Search API
Pertussis vaccination during pregnancy has been implemented in many high-resource countries, and recent data from the United States, the United Kingdom, and South America demonstrate its effectiveness in reducing infant pertussis in the first two months of life.
What has not been well established is whether 'blunting' (maternal immunological interference) has clinical consequences.
Blunting of the infant’s subsequent response to primary immunization by maternally derived antibodies has been demonstrated for many antigens.
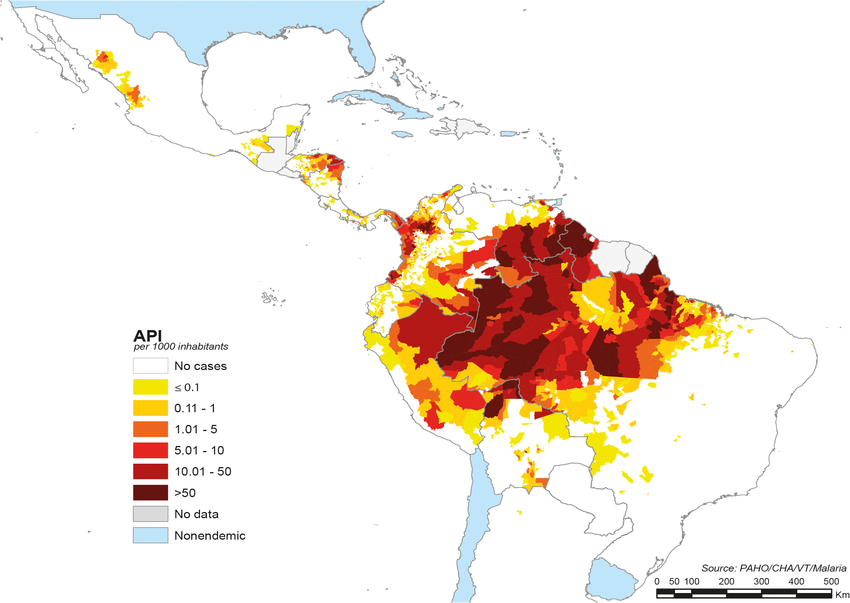
A new study published in the journal Pediatrics disclosed pertussis vaccination near 28 weeks' gestation was associated with a lower risk of infection among infants through 8 months of age.
These Australian researchers reviewed records from 2013 through 2017 to calculate the impact after infants received diphtheria, tetanus, and pertussis DTaP doses.
The vaccine effectiveness (VE) from maternal vaccination in infants younger than two months was 70.4% (95% confidence interval 50.5% to 82.3%).
Published on October 9, 2023, these researchers observed slightly lower VE point estimates for the third dose of infant pertussis vaccine among maternally vaccinated compared with unvaccinated infants (76.5% vs. 92.9%, P = .002) and did not observe higher rates of pertussis infection (hazard ratio, 0.70; 95% CI, 0.61–3.39).
The vaccine was given usually between 28 to 31 weeks gestation.
A commentary by Kathryn Edwards, MD, professor of pediatrics at Vanderbilt University Medical Center, stated, 'The consequences of maternally derived antibody on infant responses will need to continue to be monitored, as was done in the carefully conducted study of pertussis reported in this issue of Pediatrics. It will be critical to assess the burden of vaccine-preventable diseases and affirm that blunting from maternal immunization has no material impact on disease control.'
World Polio Day is an opportunity to highlight efforts toward a polio-free world and honor the tireless contributions of those on the frontlines in the fight to eradicate polio from every corner of the globe.
The Global Polio Eradication Initiative (GPEI) confirmed World Polio Day is on October 24, 2023, and has launched a Make Polio History campaign to rally existing and new supporters of polio eradication..... through vaccination programs.
Over the past few years, the GPEI has led the deployment of the type 2 novel oral polio vaccine (nOPV2), which is genetically more stable than existing oral polio vaccines, with a lower risk of reversion to neurovirulence.
Approximately 750 million nOPV2 doses have been administered in more than 35 countries worldwide.
Unfortunately, polio outbreaks have been confirmed in various countries in 2023, led by Afghanistan and Pakistan.
The World Health Organization confirmed on August 25, 2023, that the spread of poliovirus remained a Public Health Emergency of International Concern and extended the emergency notice for an additional three months.
