Search API
The World Health Organization (WHO) has revealed its recommended composition for influenza vaccines for the northern hemisphere flu season of 2024-2025.
Both trivalent and quadrivalent vaccines are recommended as of February 23, 2024.
These WHO recommendations are utilized by national vaccine regulatory agencies and pharmaceutical companies to develop, produce, and license influenza vaccines for the following influenza season.
Previously, the WHO urged manufacturers to eliminate the B/Yamagata component from flu vaccines for 2024-2025.
As of February 10, 2024, over 157 million flu vaccines (egg, cell, and nasal) had been distributed in the United States during the 2023-2024 season.

Bavarian Nordic A/S, a leading pharmaceutical company, announced news today regarding its investigational chikungunya vaccine, CHIKV VLP.
The European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP) has granted accelerated assessment for this chikungunya vaccine candidate's Marketing Authorisation Application (MAA).
The CHMP has recognized that the vaccine candidate is of significant interest to public health and therapeutic innovation.
With this positive development, the company is taking steps toward addressing the unmet medical needs of millions worldwide affected by the Chikungunya virus.
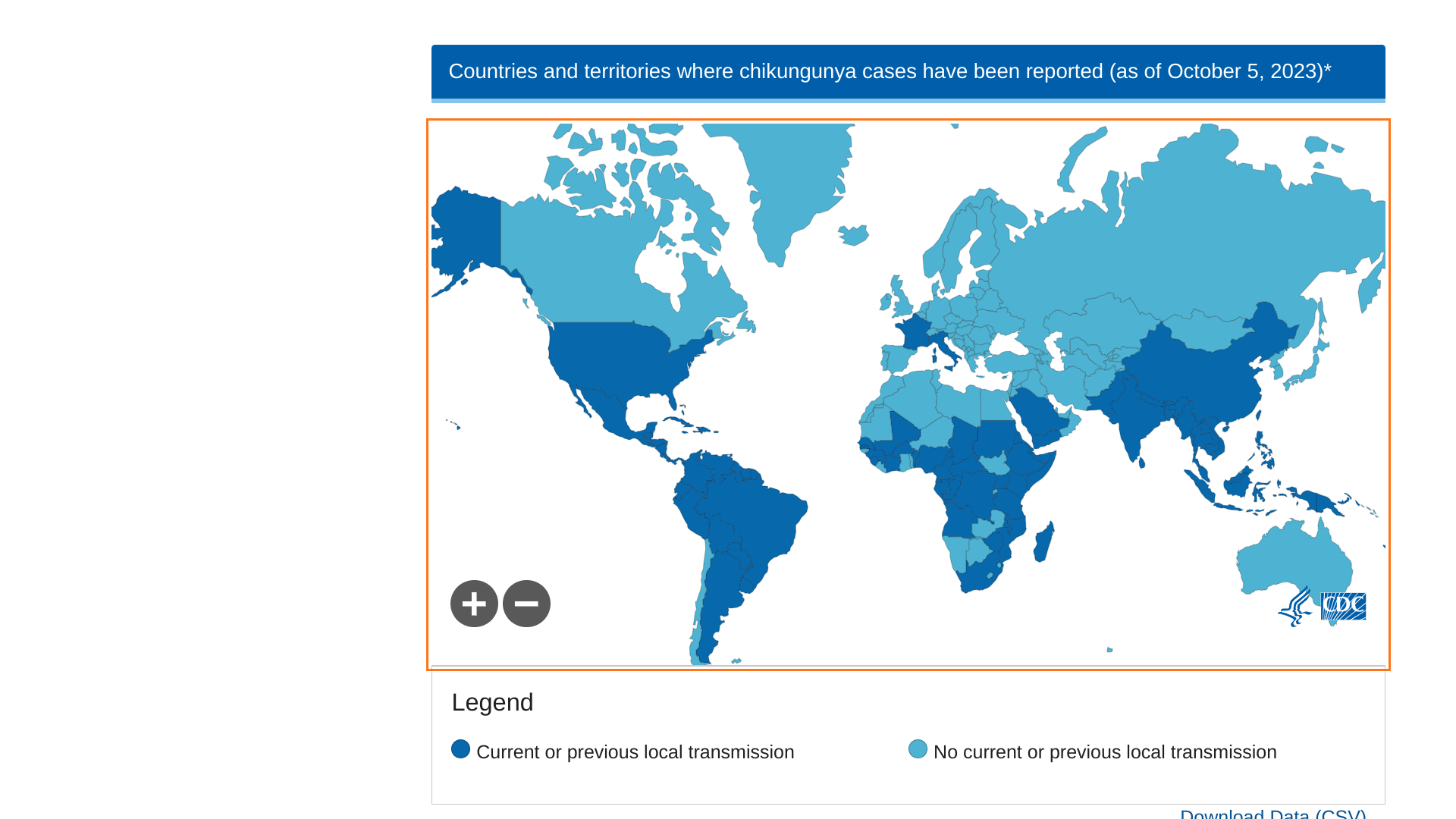
Chikungunya outbreaks continue to be reported in 2024.
