conducting clinical studies, such as herpes.

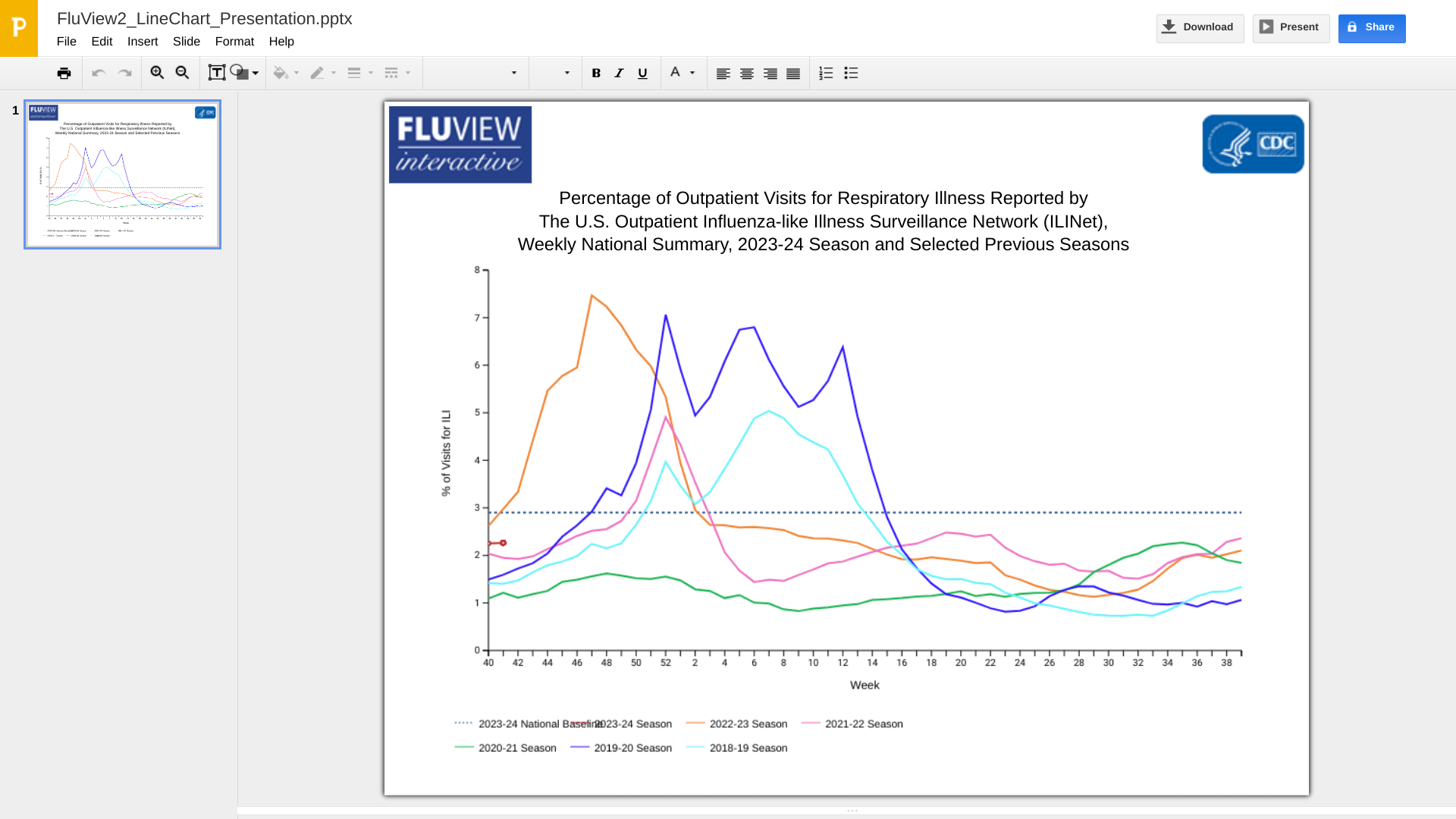
As the United States enters the traditional beginning of the annual flu season, the U.S. Centers for Disease Control and Prevention (CDC) today announced seasonal influenza activity remains low nationally.