"It's Time to Make Polio History,' says U.S. CDC Director Mandy K. Cohen, MD, MPH, in a related video message posted on YouTube on October 4, 2023. "It won't be easy, but making history never is. We have the tools and knowledge to ensure no child is paralyzed by polio again."
In the United States, the IPV polio vaccine is offered at most health clinics and pharmacies.

LimmaTech Biologics AG announced today the closing of a USD 37 million Series A financing round that will empower its proprietary technology platform and accelerate its preclinical and clinical vaccine candidates against increasingly dangerous bacterial infections, including programs addressing shigellosis and gonorrhea.
Antimicrobial Resistance is responsible for approximately 5 million deaths annually. Infections that were once easily treatable have now become difficult, if not impossible, to cure.
As a leading example of this threat to global health, half of the approximately 700,000 annual gonorrhea infections in the U.S. are already resistant to antibiotics, and there is a real threat of gonorrhea soon becoming untreatable.
While there are no gonorrhea vaccines available, off-label vaccines and treatments are in use.
Later-stage clinical development efforts will focus on the company's Shigella vaccine program, which LimmaTech developed with GSK. The company expects to announce preliminary results from the Shigella program's ongoing Phase 2 clinical trial in the second half of 2023.
Shigella cause an estimated 450,000 infections in the U.S. each year.
According to the U.S. CDC, people can get a Shigella infection (shigellosis) after putting something in their mouth or swallowing something that has come into contact with the stool of someone with a Shigella infection.
"Within the next decade, multiple bacterial infections will become untreatable due to antimicrobial Resistance, which is already a significant burden on global health. By advancing our innovative technology platform, LimmaTech has the potential to simultaneously provide vaccine-induced protection against bacterial infections, mitigate the increasing risk of antibiotic resistance, and move toward the control of several highly transmissible pathogens," commented Dr. Franz-Werner Haas, CEO of LimmaTech, in a press release on October 9, 2023.
".....With this support and our team of proven experts in bacterial vaccine development and manufacturing, we look forward to providing life-changing vaccines to address a major global medical need."
