Search API
The journal Vaccines recently published a Short Communication that presented a benefit–risk assessment for the Novavax COVID-19 protein-based vaccine (NVX-CoV2373).
Published on March 16, 2024, this analysis used data on myocarditis/pericarditis cases observed in the NVX-CoV2373 clinical studies, real-world data of mRNA COVID vaccine effectiveness against predominant SARS-CoV-2 strains in early 2023, and recent COVID-19 burden of disease data from the U.S.
The benefits of NVX-CoV2373 vaccination were estimated as the number of COVID-19 cases, hospitalizations, and deaths prevented. The risks of myocarditis/pericarditis cases and related hospitalizations and deaths occurring within seven days of vaccination were also estimated.
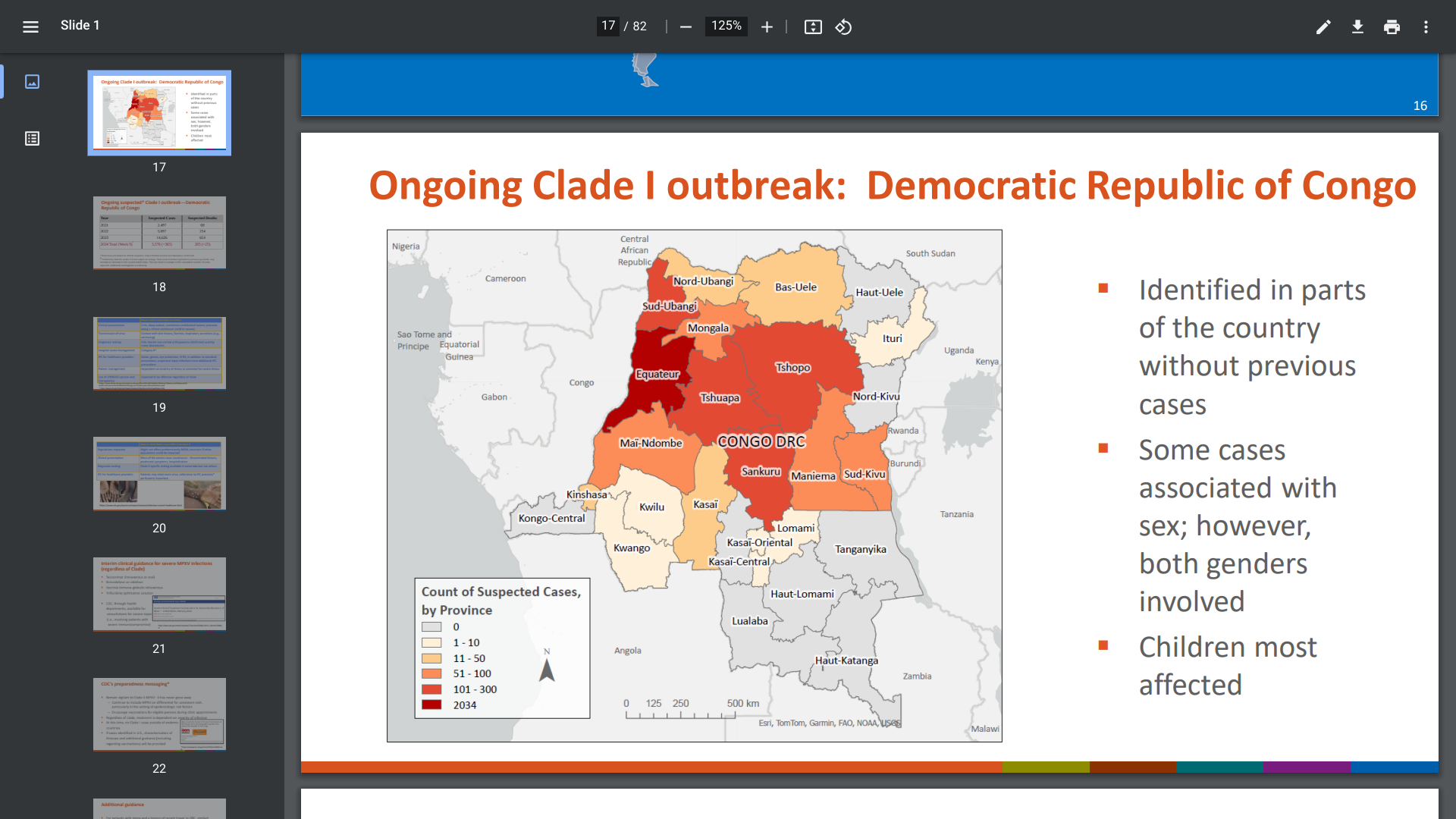
In our analysis, vaccination with NVX-CoV2373, per 100,000 vaccinated, prevented an estimated 1805 COVID-19 cases, compared with an estimated 5.3 excess myocarditis/pericarditis cases.