Before 2013, chikungunya virus cases and outbreaks had been identified in countries in Africa, Asia, Europe, and the Indian and Pacific Oceans. In late 2013, the first local transmission of chikungunya virus in the Americas was identified in Caribbean countries and territories, according to the U.S. CDC.
Bavarian Nordic also confirmed on February 23, 2024, that it is on track to submit its MAA for CHIKV VLP to the EMA during H1 2024. As a result, the review of the MAA may now take as little as 150 days instead of the usual 210 days.
This means that the vaccine could be available in Europe sooner than expected.
"We are pleased to receive the accelerated assessment in recognition of our chikungunya vaccine candidate and our efforts to bring this novel product to the market. With this, we can accelerate the approval and launch timelines for the vaccine in Europe. As part of our global strategy, we also plan to submit our biologics license application (BLA) for the vaccine candidate to the U.S. Food and Drug Administration later this year," said Paul Chaplin, President and CEO of Bavarian Nordic, in a press release.
In 2023, Bavarian Nordic successfully completed two Phase 3 studies of CHIKV VLP.
The CHKV-VLA vaccine candidate received the U.S. Food and Drug Administration (FDA) Fast Track designation in May 2018.
Recently, the FDA issued approval for Valneva SE's IXCHIQ® Chikungunya Vaccine. However, the CDC has not given its approval.
The UK Health Security Agency (UKHSA) today reported an additional 60 laboratory-confirmed measles cases have been confirmed in England during the ongoing outbreak.
This brings the total number of measles cases since October 2023 to 581.
As of February 22, 2024, the West Midlands accounted for most of these (47%, 79 of 169) measles cases, mainly in Birmingham, the largest city in the West Midlands, with over 1.1 million residents.
Furthermore, the majority (379 of 581, 65%) of these cases are in children.
In a press release, Dr. Vanessa Saliba, UKHSA Consultant Epidemiologist, said, "We're urging parents to protect their children from this serious illness with the MMR vaccine before it spreads further."
"...but 100s of thousands of unvaccinated children are at risk of severe illness or life-long complications."
"The disease spreads very quickly among those who are unvaccinated, especially in schools and nurseries. However, measles is entirely preventable with vaccination."
