Search API
A new survey published in the journal Vaccine revealed about 68% of parents in high- and emerging-incidence states would vaccinate their children against Lyme disease.
With Lyme disease vaccines approaching approval, there appears to be significant consumer demand in some U.S. states.
Announced on February 28, 2024, this survey found addressing safety concerns important, and a healthcare provider recommendation could also encourage those unsure or unwilling to be vaccinated.
Given the slight preference for monoclonal antibody passive immunization over-vaccination, particularly in rural areas, access to both may increase Lyme disease prevention.
Initially developed by Valneva SE, the VLA15 Lyme disease vaccine candidate development program was granted Fast Track designation by the U.S. Food and Drug Administration in 2017.
The journal The Lancet Infectious Diseases published results from a Jully 2023 study that concluded Valneva's novel multivalent Lyme vaccine candidate was safe and immunogenic.
VLA15 is currently in advanced phase 3 clinical study.

The U.S. Centers for Disease Control and Prevention's (CDC) Advisory Committee on Immunization Practices (ACIP) voted to recommend that individuals aged 65 and older receive an additional dose of 2023-2024 Formula COVID-19 vaccine four months after receiving the last dose.
According to a Novavax Inc. press release on February 28, 2024, while today's recommendation focuses on protection for older adults at increased risk of COVID-19, information shared at this ACIP meeting also reinforces the ongoing benefit of vaccination for all eligible.
Data from clinical trials continue to show broad long-lived neutralization responses to currently circulating forward-drift variants, including JN.1 and JN.4, for Novavax's protein-based non-mRNA COVID-19 vaccine while maintaining a favorable side effect profile.
Peer-reviewed real-world effectiveness data that shows the immune responses seen in our trials translate into COVID-19 prevention in the real world is being published.
Those interested in a non-mRNA protein-based vaccine should ask their pharmacists about the Novavax COVID-19 Vaccine, Adjuvanted (2023-2024 Formula) (NVX-CoV2601) at major retail pharmacies nationwide, including, but not limited to, Albertsons, CVS Pharmacy and Rite Aid.
Vaccine doses can also be located at us.novavaxcovidvaccine.com or vaccines.gov.
Novavax's Nuvaxovid™ XBB.1.5 is one of 13 COVID-19 vaccines the World Health Organization listed as of early 2024.

The Advisory Committee on Immunization Practices (ACIP) is scheduled to review this draft agenda today at 8 AM EST. This two-day ACIP meeting includes digital access; no registration is required to watch the webcasts.
On the agenda, vaccines to be discussed include COVID-19, Chikyngyna, Influenza, Polio and others.
As previously announced, the ACIP has undergone significant member changes in 2024.
The ACIP develops recommendations for U.S. immunizations, including ages when vaccines should be given, number of doses, time between doses, and precautions and contraindications.
Recent ACIP meeting recommendations are available at this link.
These recommendations are subject to the approval of the U.S. Centers for Disease Control and Prevention director, Dr. Mandy Krauthamer Cohen.