The number of COVID-19 hospitalizations and deaths prevented were also greater than vaccine-associated myocarditis/pericarditis hospitalizations and deaths.
Our analysis indicates a positive benefit–risk balance for NVX-CoV2373, concluded these researchers.
In October 2023, the U.S. Food and Drug Administration amended the EUA of the Novavax COVID-19 Vaccine, Adjuvanted (Nuvaxovid™ XBB.1.5) for use in individuals 12 and older, to include the 2023-2024 formula.
In the U.S., the current Novavax COVID‑19 Vaccine is available at certain pharmacies, including Costco, CVS Pharmacy, Giant, Publix, Rite Aid, and Stop & Shop.

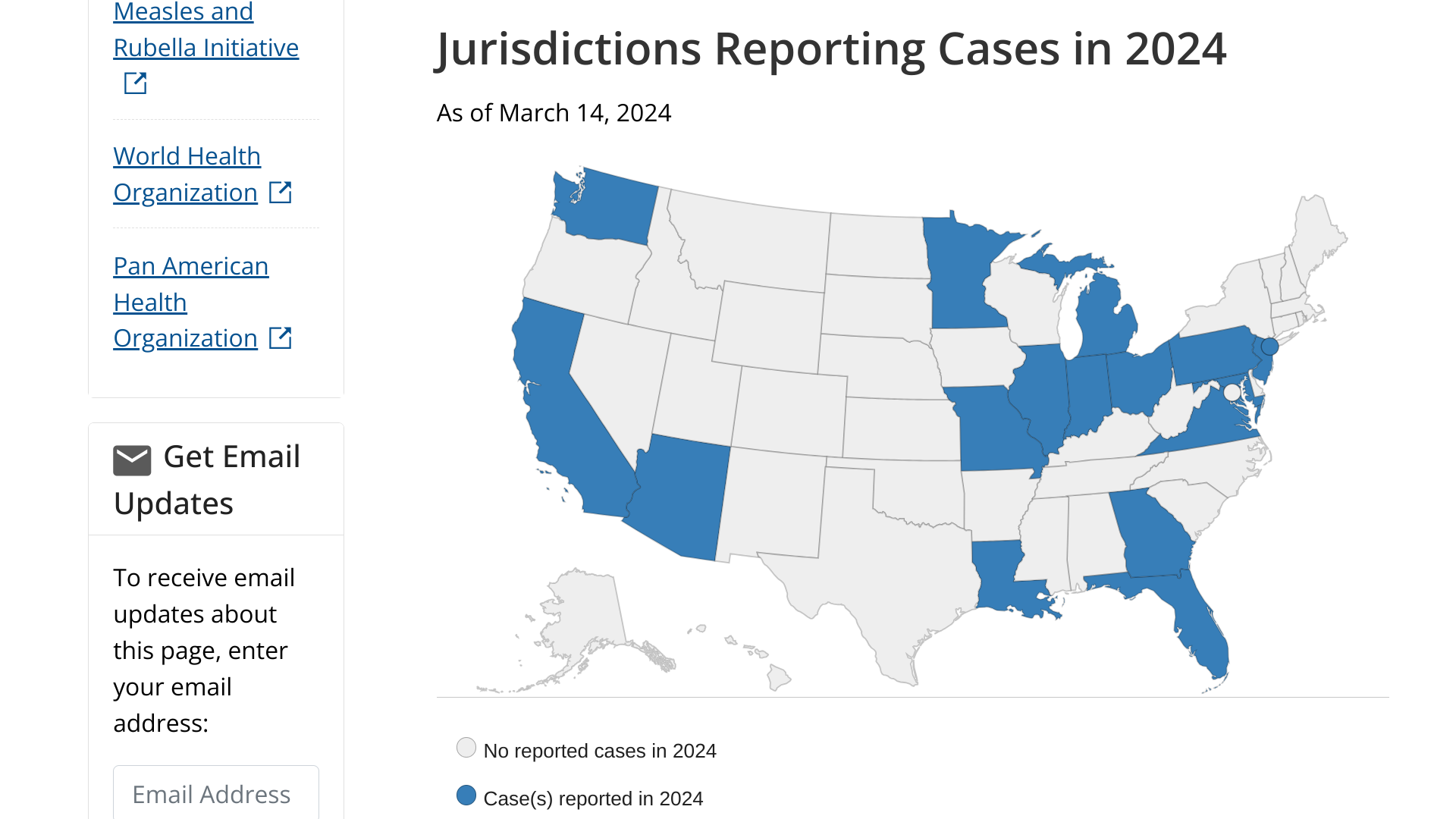
The U.S. Centers for Disease Control and Prevention (CDC) and the World Health Organization say the increased number of measles outbreaks is a global threat in 2024.