In the United Kingdom, two MMR vaccine brands are available: Priorix and MMRVaxPro.
The data published in this UKHSA epidemiological overview is currently provisional.
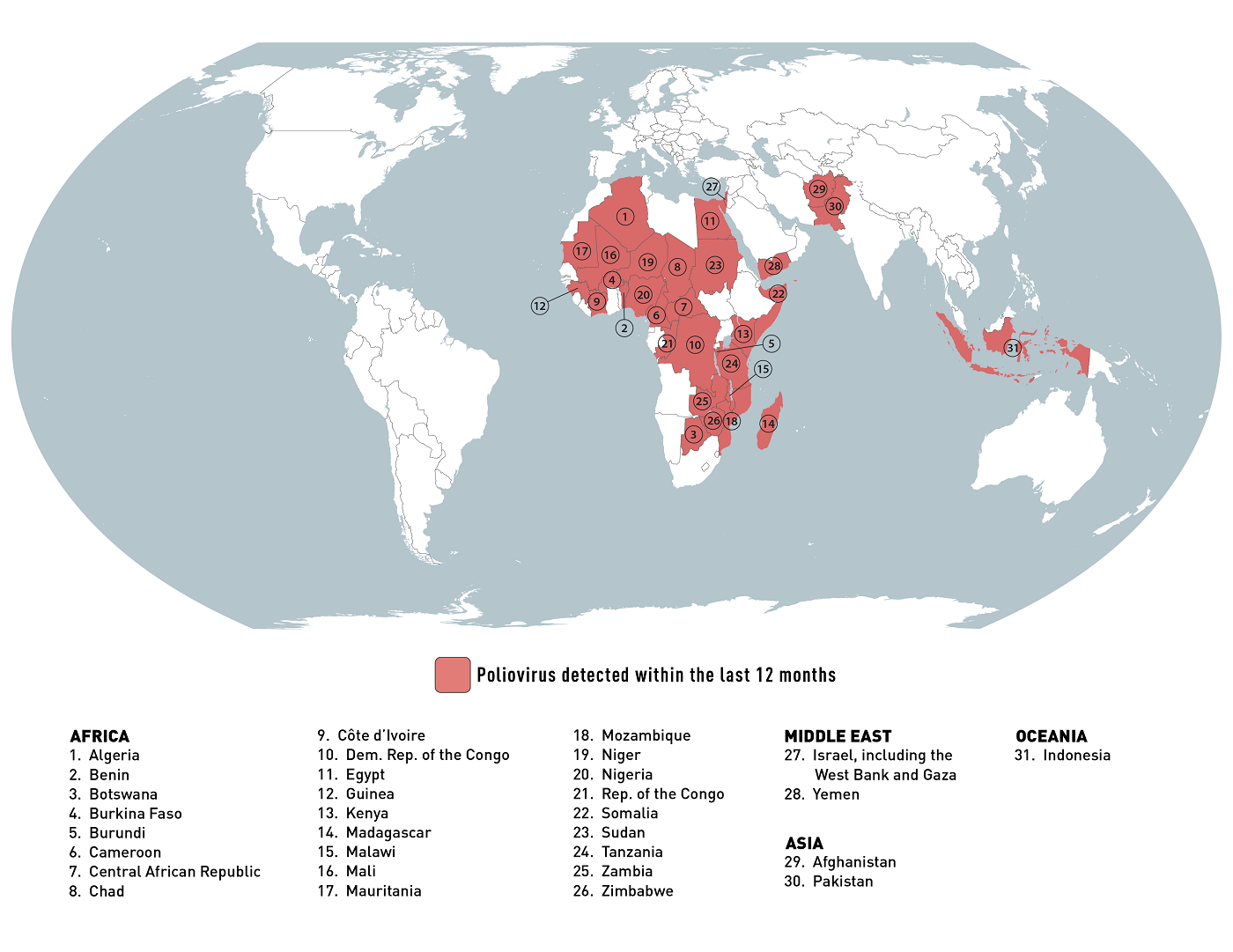
According to the AP, Zimbabwe has initiated a polio vaccination campaign to protect over four million children after identifying three polio cases caused by a rare mutation of the weakened virus found in an older version of the oral vaccine.
On February 20, 2024, the authorities confirmed they would use the type 2 novel oral polio vaccine (nOPV2) polio vaccine designed to minimize virus mutations and reduce polio outbreaks.
Zimbabwe aims to provide this vaccine to approximately 4 million children in February and March 2024.
About 1.4 billion nOPV2 doses have been produced since March 2021.
Since its launch, approximately 1 billion nOPV2 doses have been administered in more than 35 countries worldwide.
As of February 2024, the nOPV2 has not been authorized by the U.S. Food and Drug Administration. However, the U.S. CDC's vaccine committee is reviewing its use case on February 28, 2024.
Zimbabwe is one of 31 countries the CDC lists in its January 2024 global polio outbreak travel alert.
Moderna Inc. today reported $2.8 billion in Spikevax® vaccine sales in the fourth quarter of 2023. The majority of Spikevax sales ($2 billion) were in international sales.
For all of 2023, Spikevax generated $6.7 billion in vaccine sales.
Moderna confirmed in a press release on February 24, 2024, that it achieved 48% cumulative market share in the U.S. retail segment during the fall 2023 COVID season, up from 37% in 2022.
The Company reaffirmed its 2024 product sales outlook as it entered the second year of the U.S. commercial endemic COVID market.
Moderna is also prioritizing key international markets for greater commercial focus and is participating in the EU Health Emergency and Response Authority's tendering procedure for up to 36 million doses of mRNA COVID-19 vaccines per year for up to four years.
As of February 2024, Spikevax is one of 13 COVID-19 vaccines Listed by the World Health Organization.
"2023 was a year of transition for Moderna as we adapted to the endemic market. At the same time, our development team made significant pipeline advancements across infectious diseases, oncology, and rare diseases, while our commercial team increased our COVID-19 market share in the U.S.," said Stéphane Bancel, Chief Executive Officer of Moderna, in a press release.

