Search API
While Diphtheria is a vaccine-preventable disease, recent health alerts indicate various countries have under-vaccinated populations.
As of September 22, 2023, the U.S. Centers for Disease Control and Prevention (CDC) issued Travel Health Notices for Diphtheria outbreaks in the Republics of Guinea and Nigeria in Africa and Vietnam in East Asia.
As an example of recent outbreaks, during July and August 2023, a total of 5,898 suspected Diphtheria cases were reported from Local Government Areas in eleven Nigerian states.
The CDC says if you travel to any affected area, you should be up to date with your diphtheria vaccines. The U.S. agency recommends vaccinating everyone two months and older to protect against Diphtheria.
And if you have immunization questions, discuss the need for a booster dose with your healthcare professional before traveling abroad.
Diphtheria is a serious infection caused by strains of Corynebacterium diphtheriae bacteria that make a toxin that can cause people to get very sick.
This bacteria spread from person to person through respiratory droplets, like from coughing or sneezing, and from touching open sores or ulcers of people sick with Diphtheria.
For some people, respiratory Diphtheria can lead to death. Even with treatment, about 1 in 10 patients with respiratory Diphtheria die. Without treatment, up to half of patients can die from the disease, says the CDC.
Globally, Diptheria cases have been declining since 2019, when 22,986 cases were reported by the World Health Organization (WHO). In 2022, 5,856 cases were confirmed.
Furthermore, the WHO's current vaccination data indicates a high percentage for most countries, including the United States.
During the ongoing mpox outbreak, older smallpox vaccines were used to prevent infection and reduce disease severity in those at increased risk for infection.
However, the effectiveness of smallpox vaccination against mpox was unknown.
On September 21, 2023, the New England Journal of Medicine published a Correspondence that stated previous vaccination at a median of 13 years earlier with either a first- or second-generation smallpox vaccine reduced the likelihood of testing positive for orthopoxvirus, including mpox, among current or former military personnel for whom vaccination data were available.
The estimated vaccine efficacy was 72% for Dryvax (1st generation) and 75% for ACAM2000 (2nd generation).
Among the participants who tested positive for orthopoxvirus, 121 (41%) had been diagnosed with HIV infection (odds ratio, 2.34; 95% CI, 1.65 to 3.35).
Within the VA portion of the study, 19 of 186 participants (10%) required hospitalization; there were no deaths from mpox, and all the participants had an uneventful recovery.
The Department of Defense electronic laboratory data and the Veterans Affairs Corporate Data Warehouse identified eligible participants. The study was approved by the institutional review board of Emory University and was granted an exemption for consent.

The 2019 Lancet Commission on Tuberculosis laid out an optimistic vision for how to build a tuberculosis-free world through smart investments based on sound science and shared responsibility.
Since then, several significant strides have been made toward ending tuberculosis, according to a Review published by The Lancet on September 13, 2023.
With excellent short-course preventive regimens and several late-stage vaccine candidates, tuberculosis prevention is also on the cusp of a revolution.
These clinical tools are needed as more than 7 million people worldwide have died of tuberculosis in the past five years.
Still, much more can be done to implement the Commission's recommendations fully.
For example, the Bill and Melinda Gates Foundation and Wellcome pledged $550 million to support late-stage clinical trials for a new tuberculosis vaccine. The M72/AS01E (M72) subunit vaccine candidate is conducting phase 3 clinical research.
And in the United States, the 100-year-old BCG vaccine is considered for people who meet specific criteria.
According to the U.S. Centers for Disease Control and Prevention, BCG vaccination is considered for children under specific conditions.
