Search API
While there is currently no cure for cat allergy, a prevalent, life-long condition, an innovative therapeutic vaccine for humans has begun clinical trials.
Angany Inc. today announced that it has received clearance from U.K.'s Medicines and Healthcare Products Regulatory Agency for the first clinical study to be conducted on its vaccine candidate ANG-101 to treat human allergy to cats.
ANG-101 is a therapeutic vaccine that provides a disruptive disease-modifying approach for treating cat allergy.
Derived from its proprietary eBioparticle-Potentiated Immunotherapy™ technology, ANG-101 active immunotherapeutic ingredient is a unique 140 nm enveloped bioparticle (eBioparticle™) that mimics a virus in shape and size with its surface covered with thousands of copies of cat major allergen Fel d 1.
This clinical study is a first-in-human, open-label, and single-site evaluation of the new vaccine's safety, allergenicity, and immunogenicity in adult patients allergic to cat dander.
This early-stage clinical trial will be conducted under the guidance of Professor Stephen Durham and Dr Guy Scadding, two leading clinical allergy experts from Imperial College London.
Professor Durham commented in a press release on October 5, 2023, "The potential treatment of cat allergy using an auto-adjuvanted vector builds upon its known ability to induce strong allergen-specific IgG antibody responses, as observed in animal models."
Unlike prophylactic vaccines, Angany’s therapeutic vaccines are a new generation of immunotherapy biologics used to treat established pathologies.
They are meant to restore or boost natural immune mechanisms and create sustainable immune protection and vigilance.
Human allergies to cats and dogs affect 10 to 20% of the world’s population, says the Asthma and Allergy Foundation of America.
When you have a pet allergy, you are not allergic to the pet’s hair, fur, or feathers but to the protein found in the pet’s dander, saliva, and urine.

Two world-class pharmaceutical companies announced an agreement for a potential first-in-class vaccine against extraintestinal pathogenic E. coli.
On October 3, 2023, France-based Sanofi confirmed it has agreed with Janssen Pharmaceuticals, Inc., a Johnson & Johnson company, to develop and commercialize a 9-valent vaccine candidate for extraintestinal pathogenic E. coli currently in Phase 3 study.
The agreement combines Janssen's robust science behind this potential first-in-class product, Sanofi's worldwide manufacturing footprint, and the recognized world-class expertise of these companies in launching innovative vaccines.
This is important news since Extraintestinal pathogenic E. coli is a leading cause of sepsis, particularly in older adults, with an approved vaccine available in 2023.
Sepsis is a life-threatening bloodstream infection accompanied by severe illness and widespread organ damage generated by the body's self-destructive response to the infection.
Thomas Triomphe, Executive Vice President, Vaccines, at Sanofi, commented in a press release, "E. coli is a significant cause of sepsis, mortality, and antimicrobial resistance in older adults, and the number of cases is rising as the population ages."
"In line with our commitment to design and deliver first- or best-in-class medicines and vaccines, this agreement with Janssen aims to positively impact public health by reducing hospitalization costs and the burden on health systems associated with ExPEC and help older adults around the world to live longer healthier lives."
The ongoing Phase 3 E.mbrace clinical trial is designed to evaluate the efficacy of the 9-valent extraintestinal pathogenic E. coli vaccine (ExPEC9V) compared to placebo in preventing invasive E. coli disease caused by ExPEC9V O-serotypes.
The study was started in 2021 by Janssen and continues to enroll patients. To learn more, visit https://classic.clinicaltrials.gov/ct2/show/NCT04899336.
Under the terms of the new agreement, both companies will co-fund current and future research and development costs.
Sanofi will pay USD 175M upfront to Janssen, followed by development and commercial milestones. A profit-share arrangement will exist in the U.S., EU4 (France, Germany, Italy, Spain), and the U.K. In the rest of the world, Janssen will receive tiered royalties and sales milestones.

