Search API
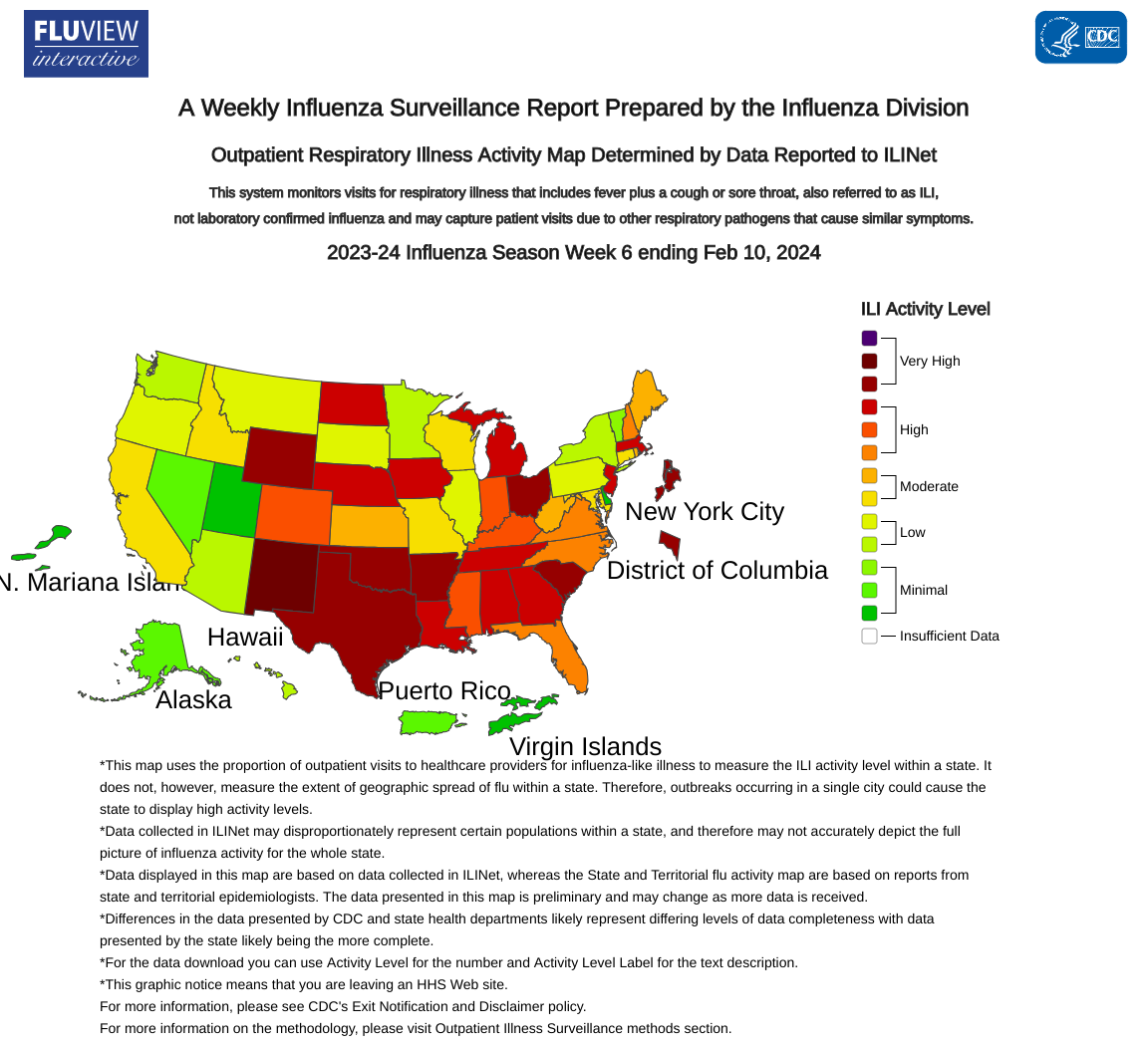
The U.S. Centers for Disease Control and Prevention (CDC) has released an update for Week #6 of the 2024 flu season.