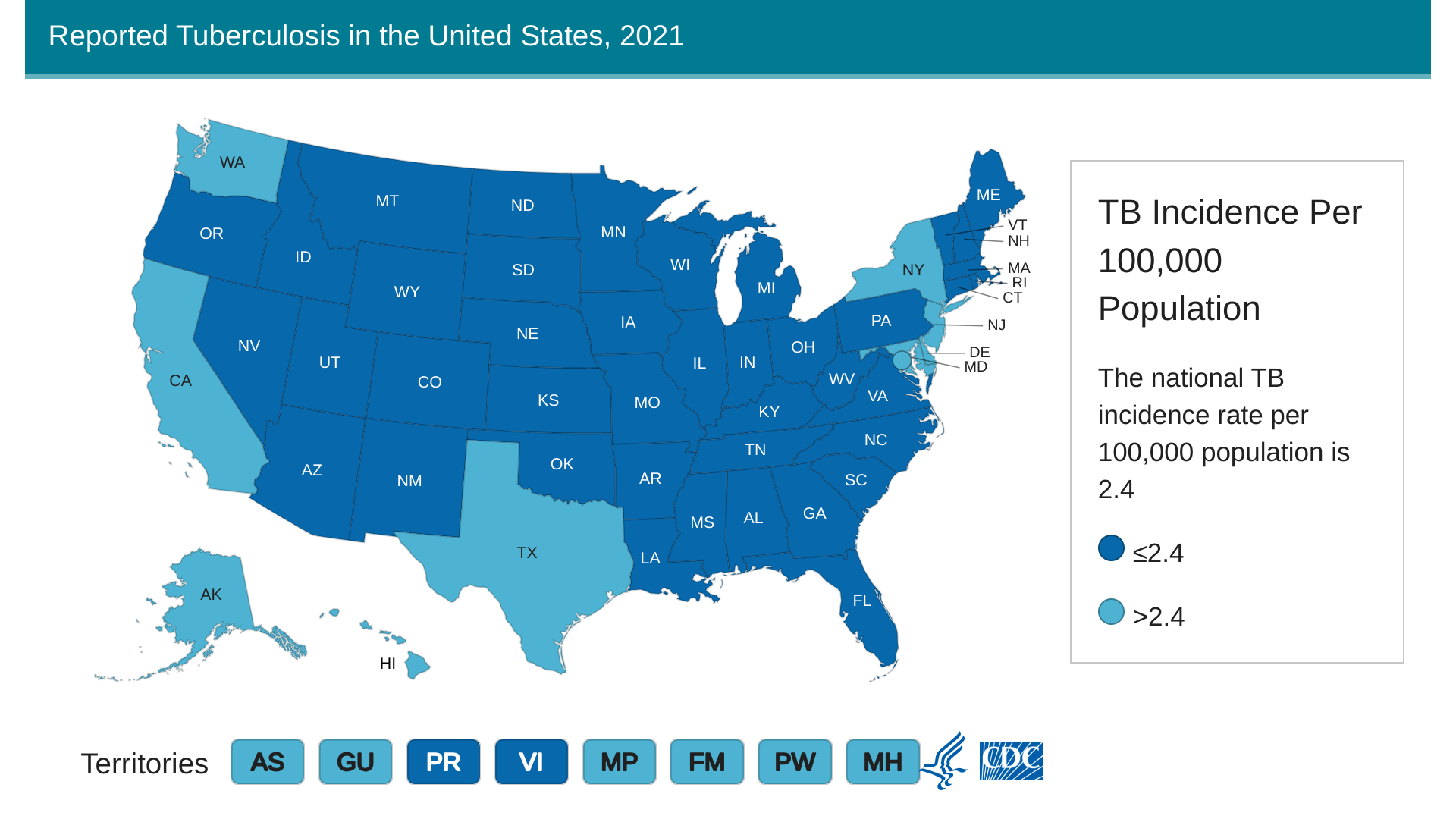
The CDC reported in March 2023 that TB cases increased by 5% in 2022, with 60 U.S. states, the District of Columbia, and territories provisionally reporting 8,300 TB cases.
When the U.S. Food and Drug Administration approved Pfizer Inc.'s ABRYSVO™ Respiratory Syncytial Virus vaccine on August 21, 2023, for use in pregnant women to prevent lower respiratory tract disease (LRTD) and severe LRTD caused by RSV in infants from birth through six months of age, it initiated questions regarding a previous FDA approval.
On July 17, 2023, the FDA approved AstraZeneca and Sanofi's Beyfortus™, a single-dose, long-acting monoclonal antibody (mAb) designed to protect children from LRTD through their first and second RSV season.
Beyfortus has been approved in Canada, Europe, Japan, and the United Kingdom.
With two RSV prevention solutions approved by the FDA, expecting mothers are seeking guidance on 'best practices.'
Hopefully, the U.S. CDC's Advisory Committee on Immunization Practices (ACIP) meeting on September 22, 2023, will add clarity to this clinical decision.
Led by Dr. Grace Lee (Chair) and Dr. Sarah Long (RSV Workgroup Chair), this ACIP's meeting agenda will review and opine on preferred recommendations regarding RSV prevention for pregnant women.
Another consideration is 'when' to become immunized.
RSV seasons in the U.S. generally start in Florida and then spread nationwide.
This means that to optimize RSV protection, make a protection selection before this respiratory virus is detected in your area.
The ACIP develops recommendations regarding U.S. immunizations, which are then ratified or not by the CDC's Director Mandy K. Cohen, MD, MPH.
This ACIP digital meeting is open to the public at this IBM link and starts at 10:00 AM ET this Friday, September 22.

While there are no approved vaccines to prevent human immunodeficiency virus (HIV) infection, several vaccine candidates are progressing in clinical studies.
As an example, Vir Biotechnology, Inc. today announced that the first participant has been dosed in a Phase 1 clinical trial evaluating the safety, reactogenicity, and immunogenicity of VIR-1388, an investigational novel T cell vaccine for the prevention of HIV.