As of October 20, 2023, the CDC reported all 10 HHS regions are below their respective baselines, and the number of flu hospital admissions remains low this flu season.
A recent update from the Walgreens Flu Index shows a few southern states are leading indicators of potential influenza outbreaks in states such as Texas.
From a severity perspective, the National Center for Health Statistics Mortality Surveillance reported that 20 influenza-related deaths occurred during the week ending October 14, 2023 (week #41).
Last flu season, the highest weekly count of deaths due to influenza was 1,048, occurring the week ending December 17, 2022.
The good news is that flu shot availability is unconstrained.
As of October 10, 2023, 128.35 million influenza vaccine doses have been distributed in the U.S.
For the 2023-2024 flu season, egg-based, cell-based, and nasal influenza vaccines are approved and available at most health clinics and pharmacies in the U.S.
Vaxcyte, Inc. today announced that the U.S. Food and Drug Administration (FDA) has cleared the Company's adult Investigational New Drug application for VAX-31, a 31-valent pneumococcal conjugate vaccine (PCV) candidate designed to prevent invasive pneumococcal disease (IPD).
Vaxcyte expects to initiate the VAX-31 Phase 1/2 clinical study in 2023 and announce topline results in the second half of 2024.
"The FDA clearance of the VAX-31 IND application represents an important step toward our goal of building a best-in-class PCV franchise, including VAX-31 and VAX-24, the 24-valent PCV for which we achieved positive results in two adult Phase 2 clinical studies," said Grant Pickering, Chief Executive Officer and Co-founder of Vaxcyte, in a press release on October 19, 2023.
"Given that VAX-31, which will be the broadest-spectrum PCV to enter the clinic, leverages the foundation already established with VAX-24, we are very excited about the promise of this vaccine candidate."
The VAX-31 Phase 1/2 clinical study is a randomized, observer-blind, active-controlled, dose-finding clinical study designed to evaluate the safety, tolerability, and immunogenicity of VAX-31 compared to Prevnar 20® (PCV20) in approximately 1,000 healthy adults aged 50 and above.
Pneumococcal disease (PD) is an infection caused by Streptococcus pneumoniae (pneumococcus) bacteria. It can result in IPD, including meningitis and bacteremia, and non-invasive PD, including pneumonia, otitis media, and sinusitis.