As of March 14, 2024, a total of 58 measles cases were reported by 17 U.S. jurisdictions: Arizona, California, Florida, Georgia, Illinois, Indiana, Louisiana, Maryland, Michigan, Minnesota, Missouri, New Jersey, New York City, Ohio, Pennsylvania, Virginia, and Washington.
In 2023, a total of 58 measles cases were reported by 20 U.S. jurisdictions.
In 2024, the Chicago Department of Public Health (CDPH) confirmed twelve measles cases, including an active outbreak at a local shelter in Pilsen. CDPH is coordinating a comprehensive, city-wide response to the first measles cases in Chicago in five years.
CDPH is encouraging all new arrivals to Chicago and every Chicagoan to get the measles-mumps-rubella (MMR) vaccine if they haven't already done so.
The vast majority of Chicagoans are vaccinated against measles and therefore not at high risk," said CDPH Commissioner Olusimbo 'Simbo' Ige, MD, MPH, in a press release on March 12, 2024.
"But those who are unvaccinated need to take precautions. If they're exposed, quarantine immediately and connect with their healthcare provider. Above all else, get vaccinated so you, too, can be protected from this virus."
In the U.S., MMR vaccines are available at most community pharmacies as of March 18, 2024.
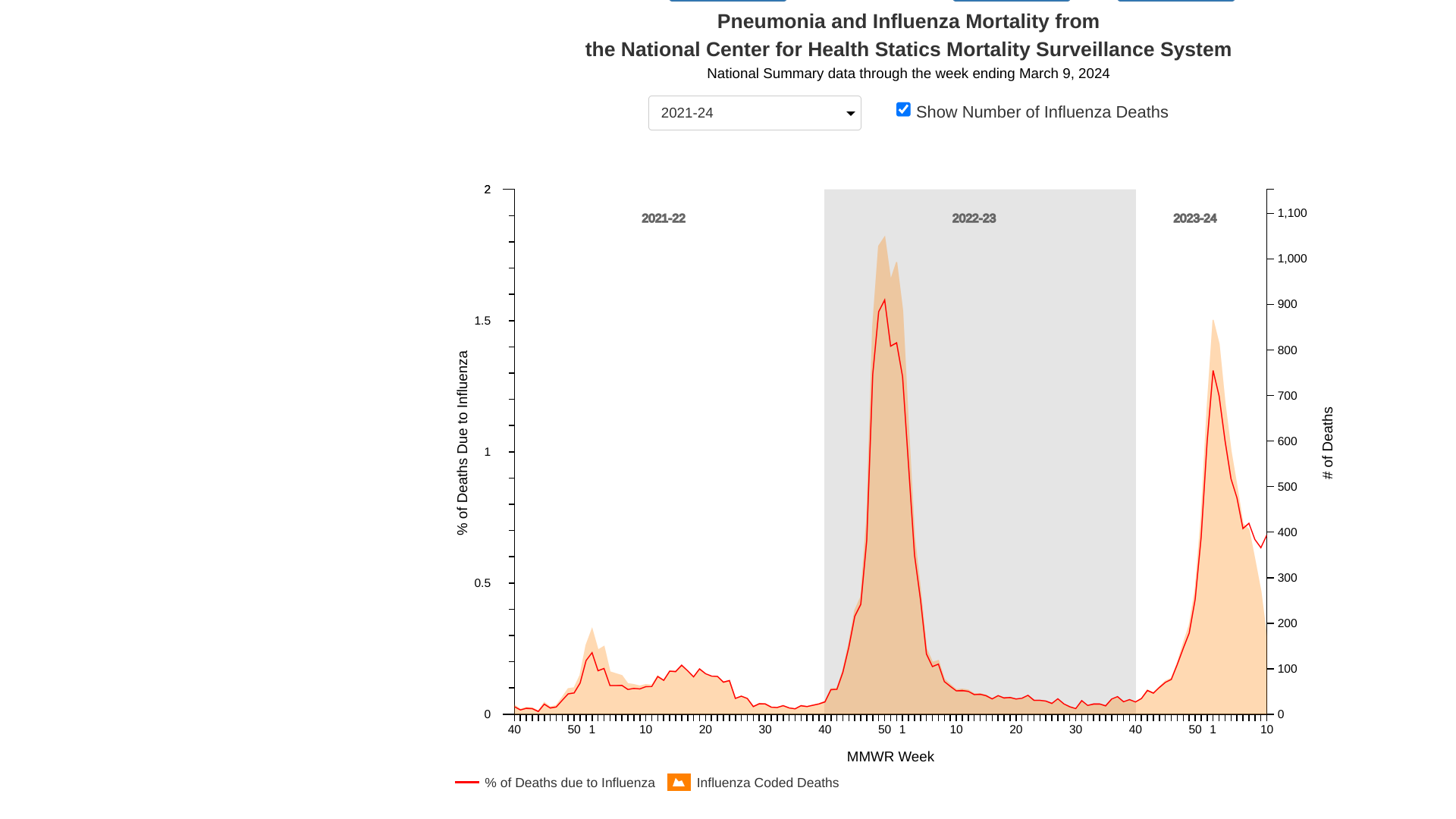
The U.S. National Center for Health Statistics (NCHS) Mortality Surveillance data available on March 14, 2024, indicates that 0.7% of the deaths that occurred during the week ending March 9, 2024 (Week 10) were due to influenza.
