Search API
In 2024, an unusual number of measles outbreaks have already been confirmed in the United States.
The Centers for Disease Control and Prevention (CDC) reported 35 measles cases recently. Recent measles cases have been reported in Indiana and New Orleans.
In 2024, there has been an alarming increase in the number of measles cases in the United States. The Centers for Disease Control and Prevention (CDC) has recently reported 35 confirmed measles cases.
And in Florida, a media statement issued by the Florida Department of Health on February 23, 2024, confirmed six measles cases in Broward County and a travel-related case in Central Florida (Polk County).
This area of Florida is home to about 2 million people and is adjacent to various entertainment parks and convention centers.
The Florida Department of Health in Broward County confirmed on February 20, 2024, that students at Manatee Bay Elementary School in Weston were infected, and about 200 classmates did not attend class last week.
Suspected measles cases are required to be reported immediately to county health departments or the Department's Bureau of Epidemiology to ensure prompt response and public health efforts. If a measles infection is suspected, please call your local county health department or the Department's Bureau of Epidemiology at 850-245-4401.
These cases relate to the global measles outbreak, generally impacting unvaccinated children and international travelers.
Last year, the CDC published a global Watch-Level 1, Practice Usual Precautions, Travel Health Notice identifying measles outbreaks in 47 countries.
In 2023, over 500,000 suspected measles cases were reported in 169 destinations.

The U.S. Centers for Disease Control and Prevention (CDC) has confirmed Gonorrhea can be cured with the proper treatment for several years.
However, in 2024, the bacteria causing Gonorrhea is recognized by the World Health Organisation as a priority pathogen, for which resistance to existing treatments is rising.
According to industry news, an innovative product may offer better options than the CDC's recommended therapy (ceftriaxone).
GSK plc announced on February 26, 2024, that they had obtained positive headline results from the pivotal EAGLE-1 phase III trial for gepotidacin.
The clinical trial met its primary efficacy endpoint, with gepotidacin (oral, two doses of 3,000mg) demonstrating non-inferiority to intramuscular (IM) ceftriaxone (500mg) plus oral azithromycin (1,000mg), which is a leading combination treatment regimen for Gonorrhea.
Gepotidacin is a potential first-in-class oral antibiotic that has a novel mechanism of action for treating uncomplicated urogenital Gonorrhea in adolescents and adults.
In a press release, Chris Corsico, Senior Vice President, Development at GSK, said, "With rising incidence rates and concern around growing resistance to existing treatments, Gonorrhea threatens public health globally."
"These positive headline results demonstrate the potential for gepotidacin to provide a novel oral treatment option in the face of rising resistance and for patients who cannot take other treatments due to allergies or intolerance."
Detailed results from the EAGLE-1 trial will be presented at an upcoming scientific meeting and shared with global health authorities.
Gonorrhoea is a sexually transmitted infection caused by bacteria called Neisseria gonorrhoeae. It has been estimated that there are 82 million new cases globally each year.
Recently, repurposed vaccines have shown effectiveness against Gonorrhoea, while vaccine candidates conduct late-stage studies.

The US Food and Drug Administration (FDA) announced in a post on X that on May 16, 2024, the Vaccines and Related Biological Products Advisory Committee (VRBPAC) will hold a public discussion to recommend the selection of strain(s) to be included in the 2024-2025 formula for COVID-19 vaccines.
On February 26, 2024, the FDA confirmed that changes to the vaccine composition may be necessary based on the currently circulating strains of the virus that causes COVID-19.
After receiving any recommendations from the VRBPAC regarding the 2024-2025 formula update, the FDA plans to take appropriate regulatory actions on updated COVID-19 vaccines so that manufacturers can make them available by September 2024.
The FDA expects that the composition of COVID-19 vaccines may need to be updated annually, as is done for seasonal influenza vaccines.
Background material and the link to the online teleconference and/or video conference meeting will be available on the Advisory Committee calendar no later than two business days before the VRBPAC meeting.
Oral presentations from the public will be scheduled between approximately 1 p.m. and 2 p.m. Eastern Time.
Contact Information: Sussan Paydar or Prabhakara Atreya, Center for Biologics Evaluation and Research, FDA, 202-657-8533 or [email protected].

It has been reported by local media that the new Pfizer COVID-19 vaccine is now available at some pharmacy chains in Mexico.
According to Mexico News Daily, certain branches of Farmacias del Ahorro, Farmacias Benavides, Farmacias San Pablo, and Farmacias Guadalajara, among others, offer nurse-administered vaccinations.
As of February 20, 2024, Pfizer's updated Comirnaty Omicron XBB 1.5 vaccine costs between $49.78 to $58.65.
The Mexican health regulatory agency authorized the sale of Pfizer's vaccine in December 2023.

In 2024, there have been several reports of avian influenza virus infections in birds, mammals, and humans. As a result, government agencies are taking measures to prepare for potential pandemics.
On February 23, 2024, the Human Medicines Committee of the European Medicines Agency (EMA) issued its recommendations for avian influenza vaccines.
The committee adopted positive opinions for two vaccines intended for active immunization against the H5N1 subtype of influenza A virus, also called bird flu.
One of them, Celldemic (zoonotic influenza vaccine (H5N1)(surface antigen, inactivated, adjuvanted, prepared in cell cultures)), is intended for immunization during outbreaks of influenza coming from animals, including when public health authorities anticipate a possible pandemic.
The other, Incellipan (pandemic influenza vaccine (H5N1) (surface antigen, inactivated, adjuvanted, prepared in cell cultures)), is a pandemic preparedness vaccine intended for use only if a flu pandemic has been officially declared.
In the event of a pandemic, once the virus strain causing the pandemic is identified, the manufacturer can include this strain in the authorized pandemic preparedness vaccine and apply for the vaccine to be authorized as a 'final' pandemic vaccine.
Because the vaccine's quality, safety, and efficacy have already been assessed with other potential pandemic strains, the authorization of the final pandemic vaccine can be accelerated, wrote the EMA.
According to the U.S. Centers for Disease Control and Prevention, about 20 million H5N1 and 12 million H7N9 vaccines were available in the National Strategic Stockpile in 2023.
The U.S. FDA authorized the Audenz™ (Influenza A(H5N1) Monovalent Vaccine, Adjuvanted) cell-based vaccine on January 31, 2020.