Novavax, Inc. and Gavi, the Vaccine Alliance (Gavi), today announced they have reached a settlement related to the 2021 Advance Purchase Agreement (APA) for Novavax's prototype COVID-19 vaccine.
To further a joint commitment to public health, Novavax and Gavi have agreed to terms that will prioritize Gavi's and Novavax's shared mission to save lives and protect people's health by increasing equitable and sustainable use of vaccines.
Under the terms, Novavax has made an initial payment of $75 million to Gavi and has agreed to make deferred payments of $80 million annually through December 31, 2028, which are due in quarterly installments and total up to $400 million.
Novavax's annual cash obligation would be offset or reduced pursuant to an $80 million yearly vaccine credit, which may be used for qualifying sales of any of the Company's vaccines funded by Gavi for supply to low-income and lower-middle-income countries.
Using the annual vaccine credit for qualifying sales would reduce Novavax's annual cash obligation.
In addition to the annual obligation, Novavax will provide an additional vaccine credit of up to $225 million, should there be additional demand, which can be applied towards qualifying dose purchases of any of the Company's vaccines in such countries throughout the five-year term.
"Novavax is pleased to have reached this agreement with Gavi as it gives us the ability to continue to work together toward our shared mission of ensuring equitable access to safe and effective vaccines," said John C. Jacobs, President and Chief Executive Officer, Novavax, in a press release on February 22, 2024.
"We look forward to a long-term partnership with Gavi to provide continued access to our protein-based non-mRNA COVID-19 vaccine."
This agreement brings the pending arbitration related to the APA to a close.
As of February 2024, Novavax's COVID-19 Vaccine (Nuvaxovid™XBB.1.5 dispersion for injection, NVX-CoV2601) is the leading protein-based vaccine used in various countries, including the United States.
As of February 22, 2024, Nuvaxovid is one of 13 COVID-19 vaccines Listed by the WHO.
Gavi is a public-private partnership that worked with the World Health Organization and various countries to supply vaccines during the recent pandemic.

ImmunityBio today announced that enrollment and initial follow-up have been completed for the safety portions of a phase 2b clinical trial that is studying an investigational cancer vaccine of a tri-valent combination of antigens delivered by a second-generation Adenovirus vector (Tri-Ad5 CEA/MUC1/brachyury) together with its IL-15 superagonist N-803 for participants with Lynch syndrome.
Each of the three vaccines in Tri-Ad5 targets different proteins associated with precancer and cancer cells.
The vaccine combination studies whether activating dendritic cells and training the immune system to recognize those proteins will destroy the precancer cells before the cancer occurs.
The IL-15 superagonist N-803 is designed to enhance the effects of the vaccines by increasing the proliferation and activation of natural killer and T cells, thereby increasing the potential for cancer prevention in study participants.
The study will include up to 186 participants when fully enrolled and is now open to the randomized controlled portion of the trial.
“We are pleased to be selected to participate in this important and innovative cancer prevention study, one that could provide insights into how the immune system could be harnessed to prevent cancer in individuals with hereditary risk,” said Patrick Soon-Shiong, M.D., Executive Chairman and Global Chief Scientific and Medical Officer at ImmunityBio, in a press release on February 21, 2024.
“With an estimated 5 to 10 percent of cancers inherited, understanding mechanisms that might prevent or delay their onset could potentially change the prospects for tens of thousands of people annually.”
Lynch syndrome is associated with a genetic mutation present in an estimated one million Americans who are more likely to be diagnosed with cancer at a younger age and are at increased risk of developing multiple types of cancers during their lifetime.
ImmunityBio’s Tri-Ad5 Vaccines and N-803 are investigational and are not commercially approved. The safety and efficacy of these investigational agents have not been established by any Health Authority, including the U.S. FDA.
The National Cancer Institute sponsors this study.

The global tuberculosis outbreak continues to impact millions of people in 192 countries and areas. As of 2023, the United Kingdom is no exception.
According to the annual report of UK Health Security Agency (UKHSA) on tuberculosis, there was a 10.7% increase in the number of tuberculosis cases in England in 2023, compared to 2022.
Dr Esther Robinson, Head of the TB Unit at UKHSA, said in a press release on February 15, 2024, "A cough that usually has mucus and lasts longer than three weeks can be caused by a range of other issues, including TB."
"Please speak to your GP if you think you could be at risk."
In 2022, TB notification rates varied widely across England, with the highest in London (1,575 individuals, rate of 17.9 per 100,000) and the lowest in the South West.