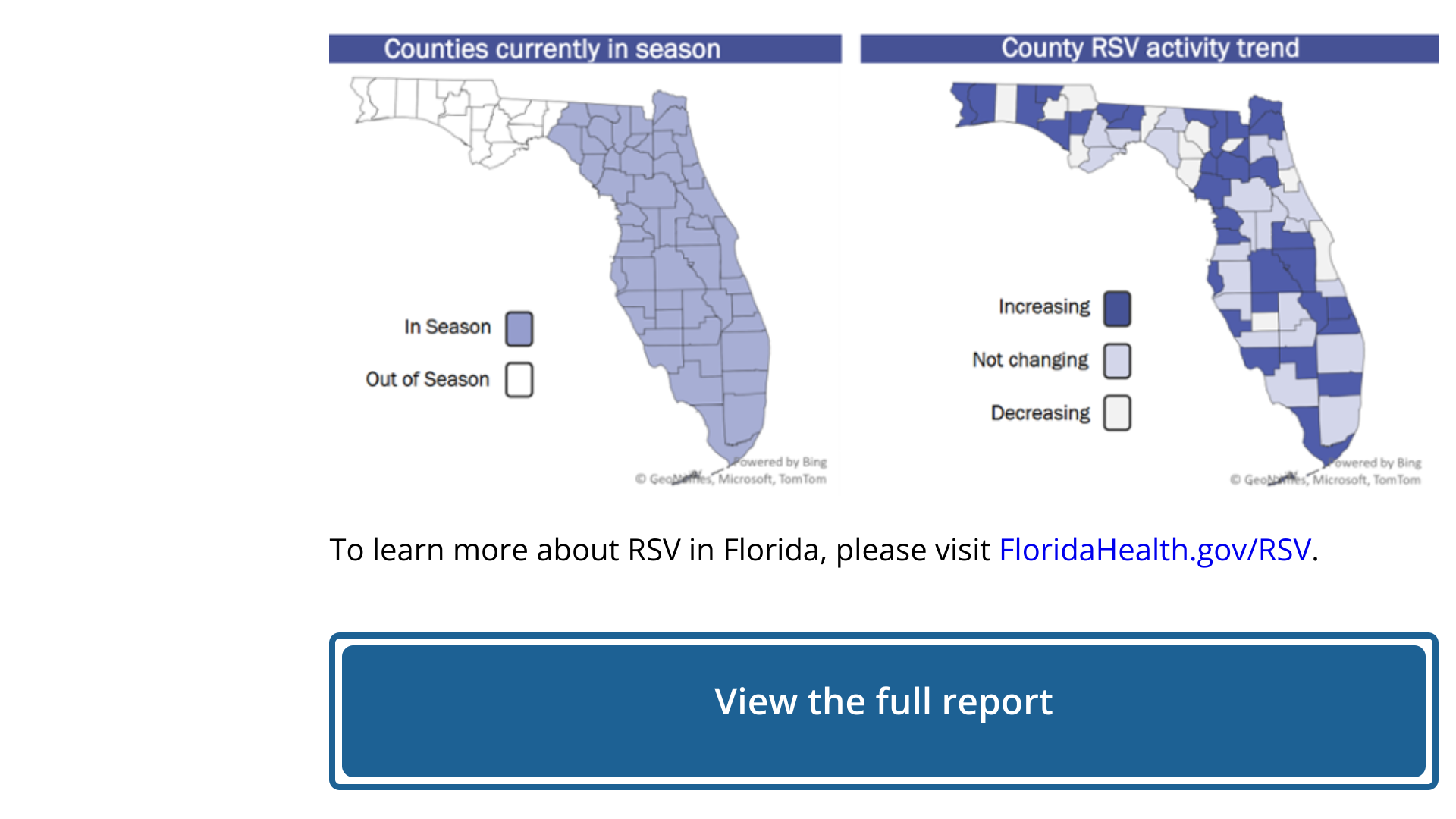
Across both prepandemic and pandemic years, the respiratory syncytial virus (RSV) outbreaks in the United States began in Florida, the southeast, and later in the north and west regions.
Florida's RSV season is longer than the rest of the U.S. and has distinct regional patterns, says the U.S. Centers for Disease Control and Prevention (CDC).
The Florida Department of Health reported as of week #39, September 30, 2023, RSV activity was increasing with higher test positivity, hospital admissions, and emergency room rates, with three previous outbreaks but no current RSV outbreak.
Furthermore, NREVSS data show Florida's PCR positivity 3-week moving average was about 7.7% as of September 28, 2023.
Globally, the World Health Organization (WHO) Influenza Update N° 454 indicates RSV activity was found to be generally low, except in some parts of Western Australia and some Central and temperate South American countries.
Additional WHO and CDC RSV outbreak data are posted at Precision Vaccinations.
As of October 4, 2023, RSV vaccines and monoclonal antibody therapies are available in the U.S.
For the first time, the U.S. government has authorized an updated protein-based COVID-19 vaccine.