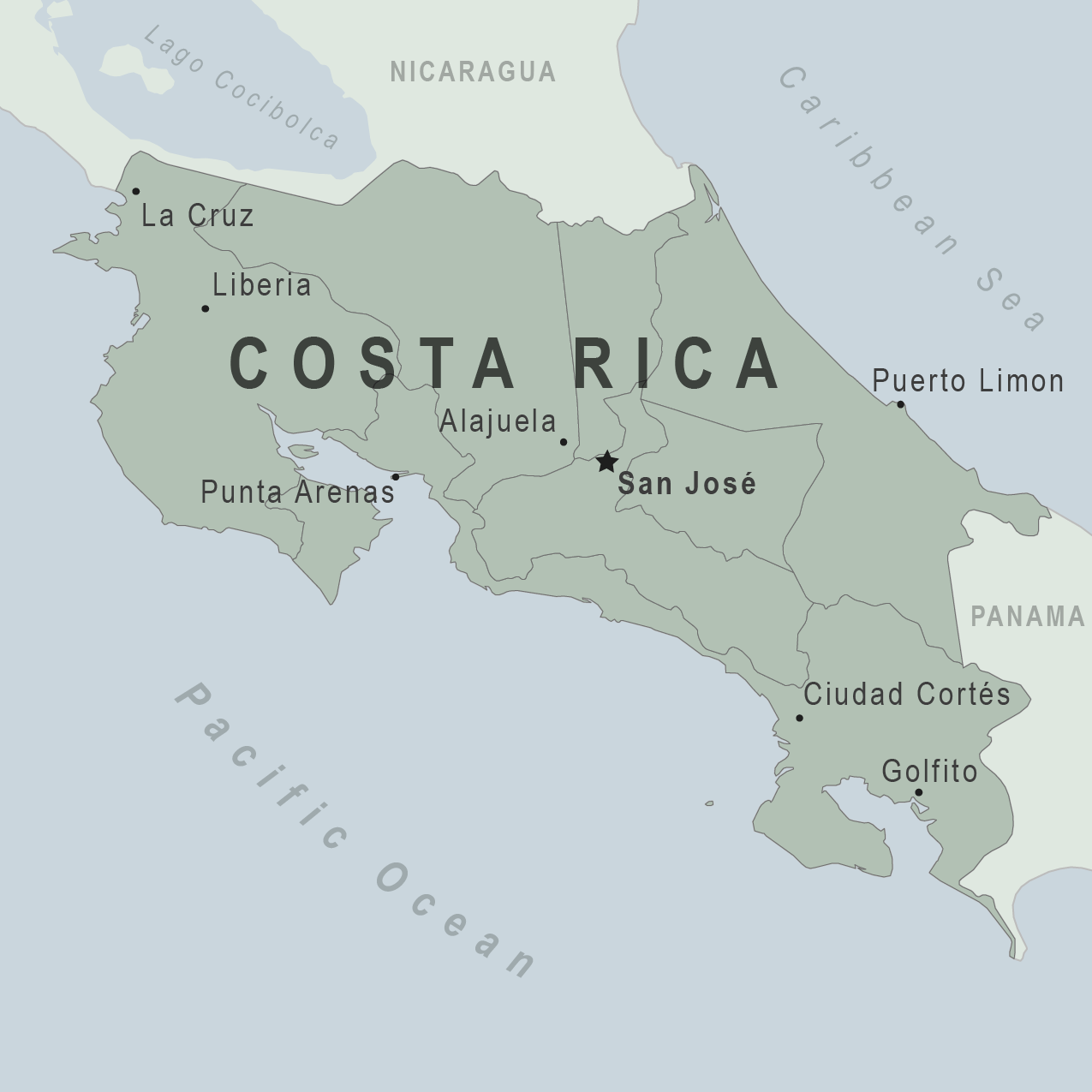
While the global dengue outbreak continues in 2024, many parts of the Americas are at risk. A trendy vacation destination in Central America reports a significant surge in dengue cases this year.
According to the Costa Rica Ministry of Health, there have been 4,787 confirmed cases of dengue during the first six reporting periods of this year.
As of February 23, 2024, Costa Rica's Central North region concentrates the highest accumulated notification of dengue cases this year with 1,228 cases, followed by the Central Pacific with 832 and the Central South with 762 cases.
During 2023, there were over 24,914 dengue cases, an increase from the 7,485 dengue patients in 2022.
To alert international travelers of this mosquito-transmitted health risk, Costa Rica was included in the U.S. CDC's Level 1 - Practice Usual Precautions, dengue notice on February 09, 2024.
The Ministry of Health has called on the population to constantly clean and empty the containers in which water is stored for domestic use. Likewise, residents are urged to collaborate with our officials when they visit your homes for fumigation, presenting proper identification.
The Vector Control Program team recently carried out 16,645 fumigations in the towns of La Carpio, Pavas, and Alajuela.
As of 2024, the Dengvaxia® vaccine was sold in private pharmacies in Costa Rica. The price is around $130.
However, the second-generation QDENGA® dengue vaccine is not offered in Costa Rica.