As of February 16, 2024, the overall national percent positivity for influenza has remained stable.
However, there has been a slight decrease in percent positivity for influenza A and a slight increase in percent positivity for influenza B. This change has been primarily driven by U.S. regions 3, 5, and 7 activity.
CDC estimates that there have been at least 16,000 deaths from influenza so far this season.
During Week 6, eight influenza-associated pediatric deaths were reported, bringing the 2023-2024 flu season total to 82.
During the last flu season, there were 182 pediatric influenza-related deaths.
The CDC added vaccination can still provide benefits this season. Everyone six months and older can get an annual flu vaccine at a local pharmacy if influenza viruses spread.
As of early February 2024, about 156 million flu shots had been distributed.
A new meta-analysis published by the Lancet concluded preventive products for respiratory syncytial virus (RSV) can have a substantial public health impact by preventing RSV-severe outcomes in preterm infants.
According to this article posted on February 14, 2024, preterm infants accounted for about 25% of RSV hospitalizations.
This systematic review and meta-analysis found early and late preterm infants had higher RSV-associated ALRI incidence and hospitalization rates than all infants of any gestational weeks.
And the increased risk of hospitalization among early preterm infants persisted into the second year of life.
One explanation is that early preterm children are at higher risk of underlying medical conditions with long-term impacts, such as chronic lung diseases and bronchopulmonary dysplasia, as shown by the ad-hoc exploratory analysis of the prevalence of comorbidities, wrote these researchers.
The U.S. CDC says preterm birth is when a baby is born before 37 weeks of pregnancy. In 2022, preterm birth affected about 10% of infants born in the U.S.
Approved RSV preventive products in 2024 include vaccines and monoclonal antibody (mAb) passive immunization.
In 2023, the phase 3 MELODY clinical trial assessed the efficacy of Beyfortus™ (Nirsevimab), a U.S. FDA-approved mAb, in infants born at a gestational age of at least 35 weeks.
This study found in term and late-preterm infants, a single dose of Beyfortus provided consistent protection against hospitalization for RSV-associated lower respiratory tract infection and severe, medically attended RSV-associated lower respiratory tract infection during an RSV season.

Following the end of the recent pandemic, many travelers explored new destinations in 2023. However, over 667,000 cholera cases and 4,000 deaths were reported last year.
In countries experiencing cholera outbreaks, international travelers protected themselves with safe and effective vaccines.
Since the beginning of 2023, there have been 24 reactive cholera vaccination campaigns implemented in 12 countries.
But, there is a global shortage of cholera vaccines. From January 2023 to January 2024, 76 million oral cholera vaccines (OCV) were requested by 14 countries, while only 38 million doses were available.
The World Health Organization (WHO) has three pre-qualified OCVs: Dukoral®, Shanchol™, and Euvichol®.
One of these OCV manufacturers reported a 72% increase in revenues today.
On February 15, 2024, Valneva SE reported its revenue for 2023. Last year, sales of Valneav's DUKORAL® vaccine amounted to €29.8 million (US32 million), a significant increase from the €17.3 million recorded in 2022.
This significant increase can be attributed to the recovery of the private travel markets and price hikes. However, the company says that foreign currency fluctuations caused a reduction of €0.9 million in DUKORAL® sales.
In a press release, Peter Bühler, Valneva's Chief Financial Officer, commented, "In 2023, Valneva successfully executed key strategic objectives despite a difficult economic environment.... we are entering 2024 in a good financial position to support our commercial and R&D objectives."
In the United States, access to cholera vaccines is limited.

Vaxxinity, Inc. announced today that it has published data from multiple non-human primate studies that demonstrate the VXX-401 vaccine candidate consistently reduces low-density lipoprotein cholesterol (LDL-C) in non-human primates.
VXX-401 is a synthetic peptide vaccine designed to stimulate the immune system to produce antibodies targeting proprotein convertase subtilisin/kexin type 9 (PCSK9), which reduces circulating LDL-C by inhibiting the breakdown of low-density lipoprotein receptor (LDLR).