Today, the U.S. Food and Drug Administration amended the emergency use authorization (EUA) of the Novavax COVID-19 Vaccine, Adjuvanted for use in individuals 12 years of age and older, to include the 2023-2024 formula.
Novavax Inc.'s updated vaccine addresses currently circulating variants to provide better protection against the serious consequences of COVID-19.
Consistent with the totality of the evidence and input from the FDA's expert advisors, the Novavax COVID-19 Vaccine, Adjuvanted, a monovalent vaccine, now includes the spike protein from the SARS-CoV-2 omicron coronavirus variant lineage XBB.1.5 (2023-2024 formula).
The FDA confirmed it has determined that the Novavax COVID-19 Vaccine, Adjuvanted (2023-2024 Formula), has met the statutory criteria for issuance of an EUA and that the known and potential benefits of the vaccine outweigh its known and potential risks in individuals 12 years of age and older.
This means individuals 12 years of age and older previously vaccinated with a COVID-19 vaccine (and who have not already been vaccinated with a recently updated mRNA COVID-19 vaccine) are eligible to receive one dose, and unvaccinated individuals receive two doses.
Peter Marks, M.D., Ph.D., director of the FDA's Center for Biologics Evaluation and Research, commented in a press release on October 3, 2023, "Today's authorization provides an additional COVID-19 vaccine option that meets the FDA's standards for safety, effectiveness and manufacturing quality needed to support EUA."
From an availability perspective, Novavax previously indicated it had produced about 20 million doses of the updated vaccine and stored them in the U.S., enabling expedited delivery to health clinics and pharmacies.
As part of today's action, the Novavax COVID-19 Vaccine, Adjuvanted (Original monovalent), is no longer authorized for use in the U.S. Additionally, global information about the Novavax COVID-19 Vaccine varies by country and is searchable at NovavaxMedInfo.com.

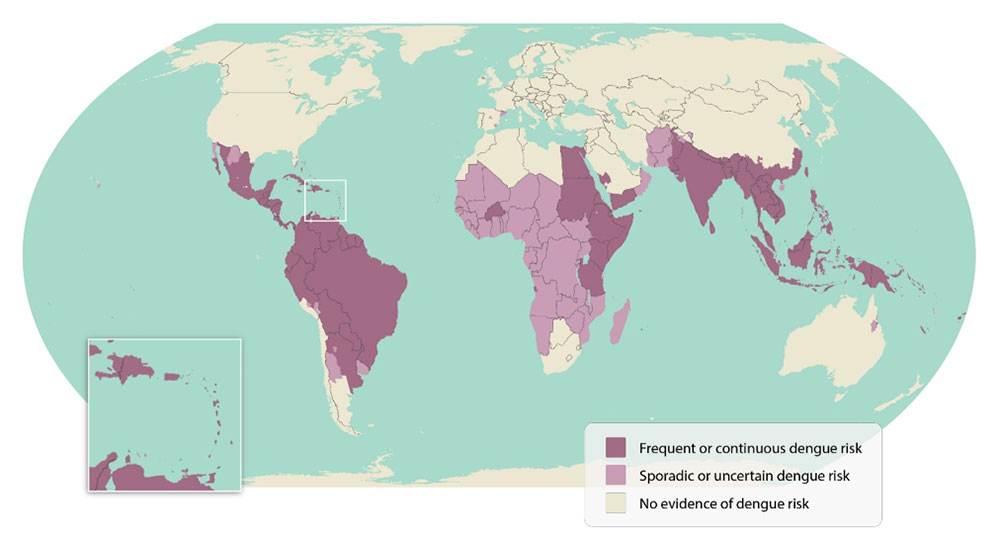
With the World Health Organization's (WHO) recent recommendation of a second dengue vaccine, this increased access may help reduce dengue outbreaks globally. About 4 billion people live in areas with a risk of dengue.