The Company is conducting a Phase I/II clinical trial in the Republic of Kenya of a 4-valent candidate vaccine to help prevent diarrheal disease caused by the Shigella bacteria in children and infants in low and middle-income regions. The Shigella study is conducted in collaboration with GSK and the Wellcome Trust.
LimmaTech is committed to translating novel scientific concepts into highly effective vaccines that benefit humanity. For more information, please visit www.lmtbio.com.

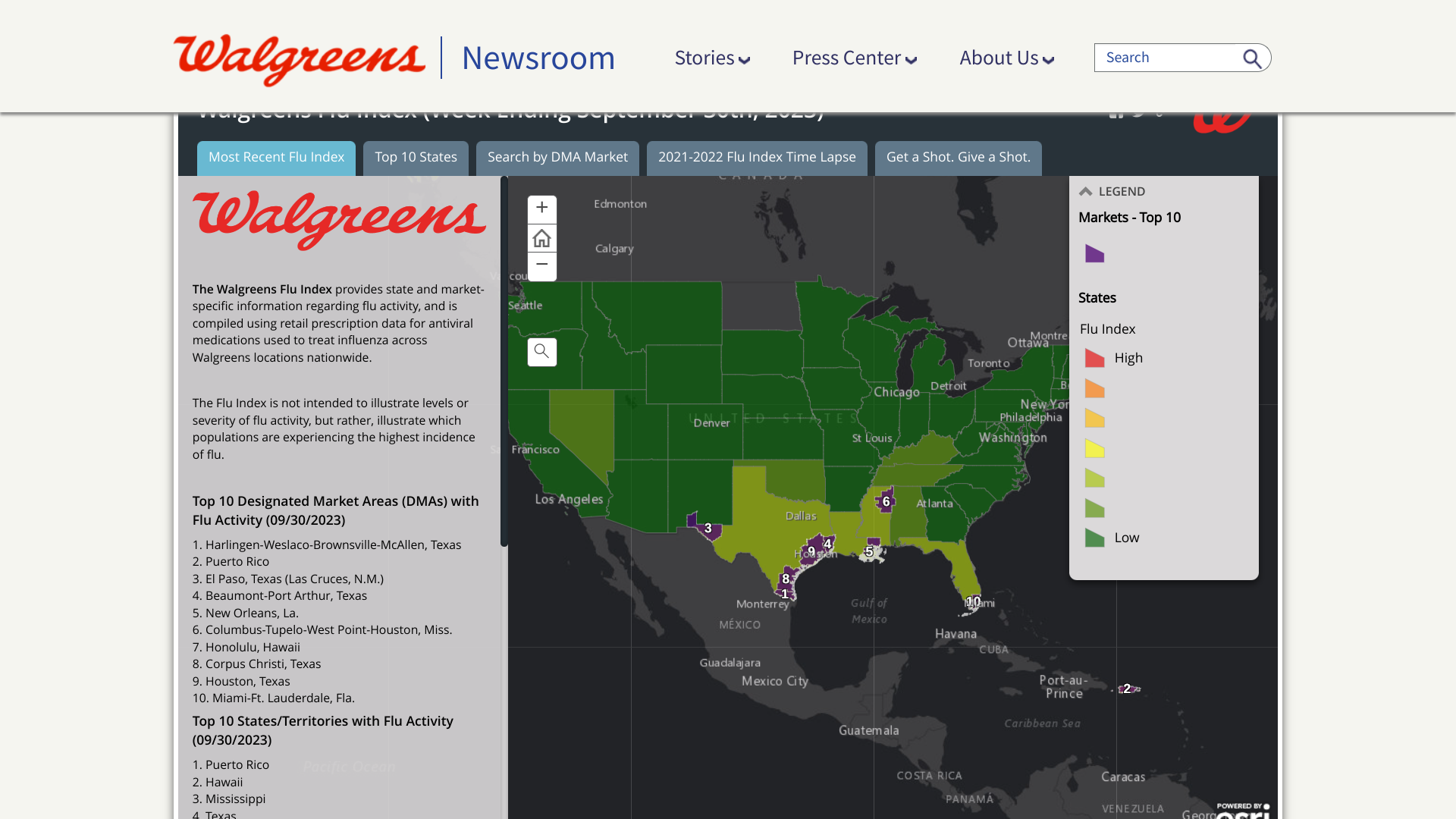
While the United States community is debating where and when the 2023-2024 flu season will arrive, the Walgreens Flu Index® ranks the top markets and states for flu activity, including Puerto Rico.
As of September 30, 2023, cities in the southern U.S. led the Top 10 Designated Market Areas with Flu Activity.
- Harlingen-Weslaco-Brownsville-McAllen, Texas
- Puerto Rico (San Juan)
- El Paso, Texas (Las Cruces, N.M.)
- Beaumont-Port Arthur, Texas
- New Orleans, La.
- Columbus-Tupelo-West Point-Houston, Miss.
- Honolulu, Hawaii
- Corpus Christi, Texas
- Houston, Texas
- Miami-Ft. Lauderdale, Fla.
From a more clinical perspective, the U.S. Centers for Disease Control and Prevention (CDC) today published its week #39 Influenza Surveillance Report.
As of October 6, 2023, seasonal flu rates last week were low nationally, with 444 (1%) positive specimens reported last week.
Additionally, 1,040 patients with laboratory-confirmed influenza were admitted to a hospital, an increased number since the previous CDC report.
And the U.S. National Center for Health Statistics mortality surveillance data distributed on October 5, 2023, confirmed that 11 additional influenza-related deaths were reported last week.
Of the flu-related deaths reported from October 2, 2022, to September 9, 2023, 9,697 (4%) listed influenza.
This data exceeded the average number of influenza-coded deaths (8,530) from 2015-16 through 2019-20.
Among 5,390 hospitalized adults with information on underlying medical conditions, 96.8% had at least one reported underlying medical condition; the most commonly reported were hypertension, cardiovascular disease, metabolic disorder, and obesity.
The CDC's new Director, Mandy K. Cohen, MD, MPH, recently answered questions and offered the most up-to-date information and common-sense solutions so you can protect yourself and your loved ones this respiratory season.
