Search API
A U.S. CDC-funded Original Research study concluded that two doses of recombinant zoster vaccine (RZV) were highly effective, although less effective against Herpes Zoster (shingles) than in the previous clinical trials.
Published by the Annals of Internal Medicine on January 9, 2024, this Real-World Setting study included nearly 2 million persons who contributed 7.6 million person-years of follow-up.
After adjustment, the vaccine efficacy (VE) of 1 dose was 64%, and VE of 2 doses was 76%.
After one dose only, VE was 70% during the first year, 45% during the second year, 48% during the third year, and 52% after the third year.
After two doses, VE was 79% during the first year, 75% during the second year, and 73% during the third and fourth years.
These findings underscore the importance of the second vaccine dose, wrote these researchers.
Currently, the CDC recommends the Shingrix® vaccine to prevent herpes zoster and related complications for most adults aged 50 and older.
There is no specific time you need to wait before administering Shingrix to patients who have had herpes zoster.
However, it would be best if you did not give Shingrix to patients who are experiencing an acute episode of herpes zoster or a pregnant woman.

According to a Medical News Brief by Emily Harris with The JAMA Network, observational data from more than 1.6 million people in the United States suggests that a high-dose vaccine may also be more effective than standard-dose vaccines for certain adults.
Reported in the New England Journal of Medicine in December 2023, the Original Article evaluated a recombinant vaccine that contains more than triple the amount of influenza hemagglutinin protein compared with standard-dose vaccines.
Flu cases were about 15% lower for people aged 50 to 64 who received the recombinant vaccine.
Because standard-dose vaccines only prevent up to 40% to 60% of influenza cases, decreasing the number of cases by an additional 15% would still “provide a substantial public health benefit, especially during more severe influenza seasons,” the researchers wrote.
As of December 31, 2023, the U.S. CDC reported that over 155 million egg, cell, and nasal-based influenza vaccines, which are generally available at local pharmacies, had been distributed this flu season.

Curevo Vaccine today announced positive data from a Phase 2 trial of amezosvatein (a non-mRNA, adjuvanted subunit vaccine also known as CRV-101) head-to-head versus the U.S. FDA-approved Shingrix® vaccine.
According to the Company's press release on January 7, 2024, Amezosvatein met all primary endpoints in the randomized, controlled, observer-blind Phase 2 trial, including demonstrating non-inferiority to Shingrix as measured by humoral immune response.
Amezosvatein also exhibited lower rates of solicited local and systemic adverse events. Amezosvatein was engineered to have a best-in-class safety profile.
Like Shingrix, amezosvatein uses a subunit protein antigen called glycoprotein 'E' (gE).
Targeting the gE antigen is proven to elicit a long-term, protective immune response to prevent shingles.
Also, like Shingrix, amezosvatein uses an adjuvant targeting the TLR4 pathway to boost the immune response to the gE antigen.
"Fewer than 5% of eligible adults in most European countries and China have received both doses of Shingrix," noted George Simeon, Curevo's Chief Executive Officer, "and two-thirds of adults in the USA still need to be immunized against shingles.
As of January 8, 2024, various shingles vaccines are available in over 30 countries.
Based upon these clinical study results, Curevo will advance amezosvatein into global Phase 3 trials in 2024 to address a market for shingles vaccination currently exceeding $4 billion.
Virtually all adults have been exposed to the varicella-zoster virus, and around 30% will develop shingles at least once in their lifetime.
Also called 'herpes zoster,' shingles occur when the varicella-zoster virus causing childhood chickenpox re-emerges from sensory ganglion nervous system cells where the virus lies dormant after initial exposure.

The Philadelphia Department of Public Health reported today that there is a cluster of measles among unvaccinated residents.
As of January 4, 2024, in addition to an index case, three locally acquired cases have been confirmed, with two of those cases being hospitalized and released.
Furthermore, there are at least two more possible cases that have been hospitalized.
If you visited any of these sites on the days listed at this link, you may have been exposed to measles.
These measles cases are connected with the notice issued on December 23, 2023.
Health Commissioner Dr. Cheryl Bettigole commented in a press release, “Unfortunately, we are seeing cases of measles that have spread to vulnerable individuals, including young children, due to people declining vaccination and also failing to adhere to quarantine recommendations."
The Health Department is strongly recommending that anyone who may have been exposed to measles should quarantine themselves by staying home and away from others.
Additionally, people who have not received both doses of a Measles, Mumps, and Rubella (MMR) vaccine should talk with their healthcare provider about getting caught up.

