Search API
The World Health Organization (WHO) Director-General, Dr. Tedros Adhanom Ghebreyesus, recently launched the TB Vaccine Accelerator Council to facilitate the development, licensing, and using new Tuberculosis (TB) vaccines.
The 100-year-old Bacille Calmette-Guérin (BCG) is the only licensed TB vaccine, with over ten versions available globally.
While it provides moderate efficacy in preventing severe forms of TB in infants and young children, it does not adequately protect adolescents and adults, who account for the majority (>90%) of TB transmission globally, said the WHO on September 22, 2023.
In a press release, Dr. Tedros Adhanom Ghebreyesus commented, "Today, we have knowledge and tools they could only have dreamed of."
"The political declaration countries approved today, and the targets they have set, are a commitment to use those tools, and develop new ones, to write the final chapter in the story of TB."
The Council aims to identify innovative sustainable financing, market solutions, and partnerships across public, private, and philanthropic sectors. It will leverage various agencies to strengthen commitment and actions for novel TB vaccine development and access.
The U.S. Centers for Disease Control and Prevention (CDC) reported that TB outbreaks increased by 5% in 2022, with 60 U.S. states, the District of Columbia, and territories provisionally reporting 8,300 TB cases last year.
According to the CDC, TB is caused by Mycobacterium tuberculosis. The bacteria usually attack the lungs, but TB bacteria can attack any body part.
As a result, two TB-related conditions exist: latent TB infection and TB disease. If not treated properly, TB disease can be fatal.
In the U.S., Merck's TICE BCG vaccine has limited availability.

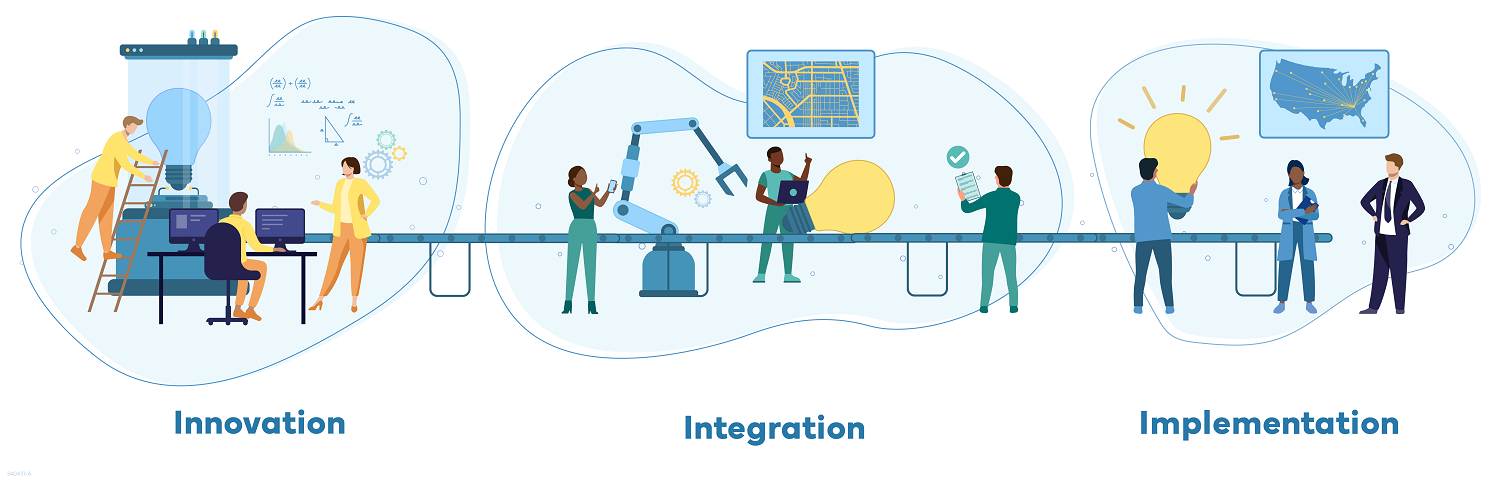
The U.S. Centers for Disease Control and Prevention (CDC) recently announced it had granted 13 funding awards to establish a first-of-its-kind national network, the Outbreak Analytics and Disease Modeling Network (OADMN).
The OADMN's goal is to improve speed, accuracy, and use of data & analytics during health emergencies, which is an important step towards ensuring Americans have the information they need to keep themselves and their families safe during outbreaks.