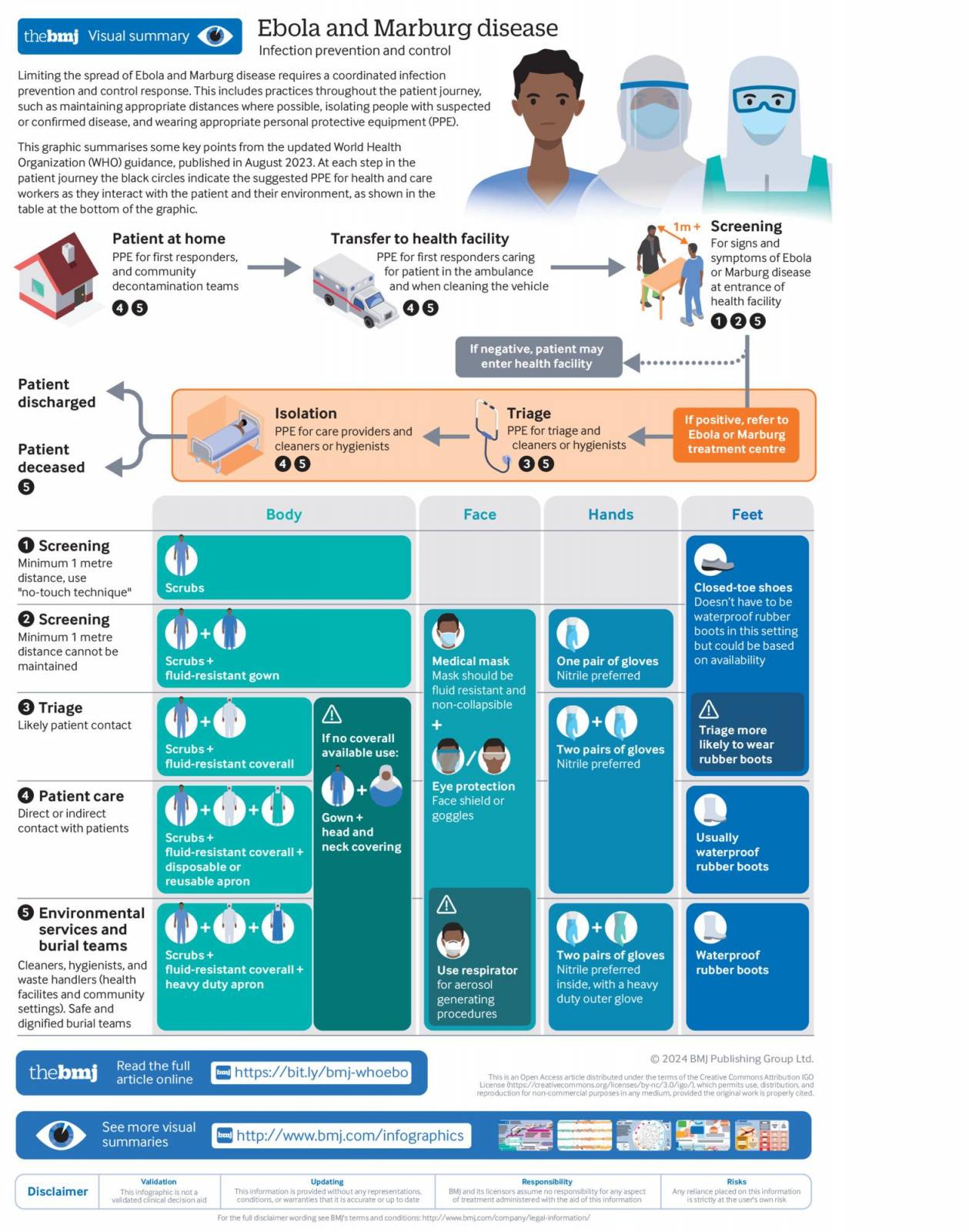
The World Health Organization (WHO) has recently updated its guidelines on infection prevention and control for Ebola disease ten years after the West African Ebola outbreak.
During outbreaks, Ebola infections have relatively high case fatality rates, averaging about 50%.
Therefore, it is crucial to have evidence-based, up-to-date infection prevention and control guidelines to ensure a safe, systematic, and standardized approach during outbreaks, said the WHO in a media release on February 27, 2024.
The complete guidelines are available on the WHO website (WHO/WPE/CRS/HCR/2023.1) and the web-based MAGICapp platform.
Since 2019, various Zaire Ebolavirus vaccines have been approved by the U.S. Food and Drug Administration, European Medicines Agency, and the WHO. These Ebola vaccines are not commercially available in 2024.