AstraZeneca and Sanofi recently announced China's National Medical Products Administration approved Beyfortus™ (Nirsevimab-alip) for the prevention of respiratory syncytial virus (RSV) lower respiratory tract infection (LRTI).
Beyfortus is the first approved single-dose (50mg) preventive option to protect infants born healthy at term, preterm, or with specific health conditions that make them vulnerable to severe RSV disease entering or during their first RSV season.
Beyfortus is anticipated to be available in China during the upcoming 2024-2025 RSV season. Its administration can be timed to the start of the local RSV season.
RSV is the most common cause of LRTD in infants, including bronchiolitis and pneumonia, and is also a leading cause of hospitalisation in all infants. China ranks among countries with a high prevalence of RSV infections.
Iskra Reic, Executive Vice President, Vaccines and Immune Therapies, AstraZeneca, commented in a press release on January 2, 2024, "Beyfortus represents the first opportunity to prevent serious respiratory disease due to RSV for all infants in China."
"We look forward to making Beyfortus available for the 2024/25 season."
While Beyfortus was approved in the United States in 2023, its availability has been limited. As of December 14, 2023, the manufacturers committed 1.4 million doses for delivery in the U.S. before February 2024.
Canada, the European Union, and the United Kingdom have also approved Beyfortus.

Pfizer Canada announced the Health Canada approved its bivalent Respiratory Syncytial Virus (RSV) vaccine ABRYSVO™ Vaccine.
ABRYSVO is indicated for the prevention of lower respiratory tract disease caused by RSV in individuals 60 years of age and older by active immunization, and the prevention of lower respiratory tract disease and severe lower respiratory tract disease caused by RSV in infants from birth through 6 months of age by active immunization of pregnant women.
"Vaccines are considered to be the most effective tool in public health for preventing illness and can help reduce the stress on our healthcare system and professionals, including nurses, doctors, and others on the frontline," said Dr. Darine El-Chaâr, Maternal Fetal Medicine physician at The Ottawa Hospital, in a press release on January 4, 2024.
"Vaccines administered through maternal immunization can also help play a critical role in decreasing the gap of vulnerability in the first few months of an infant's life, as well as helping to protect their mothers, who may be at increased risk of severe disease compared with non-pregnant women."
RSV is a common respiratory virus that causes mild disease with cold-like symptoms in the fall and winter months.
In more vulnerable populations, like adults 60 and over, an RSV infection can be severe, especially among those with underlying respiratory or cardiac conditions.
For infants, RSV infection can result in respiratory distress, especially in those less than six months of age and those with higher risk factors such as congenital heart or lung disease or prematurity.
Pfizer Canada is currently assessing the availability timeline for ABRYSVO and is committed to bringing a supply of this vaccine to Canadians as quickly as possible.
As of January 5, 2024, RSV vaccines and monoclonal antibody therapy that deliver passive immunization to infants are approved for use in the United States.