Across three preclinical studies in cynomolgus monkeys, VXX-401 induced a strong and durable antibody response against PCSK9 and robust, sustained reduction of LDL-C over time.
Prolonged exposure with VXX-401 resulted in an average of 44% LDL-C reduction.
VXX-401 was well tolerated and did not induce any toxicity or pathology beyond mild injection site reactions.
Previous studies have demonstrated that blocking PCSK9 yields lower LDL-C levels and reduces the risk of adverse cardiovascular events.
The company says these results suggest that VXX-401 could be a safe and effective anti-PCSK9 immunotherapy.
The first-in-human Phase 1 clinical trial of VXX-401 is ongoing, with topline results expected in mid-2024.
"Vaxxinity is committed to providing scalable, accessible, game-changing solutions for worldwide heart health," said Mei Mei Hu, CEO of Vaxxinity, in a press release on February 15, 2024.
"Despite multiple approved medications for LDL-C reduction, heart disease remains the number one killer in the world. A cholesterol vaccine like VXX-401 may provide a cost-effective and widely deployable solution that could benefit hundreds of millions of people at risk."
"A well-tolerated intervention that people can start early in life and remain on for many years, lowering the cholesterol' area under the curve,' has the potential to help us win the fight against heart disease."
The publication can be found in the Journal of Lipid Research (Volume 65, Issue 2, 100497, February 2024).

During the first six weeks of 2024, various cities in the United States have reported measles cases, mainly related to unvaccinated international travelers.
On February 13, 2024, the Minnesota Department of Public Health (MDH) confirmed two measles cases had been reported this year. CBS News revealed that the first case was found in the Twin Cities metro area, and the second occurred in an unvaccinated sibling.
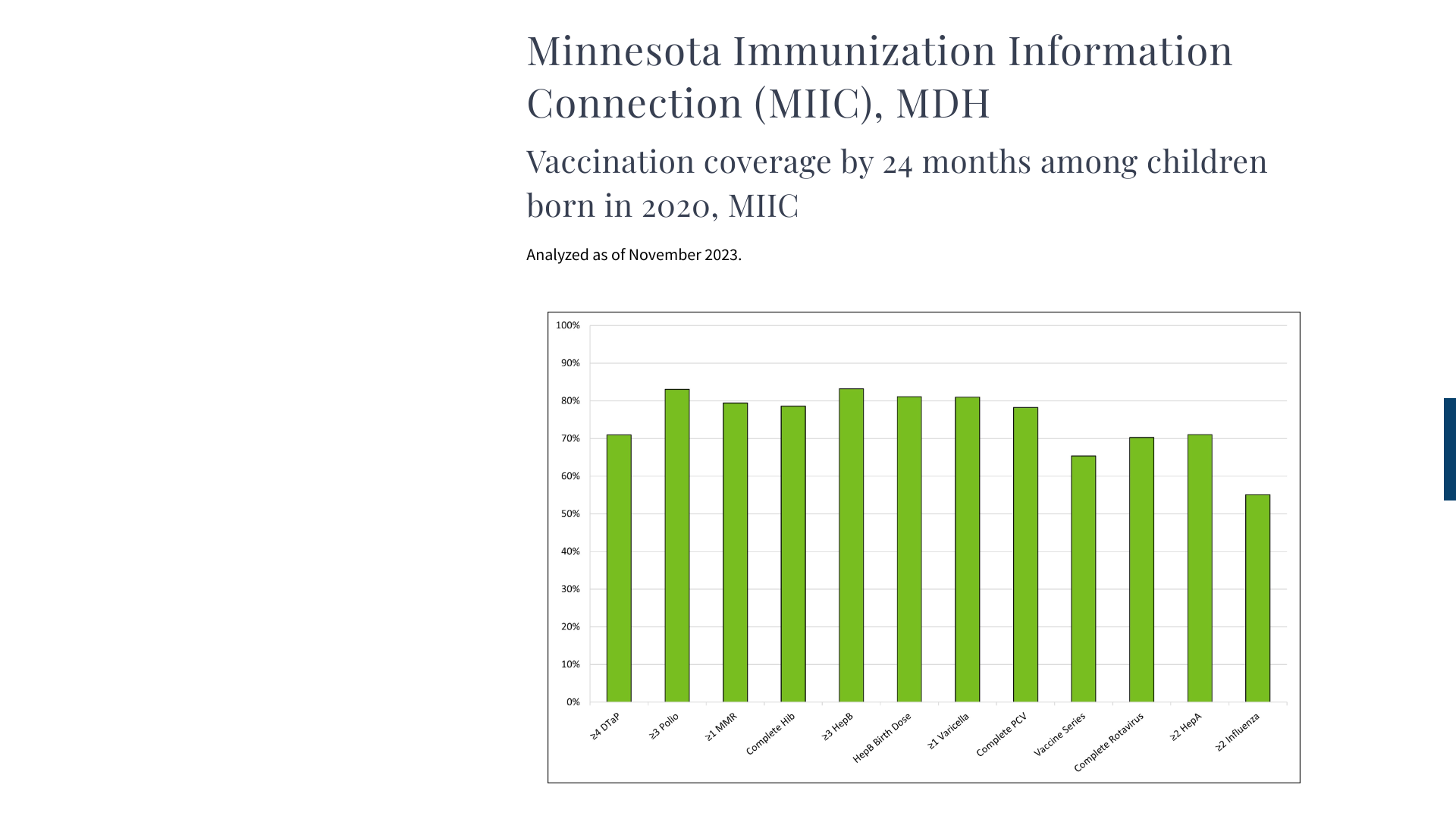
MDH says measles is a vaccine-preventable disease, but recent data indicates Minnesota is trailing the national vaccination rate.
According to MDH data, about 80% of young children were vaccinated with a measles vaccine as of November 2023. MDH reported (0) measles cases in 2023, but 22 in 2022.