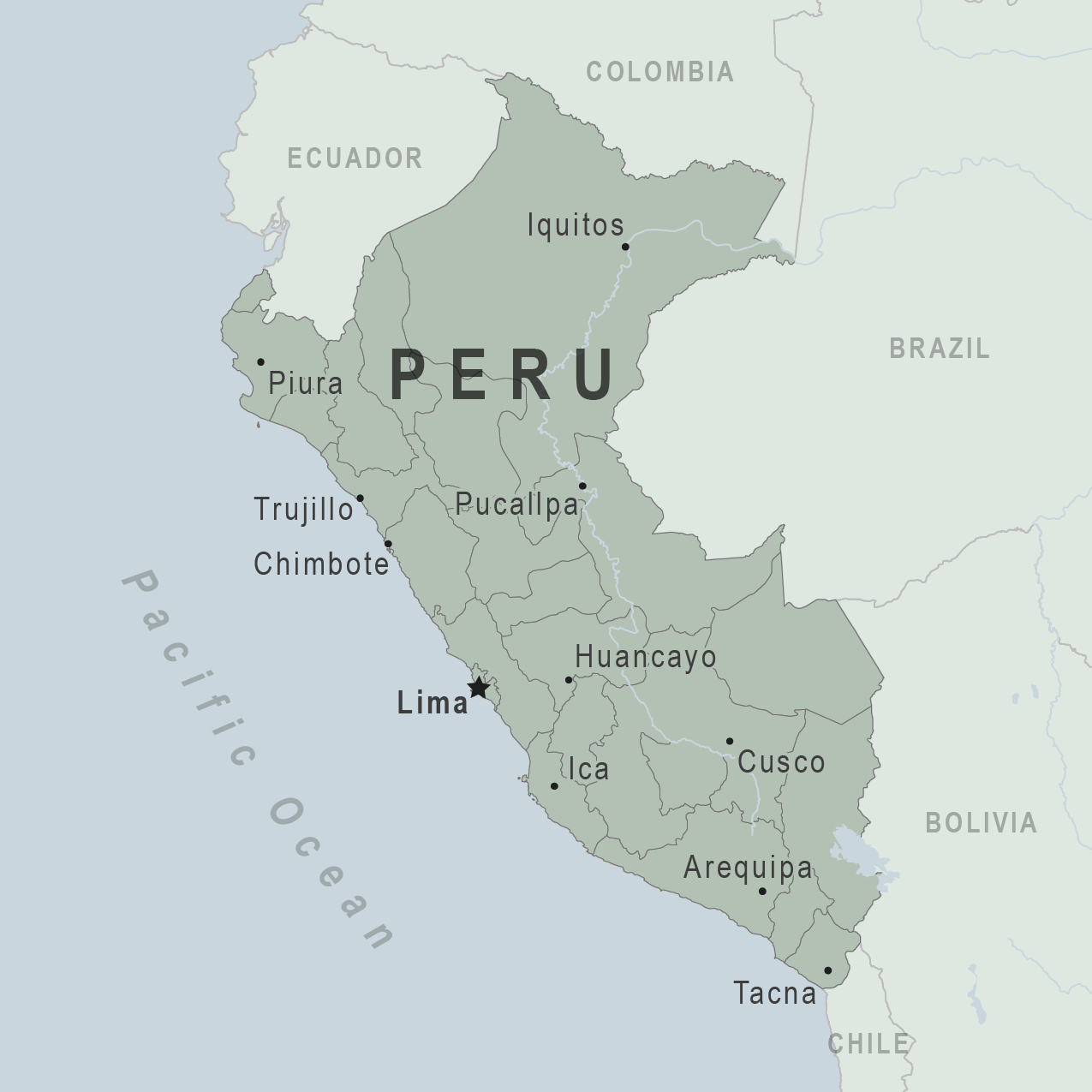
The global outbreak of dengue disease has significantly impacted the Republic of Peru in early 2024. Compared to last year, dengue cases have increased about 90%.
As of February 26, 2024, Peru's Ministry of Health (Minsa) declared a dengue emergency for 20 of the country's 25 regions for the next three months.
Lima, the capital of Peru with about 11 million residents, lies in one of the impacted regions.
However, local media has not reported dengue cases in the tourist hot spot of Machu Picchu.
César Vásquez Sánchez, Minsa's Director, explained that this health emergency declaration will allow more budget to be transferred in the following weeks to confront this disease in the regions better.