VIR-1388 is based on the human cytomegalovirus (HCMV) vector platform. It is designed to stimulate the body to produce immune cells known as T cells that recognize several HIV proteins in a way that differs from prior investigational HIV vaccines.
“HIV continues to be a major global public health challenge with no approved vaccines despite decades of research efforts,” said Carey Hwang, M.D., Ph.D., Vir’s Senior Vice President, Clinical Research, Head of Chronic Infection, in a press release on September 20, 2023.
“We are hopeful that our unique approach will help close the longstanding public health gap in HIV prevention.”
VIR-1388 was developed using applied learnings from VIR-1111, the Company’s initial investigational proof-of-concept HIV T cell vaccine based on HCMV.
The U.S. National Institute of Allergy and Infectious Diseases and the Bill & Melinda Gates Foundation financially support the clinical study.
The U.S. CDC’s Morbidity and Mortality Weekly Report publishes public health information and recommendations on HIV.

A contagious disease known for causing outbreaks on land and at sea may soon have an approved preventive vaccine. Noroviruses have emerged as the single most significant cause of gastroenteritis worldwide.
According to the U.S. Centers for Disease Control and Prevention, most norovirus outbreaks in the United States happen from November to April. In years when there is a new strain of the virus, there can be 50% more norovirus illness.
While no vaccines are available today in the U.S., U.K., or Europe, a new funding event may accelerate an approval.
HilleVax, Inc. today announced the gross proceeds from an offering, before deducting discounts, commissions, and other expenses, are expected to be $100.0 million.
HilleVax stated in a press release on September 20, 2023, that it intends to use the net proceeds from the offering to fund the clinical development of HIL-214, including certain manufacturing activities, and for working capital and general corporate purposes.
HIL-214 is an investigational virus-like particle bivalent vaccine candidate for preventing moderate-to-severe acute gastroenteritis caused by norovirus in infants.
The HIL-214 vaccine includes antigens from genotypes GI.1 and GII.4 to represent genogroups that cause most human illnesses.
HIL-214 has been studied in various clinical trials, collectively generating data from more than 4,500 subjects, including children. The ongoing phase 2/3 study was last updated in May 2023.
“HilleVax has continued to make excellent progress on our HIL-214 program, including the recent completion of enrollment of over 3,000 subjects in our NEST-IN1 clinical trial,” said Rob Hershberg, MD, PhD, Chairman and Chief Executive Officer of HilleVax, in a press release on May 12, 2023.
“We look forward to continuing to execute on our HIL-214 program and announcing top-line safety and clinical efficacy data in the first quarter of 2024 for our NEST-IN1 clinical trial.”
While other norovirus vaccine candidates are conducting clinical research, the HIL-214 vaccine appears to Precision Vaccinations to be the most advanced candidate.
As the mpox outbreak ends in most countries, other health agencies restock their reserves in case of a smallpox outbreak.
On September 13, 2023, Bavarian Nordic A/S announced that it had been awarded another contract to supply its MVA-BN® smallpox vaccine to rescEU, a strategic reserve within the European Union (EU).
This order follows an initial contract awarded in June 2023 and is also for delivery in 2024.
The vaccines will be stockpiled in another EU country, thus expanding the EU's capability to respond to medical countermeasures to its member states and other countries participating in the rescEU, an EU Civil Protection Mechanism.
Paul Chaplin, President & CEO of Bavarian Nordic, commented in a press release, "We are pleased to continue supporting the initiative from the EU to build a stockpile of smallpox vaccines across Europe."
"This secures another order for next year, confirming an ongoing demand, but also the transition to stockpiling for future outbreaks, which we see as a growing trend with other countries and organizations."
The new contract is valued at more than EUR 11 million.
The MVA-BN or Modified Vaccinia Ankara-Bavarian Nordic vaccine is marketed as IMVANEX® in Europe, IMVAMUNE® in Canada, and JYNNEOS® in the U.S.
In addition to smallpox, the U.S. Food and Drug Administration, Health Canada, and the European Commission have also approved the vaccine for use against mpox. It is the only vaccine having obtained this dual approval to date.
In the U.S., over 1.2 million doses of JYNNEOS have been distributed since the global mpox outbreak began in May 2022. Since then, about 113 countries have reported over 89,000 mpox cases.
Recently, a significant increase in mpox cases has been observed in the South-East Asia Region.
MVA-BN is a non-replicating smallpox vaccine developed in collaboration with the U.S. government to ensure the supply of a smallpox vaccine for the entire population, including immunocompromised individuals who are not recommended vaccination with traditional replicating smallpox vaccines.
RescEU was established by the European Commission as a safety net to protect citizens from disasters and manage emerging risks. It includes, among others, reserves of medical countermeasures which can be deployed in chemical, biological, radiological, and nuclear emergencies.