Many of these awardees are leading a consortium of collaborators to design, prototype, test, and scale up advances in data modeling tools and technology that can be used to support public health decision-makers at all levels of government.
"The collaboration with our public health, private, and academic partners over the last year to advance the science of disease forecasting and deliver decision support to leaders has been instrumental in improving outbreak response," said Dr. Dylan George, Director, Center for Forecasting and Outbreak Analytics, in a press release on September 22, 2023.
The grantees will be organized into three categories of performance, supporting three priority program actions:
- Innovators: these grantees will develop methods, tools, technologies, and other products to support emergency response.
- Integrators: these grantees will take lessons learned and techniques from Innovators and test them in small-scale deployments.
- Implementors: these grantees will take tested best practices and scale up to larger jurisdictions and partners.
The funding 13 recipients comprising OADMN in September 2023 include:
Emory University
Northeastern University
The University of North Carolina at Chapel Hill
Kaiser Permanente Southern California
Carnegie Mellon University
University of Michigan School of Public Health
University of California, San Diego
University of Minnesota
Clemson University
University of Utah
International Responder Systems
The University of Texas at Austin
The Johns Hopkins University
Note: The OADMN effort is in addition to the U.S. government's Disease X initiative.
During last week's Advisory Committee on Immunization Practices (ACIP) meeting, an updated adult, adolescent, and infant vaccination schedule was presented.
On September 22, 2023, the CDC's Sarah Schillie, MD, MPH, MBA CAPT, US Public Health Service, presented a draft Addendum to the 2023 Immunization Schedules.
This Addendum contains ACIP recommendations that occurred after the 2023 schedule was published earlier this year.
For 2023, two separate vaccination schedules with multiple sections summarize and aid in implementing the approved ACIP policy.
This is the most up-to-date vaccine recommendation for COVID-19, RSV, Influenza, pneumococcal, polio, and Mpox vaccines.
The CDC plans to release 2023 Immunization Schedules with Addenda this week. With this addition, all ACIP recommendations will formally be part of the Immunization Schedules.

BioArctic AB and Eisai announced today that LEQEMBI® Intravenous Infusion had been approved in Japan as a treatment for slowing the progression of mild cognitive impairment and mild dementia due to Alzheimer's disease (AD).
LEQEMBI is a humanized immunoglobulin gamma 1 monoclonal antibody (mAbs) directed against aggregated soluble and insoluble forms of Aβ.
LEQEMBI is the first and only approved treatment shown to reduce the rate of disease progression and slow cognitive and functional decline by selectively binding to and eliminating the most toxic Aβ aggregates that contribute to neurotoxicity in AD.
Japan is the second country to approve LEQEMBI, following the Food and Drug Administration (FDA) approval in the U.S. in July 2023.
"The approval of LEQEMBI in Japan is another important step in the fight against Alzheimer's disease," said Gunilla Osswald, CEO of BioArctic, in a press release on September 25, 2023.
Eisai serves as the lead of LEQEMBI development and regulatory submissions globally, with both Eisai and Biogen co-commercializing and co-promoting the product and Eisai having final decision-making authority.
BioArctic has the right to commercialize lecanemab in the Nordic region, and currently, Eisai and BioArctic are preparing for joint commercialization in the region.
According to the Alzheimer's Association, AD is a degenerative brain disease caused by complex brain changes following cell damage, leading to dementia.
As of September 2023, The U.S. FDA, the European Medicines Agency, and the United Kingdom's NHS have not authorized preventive AD vaccines.

A specialty vaccine company today announced the signing of a $32 million contract with the United States (U.S.) Department of Defense (DoD) for the immediate supply of its Japanese encephalitis (JE) vaccine, IXIARO®.
Under this new one-year contract with Valneva SE, the DoD can purchase additional doses for the next twelve months.
IXIARO® is the only JE vaccine approved for adults by the U.S. Food and Drug Administration (FDA).
Dipal Patel, Chief Commercial Officer of Valneva, commented in a press release on September 25, 2023, "We are excited to continue our long-term relationship with the DoD."
"The U.S. military has trusted IXIARO® for over ten years to help protect military personnel, their families, civilian government service personnel, and government contractors from this potentially deadly disease."
Japanese encephalitis is a deadly infectious disease found mainly in Asia. The disease is endemic in Southeast Asia, India, and China, a region with more than three billion population.
About 70,000 cases of JE are estimated to occur in Asia each year.