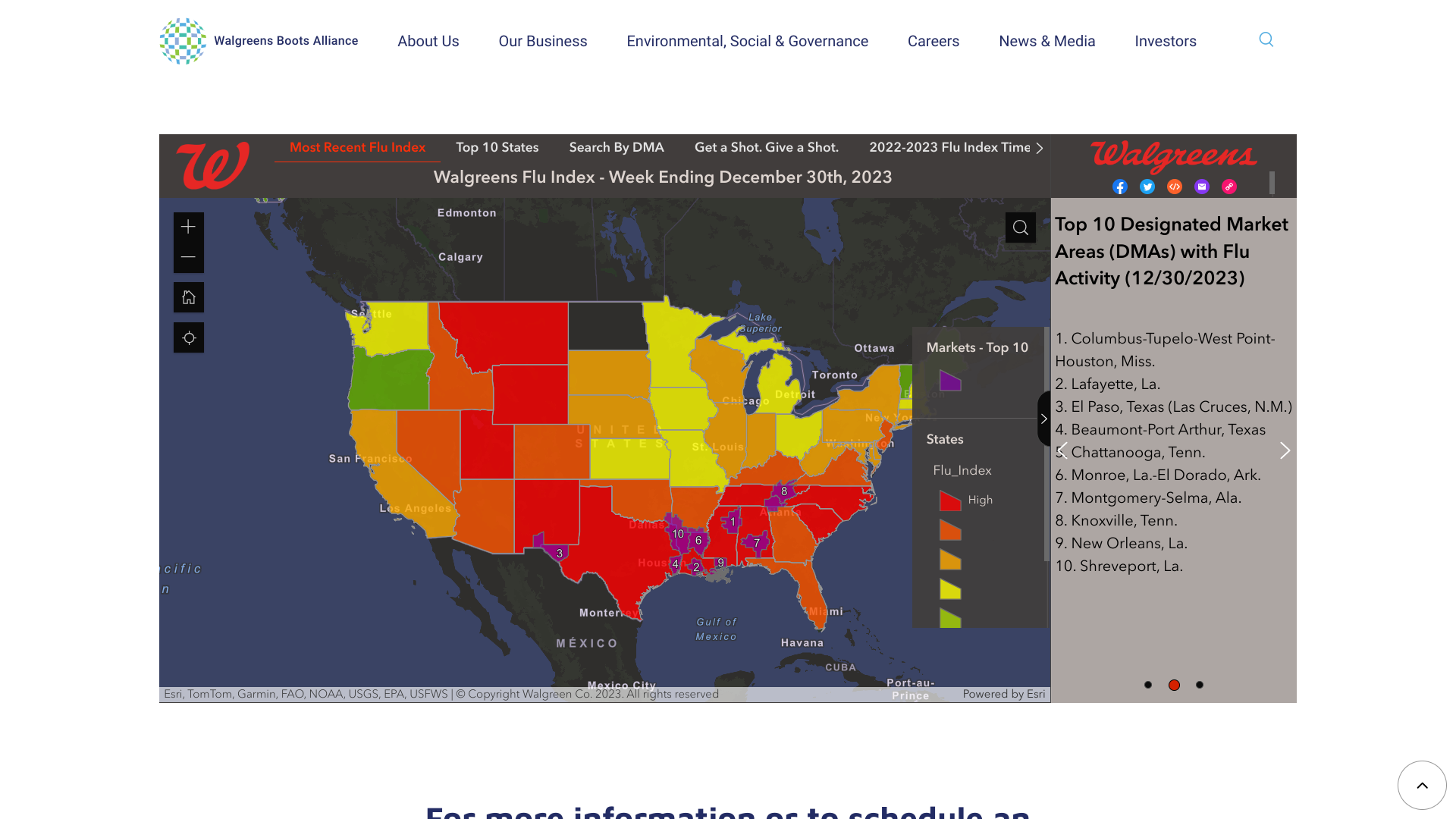
The Walgreens Flu Index® recently rendered updated information regarding influenza activity compiled using retail prescription data for antiviral medications used to treat influenza across Walgreens pharmacy locations.
As of December 30, 2023, the Index identified the top ten cities impacted by the flu last week:
- Columbus-Tupelo-West Point-Houston, Miss.
- Lafayette, La.
- El Paso, Texas (Las Cruces, N.M.)
- Beaumont-Port Arthur, Texas
- Chattanooga, Tenn.
- Monroe, La.-El Dorado, Ark.
- Montgomery-Selma, Ala.
- Knoxville, Tenn.
- New Orleans, La.
- Shreveport, La.
Previously, the U.S. CDC reported seasonal influenza activity was elevated in most parts of the country.
As of December 29, 2023, outpatient respiratory illness was above baseline for the eighth consecutive week and in all 10 HHS Regions.
From a prevention perspective, the CDC encourages most people to get an annual flu shot.
And for those who catch the flu, prescription antiviral drugs that treat influenza are available at most pharmacies in the U.S.
Sanofi Canada recently announced the National Advisory Committee on Immunization's annual Statement on Seasonal Influenza Vaccine for 2024-2025 continues to recommend Fluzone® High-Dose Quadrivalent among the preferential influenza vaccines to protect adults 65 years of age and older against influenza.
Delphine Lansac, General Manager, Vaccines Canada, Sanofi, commented in a press release on January 3, 2023, "This long-standing recommendation as a preferential influenza vaccine for adults 65 years of age and older speaks to the science that Fluzone® High-Dose Quadrivalent is built on and demonstrates our continued leadership in vaccines."
"It's been a groundbreaking year, where over 50% – more than one in two – seniors who received influenza vaccines in Canada were immunized with Fluzone® High-Dose Quadrivalent Influenza Vaccine."
Fluzone® High-Dose Quadrivalent Influenza Vaccine is designed to protect seniors with four times the active component dosage of a standard flu shot.
To date, Fluzone® High-Dose is available in more than 20 countries, including the United States, Canada, and Germany.
As of December 23, 2023, over 155 million flu shots had been distributed in the U.S. this season.