Search API
Merck and Moderna, Inc. today announced the initiation of a pivotal Phase 3 randomized clinical trial (INTerpath-002) evaluating V940 (mRNA-4157), an investigational individualized neoantigen therapy, in combination with KEYTRUDA®, Merck’s anti-PD-1 therapy, as adjuvant treatment in patients with completely resected (R0) Stage II, IIIA or IIIB (with nodal involvement) non-small cell lung cancer.
Global recruitment of the INTerpath-002 has begun, and the first patients enrolled in Australia.
“As lung cancer is the leading cause of cancer death worldwide, there is a need for continued scientific advancements to help fight this disease at earlier stages when patients have the best chance for better outcomes,” said Dr. Marjorie Green, senior vice president and head of late-stage oncology, global clinical development, Merck Research Laboratories, in a press release on December 11, 2023.
“By combining KEYTRUDA with V940 (mRNA-4157), a promising new modality, we are researching innovative new approaches for earlier stage non-small cell lung cancer.”
As previously announced, in addition to INTerpath-002, the combination of V940 (mRNA-4157) plus KEYTRUDA is being investigated in INTerpath-001, a global, randomized, double-blind, placebo- and active-comparator-controlled Phase 3 trial evaluating patients with resected high-risk (Stage IIB-IV) melanoma.
INTerpath-001 is actively screening in 14 countries, representing 38 sites.
The companies confirmed they plan to expand the comprehensive clinical development program for V940 (mRNA-4157) to additional tumor types.

The U.S. Centers for Disease Control and Prevention (CDC) continues to publish Trave Health Notices regarding diphtheria outbreaks in various countries in 2023.
On December 7, 2023, the CDC posted a Level 2 - Practice Enhanced Precautions notice regarding an outbreak of diphtheria in several districts in Guinea, which is located in western Africa.
Diphtheria is a severe infection caused by strains of Corynebacterium diphtheriae bacteria that make a toxin. The toxin can cause people to get very sick. Diphtheria bacteria spread from person to person through respiratory droplets, like from coughing or sneezing.
People can also get sick from touching open sores or ulcers of people ill with diphtheria, according to the CDC.
Diphtheria is a vaccine-preventable disease.
Unfortunately, an estimated 16% of children worldwide had no or incomplete vaccination coverage.
The U.S. CDC says most travelers visiting outbreak areas should receive an age-appropriate dose of diphtheria toxoid-containing vaccine if they are not fully vaccinated or have not received a booster dose within five years before departure.
There are 11 vaccines available for use to help protect against diphtheria in 2023. Diphtheria and other travel vaccines are offered at many clinics and pharmacies in the U.S.

The Africa Centres for Disease Control and Prevention (Africa CDC) recently welcomed the announcement from The Global Vaccine Alliance (GAVI) Board for the establishment of the African Vaccine Manufacturing Accelerator (AVMA).
The AVMA is a financing mechanism to create a sustainable vaccine manufacturing industry in Africa. It will make up to $1 billion in funds available to support vaccine manufacturing in Africa.
Dr. Jean Kaseya, Africa CDC Director General, commented in a press release on December 8, 2023, "Today is a significant moment for Africa. The targeted USD 1 Billion from GAVI to African Manufacturers is a game changer for the continent and advances our efforts towards vaccine self-reliance."
"The African Union has set a target for the continent to produce 60% of the vaccines needed by 2040, and the AVMA is indeed an accelerator towards that ambition."
"GAVI has been an incredible partner in this; we will continue to advance together on this journey of self-reliance. Together, we are united with a mission for vaccine equity."
The launch of AVMA is an important message from our partners that Africa will no longer be solely a recipient of vaccines but an active member and contributor to the global vaccine ecosystem.
The collaboration has seen several vaccine manufacturing projects taking shape, and others are in the works to guarantee self-reliance in Africa should any health emergency or outbreak hit the continent.
In addition, Africa CDC remains committed to collaborating with all partners and stakeholders in the vaccine ecosystem to facilitate the full operationalization of AVMA and expedite the attainment of health security, as envisaged in Agenda 2063.
The Coalition for Epidemic Preparedness Innovations (CEPI) recently announced it had partnered with Jurata Thin Film, Inc. to advance development of thermostable under-the-tongue vaccine films as a needle-free vaccine delivery platform.
On December 5, 2023, CEPI confirmed that it will provide up to an initial $1.2 million to support Jurata's proprietary innovative formulation platform, which, if shown to be successful, could help expand access to vaccines in underserved regions and advance the global response to future emerging infectious disease outbreaks.
CEPI's initial funding will support optimizing the composition and process of creating thin films and preclinical studies.
Under the agreement with CEPI, Jurata will create vaccine films to remain stable at 2-8 degrees, 25 degrees, and 40 degrees.
Jurata will optimise the composition of the films by testing various buffers, pH, stabilizers, sugars, salts, and different drying parameters and assessing how this affects vaccine stability and delivery.
Jurata aims to improve vaccine accessibility by stabilizing the 3D structure of mRNA-containing lipid nanoparticle vaccine materials, provided by Quantoom Biosciences, part of Univercells, into a thin thermostable film, thereby removing frozen storage needs.
The vaccine films are also lightweight and compact, simplifying the transportation process and potentially allowing for more doses to be shipped at any one time compared to current needle-and-syringe distribution.
Dr. Irnela Bajrovic, Chief Scientific Officer, Jurata, commented in a press release, "Our stabilising formulations have the potential to facilitate global access to mRNA vaccines, and our thin film delivery platform could make vaccine administration far easier than needle-and-syringe injections."
"We are grateful to CEPI for supporting our innovative technology and look forward to working with Quantoom to show the breadth of mRNA vaccines our technology can stabilize and deliver."
This is the fourth partner to be announced as part of CEPI's Call for Proposals for thermostable vaccine manufacturing innovations, announced in January 2022.
Thermostable vaccines are also identified as a preferred vaccine characteristic by the World Health Organization.