In the U.S., approximately 320,000 people get pneumococcal pneumonia yearly, estimated to result in about 150,000 hospitalizations and 5,000 deaths. Pneumococci also cause over 50% of all cases of bacterial meningitis in the U.S.
As of October 20, 2023, numerous licensed pneumococcal vaccines and candidates are conducting clinical trials.

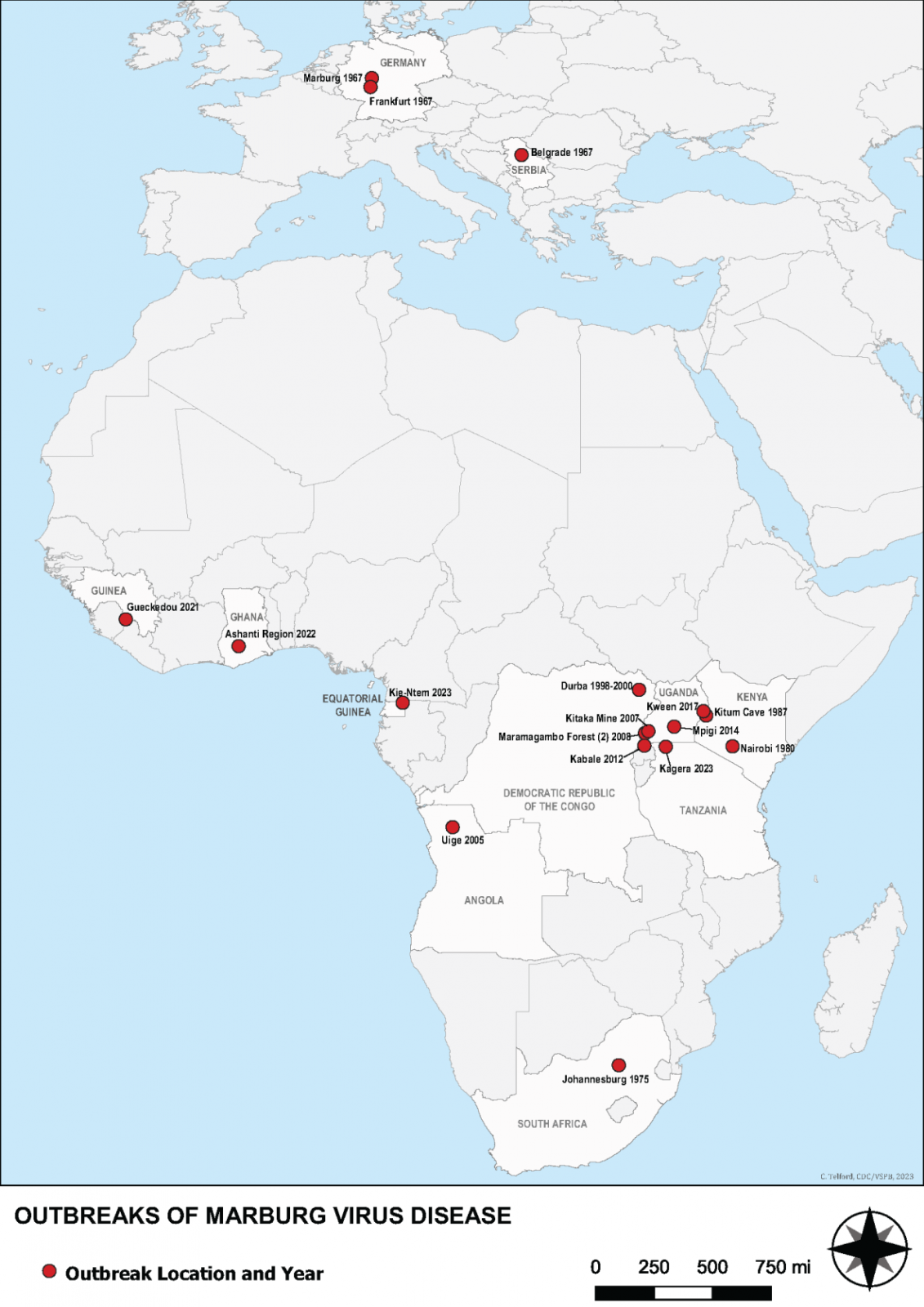
The Sabin Vaccine Institute today announced it launched a Phase 2 clinical trial for its vaccine candidate against the Marburg virus disease (MVD).
As of October 20, 2023, there are currently no vaccines or antiviral treatments approved to treat Marburg, a filovirus in the same family as the virus that causes Ebola.
Marburg has a case fatality rate of up to 88%.
Based on the ChAd3 platform, Sabin’s single-dose investigational Marburg vaccine was found to be promising in a Phase 1 clinical trial.
Dr. Betty Mwesigwa, deputy executive director of the Makerere University Walter Reed Project, is the principal investigator for the Kampala portion of the Sabin-sponsored trial.
A few weeks later, participants will also be enrolled at a second site at the Kenya Medical Research Institute, with Dr. Videlis Nduba as principal investigator.
In a press release, Amy Finan, Sabin’s Chief Executive Officer, commented, “Sabin’s Phase 2 clinical trial builds on a solid safety and immunogenicity foundation, and we hope it will generate the information needed to move the vaccine toward licensure.”
In addition to the Sabin vaccine candidates, other Marburg vaccines are conducting clinical trials.
According to Sabin, the number of MVD outbreaks in Africa has climbed steadily in recent years.
Two outbreaks of Marburg virus disease have occurred in 2023: Equatorial Guinea reported its first documented Marburg outbreak, which killed 12 people, followed by Tanzania, where six people succumbed to the virus.
Communities in Uganda and Kenya are familiar with Marburg, having been ravaged by outbreaks over multiple years in the last few decades.
MVD was first observed in 1967 during outbreaks in Germany.
The U.S. Centers for Disease Control and Prevention published Health Alert Network CDCHAN-00489 on April 6, 2023, confirming no cases of MVD have been reported in the U.S.