Vásquez Sánchez explained there have been 31,364 accumulated cases of Dengue have been reported throughout Peru and 32 related deaths this year.
"As the World Health Organization had already announced a few months ago, 2024 would be a catastrophic year for Latin America regarding dengue fever. The unusual heat wave and the absence of winter have generated a perfect panorama for the spread of Dengue," he mentioned.
While the U.S. Centers for Disease Control and Prevention (CDC) issued a Travel Health Notice regarding dengue outbreaks in the Americas on February 9, 2024, it did not include Peru.
The CDC says Dengue is a disease caused by a virus spread through mosquito bites. It can become severe within a few hours. Severe Dengue is a medical emergency, usually requiring hospitalization.
Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator (CARB-X) today announced it awarded $2.2 million to LimmaTech Biologics AG to advance the development of its novel vaccine candidate targeted to prevent Neisseria gonorrhea (NG) infections.
The CARB-X award supports the development of LimmaTech's vaccine candidate that incorporates multiple antigens commonly found in NG bacteria. The goal is to develop a cost-effective vaccine that elicits a robust immune response against different NG bacterial strains.
"Gonorrhea is becoming increasingly resistant to treatment, which reinforces the pressing need for a highly effective and safe vaccine that can protect against this serious and pervasive pathogen," said Dr. Franz-Werner Haas, CEO of LimmaTech, in a press release on February 27, 2024.
"We believe our proprietary vaccine technology offers advantages in efficacy, production scalability, and simplicity to include multiple antigens that can effectively address bacterial infectious disease threats."
The WHO Global Health Sector Strategy on Sexually Transmitted Infections has set goals for reducing gonorrhea incidence by vaccination by 90%.
As of February 2024, no gonorrhea vaccine candidate has been approved by the U.S. Food and Drug Administration or European Medicines Agency.
However, the U.K.'s Joint Committee on Vaccination and Immunisation agreed in 2023 that a targeted program should be initiated using the 4CMenB (Bexsero®) vaccine for the prevention of gonorrhea in those who are at most significant risk of infection.
LimmaTech is a Swiss clinical-stage biotech company that has built a robust pipeline of innovative vaccines to provide solutions against increasingly dangerous infections, including Staphylococcus aureus and Shigella.

Takeda and Biological E. Limited today announced a strategic partnership to accelerate access to QDENGA® (Dengue Tetravalent Vaccine [Live, Attenuated]) multi-dose vials (MDVs).
MDVs offer economic and logistical advantages for National Immunization Programs by minimizing packaging and storage expenses and reducing medical and environmental waste.
These doses will ultimately be made available for government procurement in dengue-endemic countries by 2030 to support National Immunization Programs.
"Takeda's long-term goal for our dengue program has been to make QDENGA broadly available to those at risk who may benefit from immunization. Within the last year, we've successfully launched in private markets, are now launching in some public programs, and working with partners to support a broader public health impact," said Gary Dubin, M.D., president of the Global Vaccine Business Unit at Takeda.
According to the press release on February 27, 2024, BE will ramp up to a manufacturing capacity of up to 50 million doses a year, accelerating Takeda's efforts to manufacture 100 million doses annually within the decade.
The partnership will build upon existing manufacturing capacity for the vaccine at Takeda's facility in Singen, Germany, and Takeda's long-term collaboration with IDT Biologika GmbH.
Dengue fever is among the most common mosquito-borne viral diseases worldwide. Dengue is endemic in more than 100 countries and causes an estimated 390 million infections yearly.
The Americas, Southeast Asia, and Western Pacific regions are the most seriously affected, with Asia alone representing ~70% of the global disease burden.
In 2024, the state of Florida reported two cases of locally acquired dengue from two counties. In 2023, positive samples from 186 humans were reported from five counties.
QDENGA is currently available in the private market in countries in Europe, Indonesia, and Thailand and in private and some public programs in Argentina and Brazil.
As of late February 2024, QDENGA is not approved by the U.S. FDA.