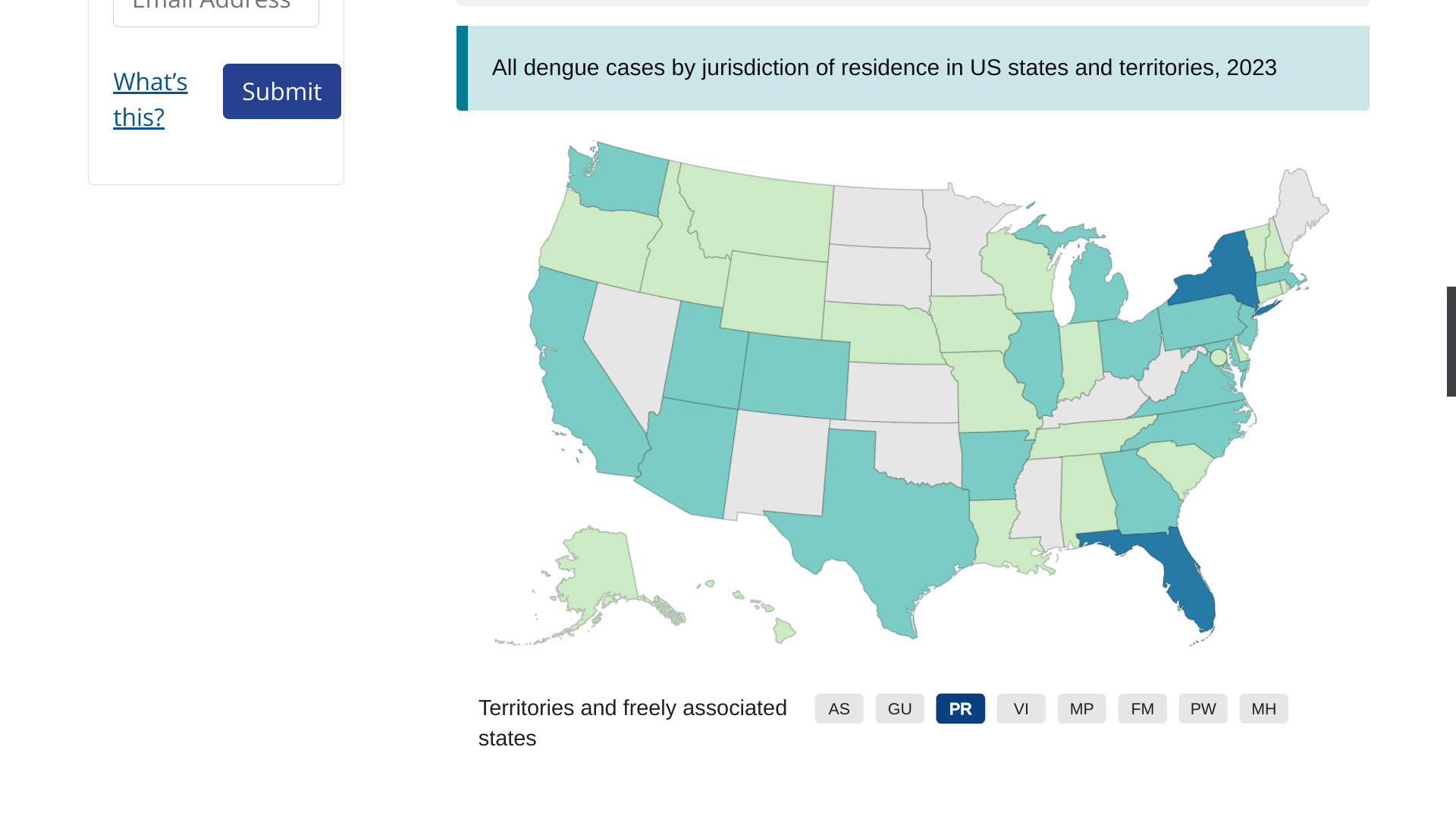
In the United States, the CDC confirmed that 44 U.S. jurisdictions had reported about 997 dengue cases as of September 27, 2023.
During the summer of 2023, dengue outbreaks have been reported by the U.S. CDC in Florida, New Jersey, New York, and Puerto Rica.
In Florida, the Department of Health issued a statewide mosquito-borne illness advisory in June 2023.
As of September 30, 2023, 38 locally acquired dengue cases have been reported this year, primarily in Miami-Dade County. Additionally, 334 travel-associated dengue fever cases were reported, primarily in people arriving from Cuba and Brazil.
Over one thousand miles to the north, New York has reported 74 dengue cases, and New Jersey has confirmed 35 this year.
In the Caribbean Sea, the U.S. territory of Puerto Rica has reported 442 cases.
Anyone who lives in or travels to dengue-endemic areas is at risk for infection. According to the CDC, the four dengue viruses are spread to people through the bite of an infected Aedes species mosquito.