This percentage increased (≥ 0.1 percentage point change) compared to last week's data.
NCHS's data also confirmed that 13 influenza-associated pediatric deaths were reported to the U.S. CDC during Week 10. The total number of pediatric deaths for the 2023-2024 flu season is now 116.
As of March 17, 2024, the CDC continues to recommend that everyone six months and older get an annual flu vaccine as long as influenza viruses are spreading. Vaccination can still provide benefits this flu season.
Various egg, cell, and nasal flu shots remain available at most community pharmacies in the U.S.
The composition of flu vaccines is reviewed annually, and new flu vaccines are manufactured each year. For the 2024-2025 flu season, many countries are transitioning to trivalent flu vaccines.
Following detecting a new emergence of variant poliovirus type 2 in six wastewater samples in the Port Sudan locality, Red Sea State, the Republic of Sudan recently announced polio vaccinations will begin in April 2024.
Sudan's Federal Ministry of Health (FMOH) announced on March 11, 2024, that this new detection comes 14 months after Sudan declared an outbreak of variant poliovirus type 2 from an emergence of the virus.
While no vaccination campaign has occurred since April 2023, surveillance for poliovirus in children focused on the most common indicator of a polio infection, acute flaccid paralysis.
While no child has been paralyzed due to the new emergence, detecting poliovirus in wastewater samples puts children across the country at high risk.
"The new detection has only redoubled our commitment to safeguarding our children's future. In collaboration with partners, we are mobilizing an outbreak response campaign to ensure that every child under five years in inaccessible areas receives the polio vaccine, and special plans will follow for hard-to-reach areas," said Dr. Dalya Eltayeb, Director-General of Primary Health Care in Sudan's FMOH, in a media statement.
In October 2022, the FMOH distributed 10.3 million doses of oral polio vaccine (OPV) in Sudan.
This year, Sudan is deploying the WHO-authorized type 2 novel oral polio (nOPV2) vaccine, which has been used 1 billion times by over 30 countries.
nOPV2 is reported to be more genetically stable than OPVs, with a lower risk of reversion to neurovirulence and less likely to mutate and cause paralysis.
While the nOPV2 vaccine is not approved in the United States, the government's vaccine committee considered its use under specific conditions during its February 28, 2024 meeting.
Various reports suggest that Africa's mpox Clade 1 outbreak has recently intensified. As of mid-March, the Democratic Republic of the Congo (DRC), Eurosurveillance, and the World Health Organization released updated mpox outbreak figures for 2024.
These reports revealed 14,626 suspected cases for the last year, with 654 fatalities. This equates to a case-fatality rate (CFR) of 4.5%.
Separately, the U.S. Centers for Disease Control and Prevention (CDC) and the Infectious Diseases Society of America estimated the CFR to be approximately 7.4% in the DRC.
Despite this concerning situation, limited genomic information is available on the circulating mpox viruses, which suggests that they belong to Clade I.
This finding demonstrates that mpox transmission through sexual contact may extend beyond clade IIb, says the CDC.
During a digital briefing on March 14, 2024, Agam Rao, MD CAPT, U.S. Public Health Service, reconfirmed the Advisory Committee on Immunization Practices recommended on October 25, 2023, vaccination with the 2-dose JYNNEOS® (MVA-BN®, IMVAMUNE®) vaccine series for persons aged 18 years and older at risk for mpox.
Since the Clade 2 outbreak began in May 2022, the overall vaccine coverage in the U.S. for one dose is 40%, and for two doses, it is 25%.
The U.S. Food and Drug Administration initially approved JYNNEOS for smallpox in 2019. JYNNEOS remains available in the U.S. at specific clinics and community pharmacies.