JE is fatal in approximately 30% of those who show symptoms, leaving half of the survivors with permanent brain damage.
In the U.S., Valneva markets and distributes IXIARO® directly to the military and private travel market.
IXIARO / JESPECT® is licensed for adults in Australia, New Zealand, Europe, Canada, Switzerland, Hong Kong, Singapore, Israel, Norway, Liechtenstein, Iceland, Singapore, Japan, the United Kingdom, and the Republic of Korea. In all other licensed territories, IXIARO/JESPECT is indicated for use by adults aged 18 years or older.
When comparing data from the past few influenza seasons, it appears the 2023-2024 flu season is starting slowly.
Globally, the World Health Organization's Influenza Update N° 454 confirmed influenza detections remained low. In the temperate zones of the northern hemisphere, indicators of influenza activity were reported at low levels or below the seasonal threshold in most reporting countries.
Within the United States, the Centers for Disease Control and Prevention (CDC) reported on September 22, 2023, that seasonal flu rates last week were low nationally, with 315 (0.8%) positive specimens reported.
Last week, 820 patients with laboratory-confirmed influenza were admitted to a hospital, an increased number since the previous report.
Separately, the U.S. National Center for Health Statistics mortality surveillance data published on September 21, 2023, confirmed 13 additional influenza-related deaths.
The FluView week #37 report confirmed two additional children died from flu-related illness. A total of 176 influenza-associated pediatric fatalities were reported during the 2022-2023 flu season.
The previous high was during the 2019-2022 flu season when 199 children died from influenza infections.
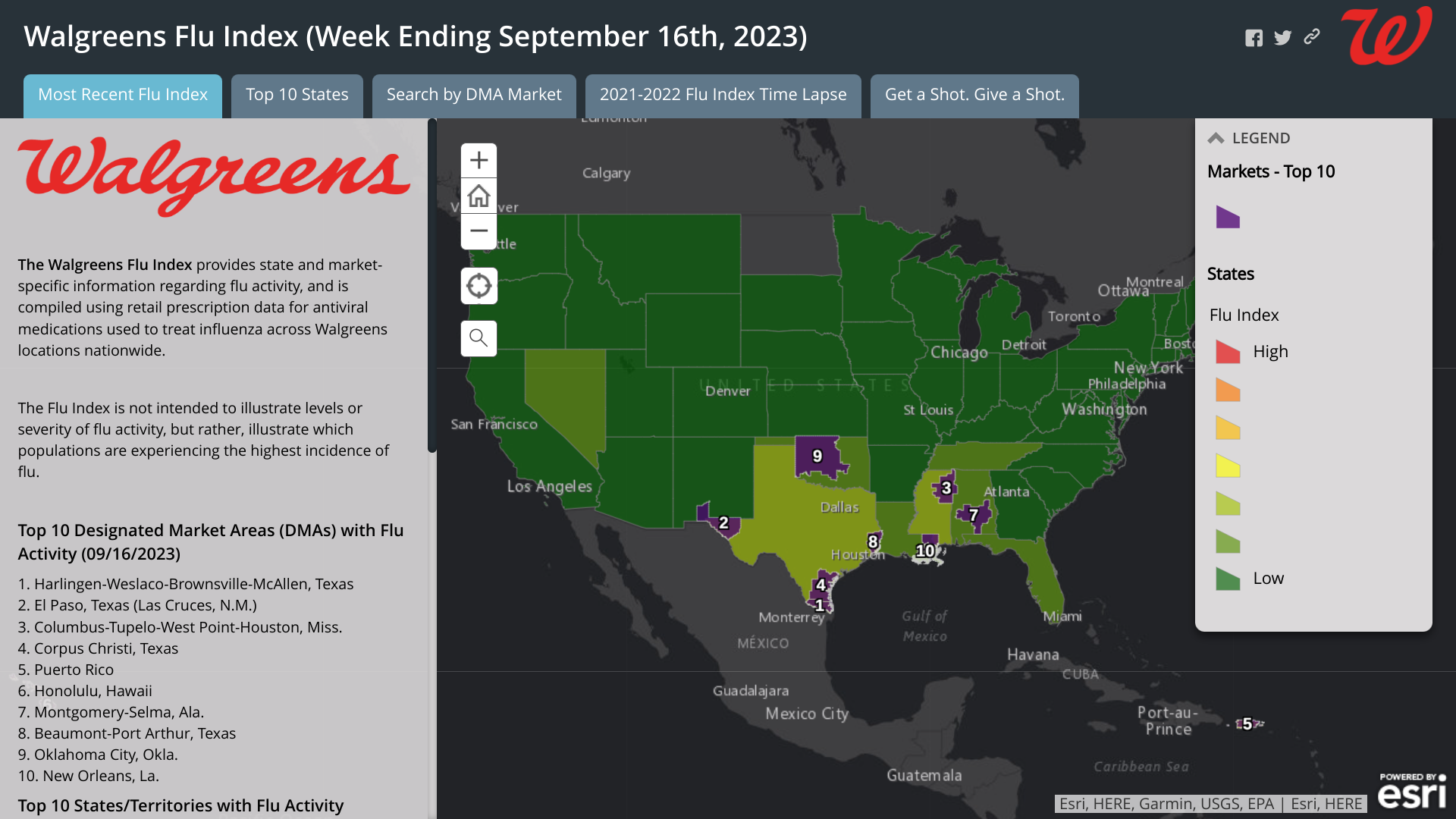
From a local perspective, the Walgreens Flu Index market-specific information indicates the top cities for influenza were led by the following:
1. Harlingen-Weslaco-Brownsville-McAllen, Texas
2. El Paso, Texas (Las Cruces, N.M.)
3. Columbus-Tupelo-West Point-Houston, Miss.