Nationally, the U.S. CDC has reported a total of 9 measles cases in four jurisdictions in 2024.
Internationally, the CDC says over the past year, Yemen (18,464), Azerbaijan, Kazakhstan, and India have reported the most measles cases. In 2023, over 534,000 suspected measles cases were reported in 169 jurisdictions.
To alert travelers, the CDC maintains a global Watch-Level 1, Practice Usual Precautions, Travel Health Notice, that identifies measles outbreaks in 47 countries.
Brii Biosciences Limited today announced that it has entered into agreements with VBI Vaccines, Inc., ensuring expansion and control of future clinical and commercial supplies of BRII-179, a late-stage clinical asset in Brii Bio's HBV functional cure portfolio.
Brii Bio confirmed on February 13, 2024, that it will initially issue a $2.5 million promissory note to VBI. This will eliminate royalty and milestone payments for PreHevbri. The note will increase to $10 million upon meeting specific conditions, securing all of VBI's intellectual properties for BRII-179, with associated payments also eliminated.
In addition, subject to certain approvals, Brii Bio and VBI will work together to transfer the manufacturing technologies of BRII-179 to a site designated by Brii Bio.
Upon completing essential activities relating to such technology transfer, subject to certain potential adjustments, Brii Bio will issue up to an additional $8 million promissory note to VBI.
After satisfaction of certain conditions, Brii Bio will also take control of VBI's Rehovot-based manufacturing facilities for BRII-179 and PreHevbrio™ (PreHevbri®, Sci-B-Vac®) for $10 million cash on or after June 30, 2024, when Brii Bio and VBI plan to enter into supply agreement under which Brii Bio will become VBI's commercial supplier for PreHevbrio and PreHevbri.
Separately, subject to achievement of certain conditions by VBI, Brii Bio will secure an exclusive license to develop and commercialize VBI-1901, VBI's glioblastoma immunotherapeutic candidate, in the Asia Pacific region excluding Japan and issue a $5 million promissory note to VBI. VBI-1901 has received fast-track and orphan drug designations from the U.S. Food and Drug Administration and is conducting a Phase 2b study.