This week's updated flu shot availability news indicated that over 100 million influenza vaccines had been distributed to health clinics and pharmacies in the U.S.
Without preventative vaccines available for three well-known sexually transmitted diseases, the U.S. Centers for Disease Control and Prevention (CDC) recently took action by opening a docket to obtain comments on proposed guidelines for the use of doxycycline post-exposure prophylaxis (PEP) for prevention of bacterial sexually transmitted infections (STI).
Announced on October 2, 2023, the proposed guidelines for bacterial STI prevention include post-exposure prophylaxis with doxycycline (doxycycline PEP) because it has demonstrated benefit in reducing chlamydia, gonorrhea, and syphilis infections.
The incidence of STIs caused by Neisseria gonorrhoeae (causative agent of gonorrhea), Chlamydia trachomatis (causative agent of chlamydia), and Treponema pallidum (causative agent of syphilis) continues to increase in the U.S., says the CDC.
The proposed guidelines provide updated clinical guidance for healthcare providers to inform the use of doxycycline PEP for preventing bacterial STI infections.
Written comments must be received on or before November 16, 2023.
Doxycycline PEP, when offered, should be implemented in the context of a comprehensive sexual health approach, including risk reduction counseling, STI screening and treatment, recommended vaccination, and linkage to HIV pre-exposure prophylaxis (PrEP), HIV care, or other services, as appropriate.
The CDC has made available a pre-recorded informational presentation to provide information about the studies considered when developing the proposed guideline, explain the public comment process, and provide an overview of important monitoring for antibiotic use and antibiotic resistance that the agency will consider to address potential risks.
You may submit comments identified by Docket No. CDC–2023–0080 at Federal eRulemaking Portal: http://www.regulations.gov. Follow the instructions for submitting comments. Do not submit comments by email, as the CDC does not accept comments by email.

An independent committee of vaccine experts unanimously endorsed removing one influenza virus strain from future influenza vaccines.
On October 5, 2023, the Vaccines and Related Biological Products Advisory Committee (VRBPAC) voted to eliminate the influenza B/Yamagata lineage viruses since it has not been detected for about three years.
The VRBPAC vote follows the World Health Organization's recent announcement that it endorsed eliminating the influenza B/Yamagata/16/88 lineage component from the 2024 Southern Hemisphere flu shot formulation.
Flu seasons in the Southern Hemisphere usually occur between April and September, while the Northern Hemisphere's is generally between October and May.
This change should not impact the effectiveness of flu shots.
A recent U.S. CDC report found the adjusted vaccine effectiveness against severe acute respiratory infection hospitalization associated with any influenza virus during the 2023 Southern Hemisphere flu season was 51.9% (95% Confidence Interval 39.2%–62.0%).
For the 2023-2024 flu season in the U.S., there should be ample supply of vaccines at health clinics and pharmacies. According to a report by the Global Healthy Living Foundation and IQVIA, about 60% of vaccinations during the 2022 flu season took place at pharmacies in the U.S.
Vaccine manufacturers have projected to supply as many as 156.2 million to 170 million doses of influenza vaccines for the 2023-2024 season.
As of September 23, 2023, the CDC says about 100 million doses had been distributed in the U.S. All influenza vaccines are quadrivalent for this flu season, such as FLUMIST® and FLUCELVAX®.

The Arkansas Department of Health (ADH) today announced it identified a case of locally acquired malaria in a resident who resides in Saline County and has no international travel history.
ADH stated in a press release on October 4, 2023, that this is the only known locally acquired case of malaria in Arkansas and the 11th in the United States in 2023
Earlier in 2023, the U.S. Centers for Disease Control and Prevention (CDC) confirmed seven locally acquired malaria infections in Florida, one in Texas, and one in Maryland.
On September 7, 2023, the Pan American Health Organization estimated that approximately 41 million people are living in areas where the risk of infection by malaria is considered moderate to high in 21 Latin American countries.
The CDC has issued various outbreak alerts for malaria-endemic countries, including Costa Rica.
According to the CDC, malaria is a vaccine-preventable disease caused by four species of protozoan parasites of the genus Plasmodium. These disease-carrying mosquitoes are found throughout the Americas.
There are two malaria vaccines in use in Africa in 2023. However, they are not available in the U.S.