4. Corpus Christi, Texas
5. Puerto Rico
The CDC's new Director, Mandy K. Cohen, MD, MPH, recently posted a video on Instagram encouraging people to speak with a healthcare provider about flu shot options and when to be protected during the new flu season.
As of late September 2023, most pharmacies offer influenza vaccination appointments and walk-in services.
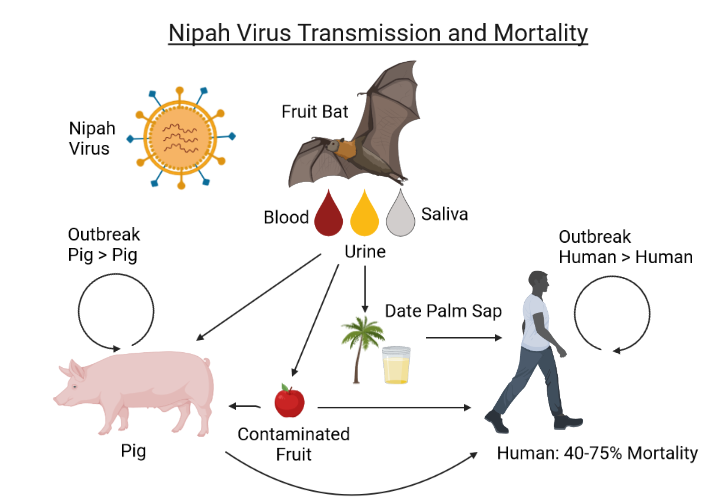
Since August 2023, the Indian state of Kerala has been experiencing its fourth Nipah virus outbreak.
Over the past two decades, the Nipah virus has spilled into humans in Bangladesh, India, Malaysia, the Philippines, and Singapore.
While no approved Nipah virus vaccines exist, several candidates are conducting clinical trials.
“The Nipah virus outbreak is a reminder of the increased risk of zoonotic diseases as the world population expands and encroaches into wild habitats. We have the technology to rapidly develop next-generation mRNA vaccines that will effectively fight against these new viruses”, commented Pascal Brandys, co-founder and CEO of Phylex BioSciences, in a press release on September 22, 2023.
“Our mRNA vaccine encoding for a highly immunogenic nanoparticle provides a flexible platform for rapid development of a new Nipah virus vaccine and will be of great value in combatting Nipah virus and other similar viruses.”
Phylex BioSciences published last year the proof-of-principle of its second-generation nanoparticle mRNA vaccine. The same technology is directly applicable to the development of a Nipah virus vaccine.
The second generation mRNA vaccine candidate encodes for a nanoparticle displaying 60 copies of the antigen-based upon the head domain of the G protein of the Nipah virus.
The anticipated advantages of the nanoparticle vaccine are superior immune response, long-term protection from cell-mediated immunity, and protection against virus dissemination in the brain.
Globally, an estimated two billion people worldwide are at risk for bat-borne Nipah virus outbreaks. The Nipah virus has a fatality rate exceeding 40%.