People with TB continue to be concentrated in large urban areas, with the highest TB notification rates being in Newham (London) and Leicester City (East Midlands).
In 2022, non-UK-born individuals continued to account for 79.1% of TB notifications in England.
People born in countries in India, Pakistan, Bangladesh, Eritrea, Nigeria, and Romania experience the highest number of cases.
The UKHSA continues to work with partners on a TB action plan, which sets out steps to improve the prevention and detection of TB.
However, the U.K. has reduced access to TB prevention vaccines.
In the United States, the U.S. CDC reported in November 2023 that TB cases increased by 5% in 2022, with 60 U.S. states, the District of Columbia, and territories provisionally reporting 8,331 TB cases.
Furthermore, about 13 million people in the U.S. are living with latent TB infection.
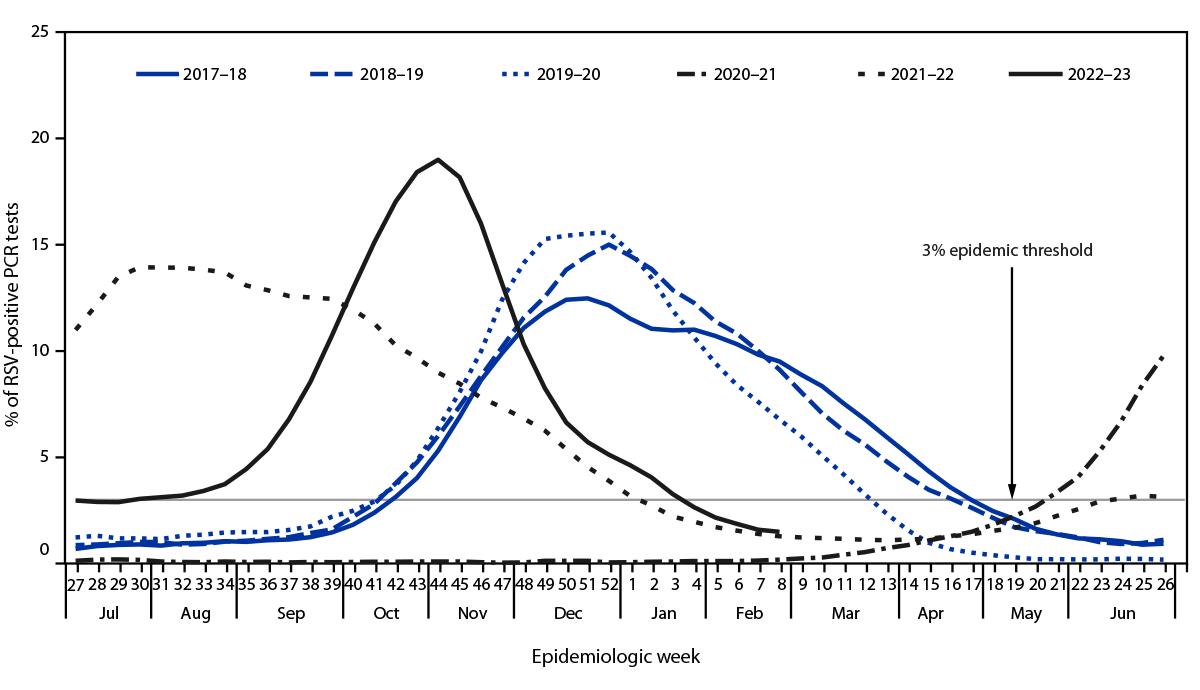
While the respiratory virus season is likely past its peak, it is not over. Respiratory syncytial virus (RSV) outbreaks exist in a few hot spots.
According to the World Health Organization (WHO) Update N° 465, in countries with RSV surveillance, activity was stable or decreased in most reporting countries except in South Africa and New South Wales (Australia), where detections slightly increased in this reporting period.
As of February 19, 2024, RSV positivity was still high in Egypt, though it slightly dropped compared with the last report.
In the United States, the Centers for Disease Control and Prevention (CDC) reported on February 16, 2024, that RSV activity remained elevated but was decreasing nationally.
Other indicators, such as emergency department visits with diagnosed RSV and RSV test positivity, are also decreasing.
From a comparison perspective, the national RSV per capita hospitalization rate remains lower than the peak for last season, says the CDC.
Furthermore, the U.S. market has ample access to approved RSV adult vaccines. At this point in the season, 22% of adults 60 years and older have received an RSV vaccination.
And a monoclonal antibody therapy (Beyfortus™) that delivers passive immunization to infants has become more available.