The CDC has issued various Travel Health Advisories regarding dengue outbreaks in 2023. Before visiting the areas, the CDC suggests discussing dengue vaccination options with a healthcare provider.
In a new study published by BMC Public Health, researchers investigated the effect of zoster vaccination on dementia risk in an extensive UK population-based data set.
On October 2, 2023, these researchers disclosed an inverse association between zoster vaccination and dementia outcome in a fully adjusted model (HR 0.78, 95% CI: 0.77–0.79).
For Alzheimer's disease, the effect size is smaller (HR 0.91, 95% CI: 0.89–0.92); however, this result is likely only significant because of the large sample size involved.
To investigate if the result seen was exclusive to HZ vaccination, we also explored the effect of influenza vaccine and dementia/Alzheimer's disease.
They found a slight decreased hazard risk with HR of 0.96 (95% CI: 0.94–0.97) for dementia and HR of 1.10 (95% CI: 1.07–1.12) for Alzheimer's disease.
It has been estimated that the number of people with dementia could increase from 57 million cases in 2019 to 152 (130.8–175.9) million cases worldwide in 2050.
These researchers wrote, 'Several population-based studies have suggested a link between herpes zoster infection or vaccine against shingles and dementia. However, other population-based studies did not show any association between zoster infection and dementia risk.'
Globally, there are several approved shingles (herpes zoster) vaccines.
In the United States, GlaxoSmithKline plc Shingrix® is generally available at health clinics and pharmacies and has been reported up to 90% effective against shingles infection in various clinical trials.

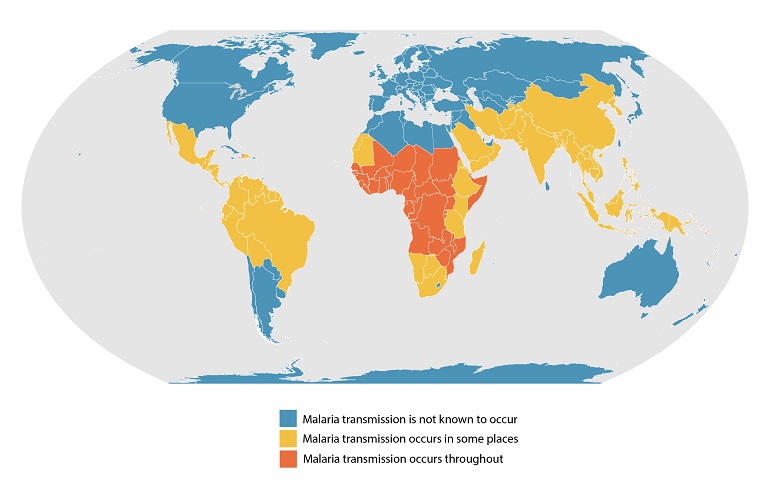
Malaria vaccines have been in development since the 1960s, with substantial progress in the last decade, says the World Health Organization (WHO). Today, the WHO announced it has recommended a second vaccine, R21/Matrix-M, for preventing malaria in children.
As of October 2023, the R21 vaccine is the second malaria vaccine recommended by WHO, following the Mosquirix™ (RTS,S/AS01) vaccine, which received a WHO recommendation in 2021.
Demand for malaria vaccines is unprecedented in 2023. However, the available supply of RTS,S is limited.
The addition of R21 to the WHO's recommended malaria vaccine list is expected to result in sufficient vaccine supply in various countries.
The following steps for R21/Matrix-M, include completing the ongoing WHO prequalification and enabling international vaccine procurement for a broader rollout in malaria outbreak countries.
The WHO's Director-General endorsed the recommendation following its regular biannual meeting on 25-29 September 2023.
"As a malaria researcher, I used to dream of the day we would have a safe and effective vaccine against malaria. Now we have two," commented Dr Tedros Adhanom Ghebreyesus, WHO Director-General, in a press release on October 2, 2023.
"Demand for the RTS,S vaccine far exceeds supply, so this second vaccine is a vital additional tool to protect more children faster and to bring us closer to our vision of a malaria-free future."
The R21 vaccine has been shown to reduce symptomatic malaria cases by 75% during the 12 months following a 3-dose series. A fourth dose given a year after the third maintained efficacy.
This efficacy is similar to that demonstrated when RTS,S is given seasonally.
The WHO says that at US $2 – US $4 per dose, the cost-effectiveness of the R21 vaccine would be comparable with other recommended malaria interventions and other childhood vaccines.