Dr. Zhi Hong, Ph.D., Chairman and Chief Executive Officer of Brii Bio, stated in a press release, "As Brii transitions to late-stage development of HBV programs, a global manufacturing strategy becomes critically important."
"We look forward to working with the biologics manufacturing experts at the Rehovot site and timely integration of our R&D and manufacturing capabilities."
According to the U.S. CDC, Hepatitis B is a vaccine-preventable liver infection caused by the HBV. It is spread when blood, semen, or other body fluids from a person infected with the virus enter the body of someone who is not infected. Not all people newly infected with HBV have symptoms, but for those that do, symptoms can include fatigue, poor appetite, stomach pain, nausea, and jaundice.

GSK plc announced today that the US Food and Drug Administration (FDA) has granted Fast Track designation for bepirovirsen, an investigational antisense oligonucleotide (ASO) for the treatment of chronic hepatitis B (CHB).
GSK said on February 12, 2024, Bepirovirsen is the only single agent in phase III development that has shown the potential to achieve clinically meaningful functional cure response when combined with oral nucleoside/nucleotide analogues (NAs).
The FDA designation was requested based on the potential for bepirovirsen to address an unmet medical need for CHB, a serious and life-threatening condition.
Data from the phase IIb trials B-Clear and B-Sure, which evaluated the efficacy, safety, and durability of the response of bepirovirsen in people with CHB, were submitted to support the application. A confirmatory phase III program, B-Well, is ongoing.
CHB affects nearly 300 million people worldwide, and current treatment options offer a less than 2-8% functional cure rate, which is not clinically meaningful.
Currently, available oral antiviral therapies only suppress the virus and do not directly lower hepatitis B surface antigen, which is essential for a functional cure.
Bepirovirsen is a triple-action investigational antisense oligonucleotide. It is also being investigated as a potential backbone therapy in future sequential regimens to pursue functional cures in a broader population of patients with CHB.
FDA Fast Track designation is intended to facilitate the development and expedite the review of drugs to treat serious conditions and fill an unmet medical need.
Bepirovirsen (previously known as 'ISIS 505358 or IONIS-HBVRX') was discovered and is jointly developed with Ionis Pharmaceuticals.
GSK is a global biopharma company that aims to unite science, technology, and talent to get ahead of disease together. Find out more at gsk.com. GSK's unedited press release is available at this link.

As Lunar New Year festivities take place worldwide, there have been reports of avian influenza outbreaks in Asia and sporadic cases of human infections.
In light of this health risk, the World Health Organization (WHO) issued an updated Disease Outbreak News (DONs) on February 13, 2024, with prevention advice titled "Avian Influenza and Lunar New Year Festivities: Vigilance and Precautions" in February 2024.
During February 2024, WHO avian influenza alerts were issued for:
- Influenza A (H1N1) variant virus - Brazil, 7 February 2024
- Avian Influenza A (H5N1) - Cambodia, 8 February 2024
- Influenza A(H1N1) variant virus - Spain, 9 February 2024
- Avian Influenza A(H10N5) and Influenza A(H3N2) coinfection - China, 13 February
The WHO says vigilance remains crucial, although most human infections have been sporadic following contact with infected poultry and/or their environments, with no evidence of sustained human-to-human transmission.
Birds are the natural hosts for avian influenza viruses.
After an A(H5N1) virus outbreak in 1997 in poultry in Hong Kong SAR, China, since 2003, this avian and other influenza viruses have spread from Asia to Europe and Africa. I
Beginning in 2013, human infections with the influenza A(H7N9) virus were reported in China.
WHO DONs provide information on confirmed acute public health events or potential events of concern. For more details, please refer to the WHO Influenza (avian and other zoonotic) factsheet.
As of 2024, the U.S. government has invested tens of millions in vaccines protecting people from certain avian influenza viruses. Furthermore, the U.S. says annual flu shots are unlikely to protect people during avian influenza (bird flu) outbreaks.