Malaria is a disease caused by four species of protozoan parasites of the genus Plasmodium and is transmitted to people by Anopheles mosquitoes.
According to the WHO's recent World Malaria Report, the global number of malaria cases reached about 240 million, with over 600,000 related fatalities.
As of October 2023, neither malaria vaccine is available in the United States.
Dengue outbreaks continue to pose significant public health burdens in endemic countries and, unfortunately, may continue to increase in incidence as disease-carrying mosquitoes expand geographically, according to the World Health Organization (WHO).
To reduce the severity of dengue outbreaks, the WHO today announced the live-attenuated quadrivalent dengue vaccine Qdenga® (TAK-003) developed by Takeda has been confirmed to demonstrate efficacy against all four serotypes of the dengue virus in baseline seropositive children (4-16 years) in endemic countries.
And against serotypes 1 and 2 in baseline seronegative children.
The WHO's Strategic Advisory Group of Experts on Immunization (SAGE) on Immunization recommended on October 2, 2023, that Qdenga be considered for introduction in settings with high dengue disease burden and high transmission intensity to maximize the public health impact and minimize any potential risk in seronegative persons.
The SAGE now recommends introducing Qdenga to children aged 6 to 16.
The vaccine should be introduced within this age range about 1-2 years before the age-specific peak incidence of dengue-related hospitalizations.
Qdenga should be administered in a 2-dose schedule with a 3-month interval between doses.
The WHO will consider the SAGE recommendation and update its paper on dengue vaccines to include final guidance on using Qdenga in public vaccination programs.
"The global impact of dengue cannot be overlooked as the incidence continues to rise. This week, the WHO's SAGE provided important recommendations for the use of QDENGA in preventing dengue," commented Gary Dubin, M.D., president of the Global Vaccine Business Unit at Takeda, in a press release on October 3, 2023.
While approved for use in Brazil and various European countries, Qdenga is unavailable in the U.S.
On July 11, 2023, Takeda announced that the Company has voluntarily withdrawn Qdenga's U.S. Biologics License Application following discussions with the U.S. Food and Drug Administration on aspects of data collection.
However, the Dengvaxia® vaccine is both FDA and WHO-recommended where appropriate.
As of September 13, 2023, 44 U.S. jurisdictions had reported about 997 dengue cases this year. Throughout the summer of 2023, dengue outbreaks have been reported in southern Florida and Puerto Rica.
Note: This article was updated on October 3, 2023, to include the Company's press release.
Without an approved Alzheimer's disease vaccine available, several bipolar disorder (BD) candidates are following U.S. Food and Drug Administration (FDA) policies to accelerate clinical research.
In the U.S., about 2.8% of the population is estimated to be affected by BD during the past year, says the U.S. NIH.
For example, Alzamend Neuro, Inc. today announced receipt of a “Study May Proceed” letter from the U.S. FDA for the initiation of study AL001-BD01, a Phase IIA clinical study of AL001 for BD type 1.
AL001 is a novel lithium-delivery system that has the potential to deliver benefits of marketed lithium salts while mitigating or avoiding currently experienced toxicities associated with lithium, a chemical entity well known for efficacy in BD type 1.
Alzamend’s goal is to revive the utility of lithium treatment by importantly improving the benefit-to-risk relationship of lithium treatment in clinical practice.
“Lithium was the first mood stabilizer approved by the FDA and is still a first-line treatment option for BD type 1,” said Stephan Jackman, Chief Executive Officer of Alzamend, in a press release on Octobe 2, 2023.
“If we are able to develop a next-generation lithium product (AL001) that would not routinely require therapeutic drug monitoring, it would constitute a major improvement over current lithium-based treatments and positively impact the seven million Americans afflicted with BD."
"We are advancing the process and expect that the first patient will be dosed in the first quarter of 2024"
Based on the favorable safety profile observed in the recently completed study and extensive safety data on the drug’s constituent components, the AL001 development program may qualify for a Section 505(b)(2) New Drug Application pathway for FDA approval, which is available to new formulations of an approved drug.
BD is a mood disorder characterized by periods of depression and periods of abnormally elevated happiness.
The condition is classified as BD Type 1 if there has been at least one manic episode, with or without depressive episodes.
And can be classified as BD Type 2 if there has been at least one hypomanic episode (but no full manic episodes) and one major depressive episode.
As of October 2023, there are several Alzheimer's disease vaccines and therapies conducting clinical trials which are seeking participants.