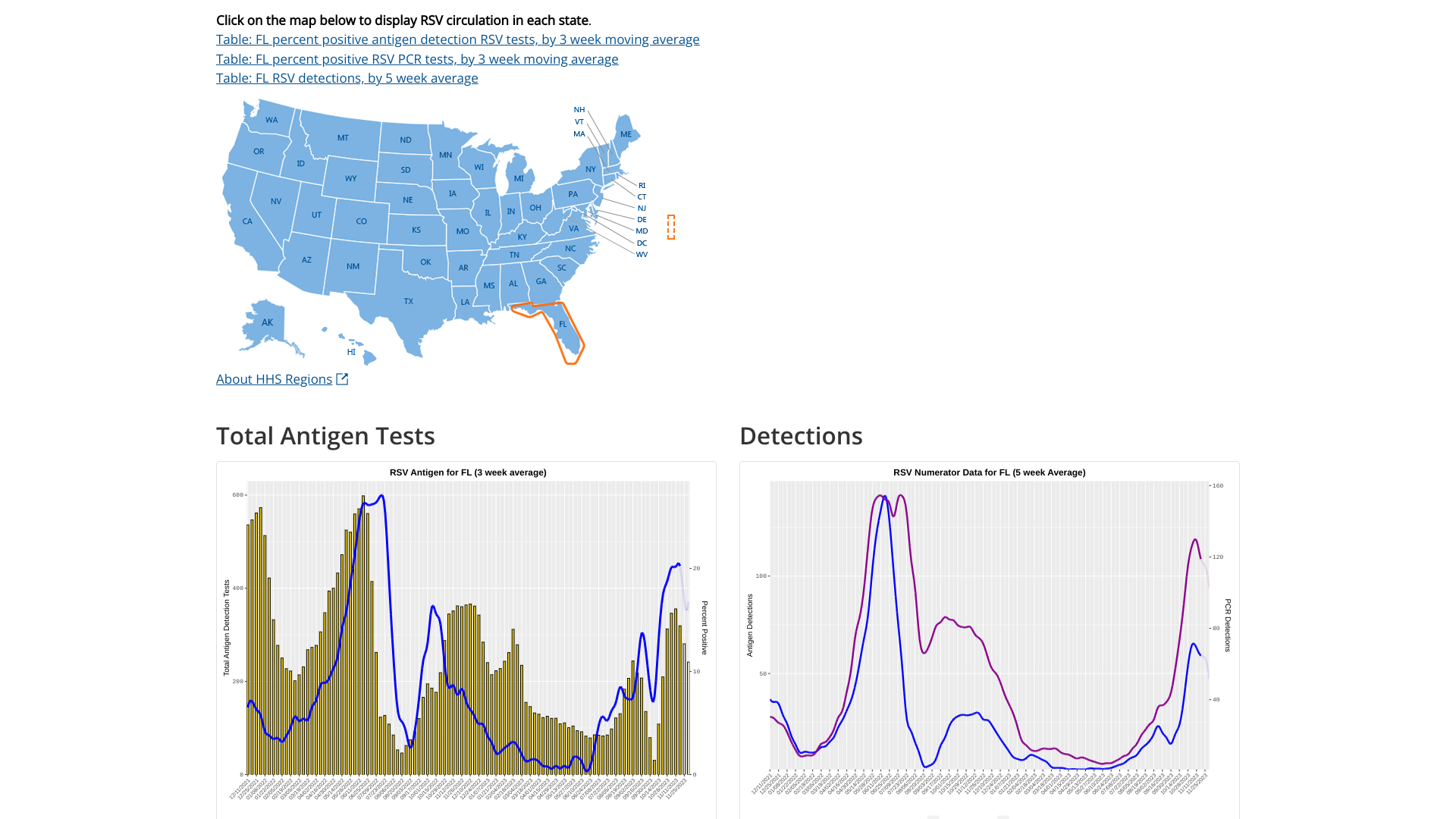
Since most respiratory syncytial virus (RSV) activity in the United States is initially recorded in the state of Florida, watching for the bending of the curve offers insights regarding future cases.
As of December 8, 2023, the U.S. Centers for Disease Control and Prevention (CDC) updated its RSV detection graphs to display the 5-week moving average. RSV infections typically occur during late fall, winter, and early spring.
There are variations in the timing of RSV outbreaks between regions and communities in the same region.
For Florida, the two charts indicate RSV's peak was in late November 2023.
Separately, the Florida Department of Health reported that as of December 2, 2023, RSV activity was decreasing in hospital admissions, positivity tests, and ER visits.
But, there was an RSV outbreak in Pinellas County.
From a prevention perspective, both RSV vaccines are available at most clinics and pharmacies in Florida. However, the CDC recently confirmed the percentage of seniors receiving an RSV vaccine was just 15.9%.
Unfortunately, the RSV antibody passive immunization for infants, Beyfortus™, remains in limited supply.
Virtually all children get an RSV infection by the time they are two years old. Most of the time, RSV will cause a mild, cold-like illness. RSV antibodies can help protect children from severe disease from an RSV infection.
RSV can be dangerous for infants and young children. Each year, thousands are hospitalized due to RSV infection, says the CDC.
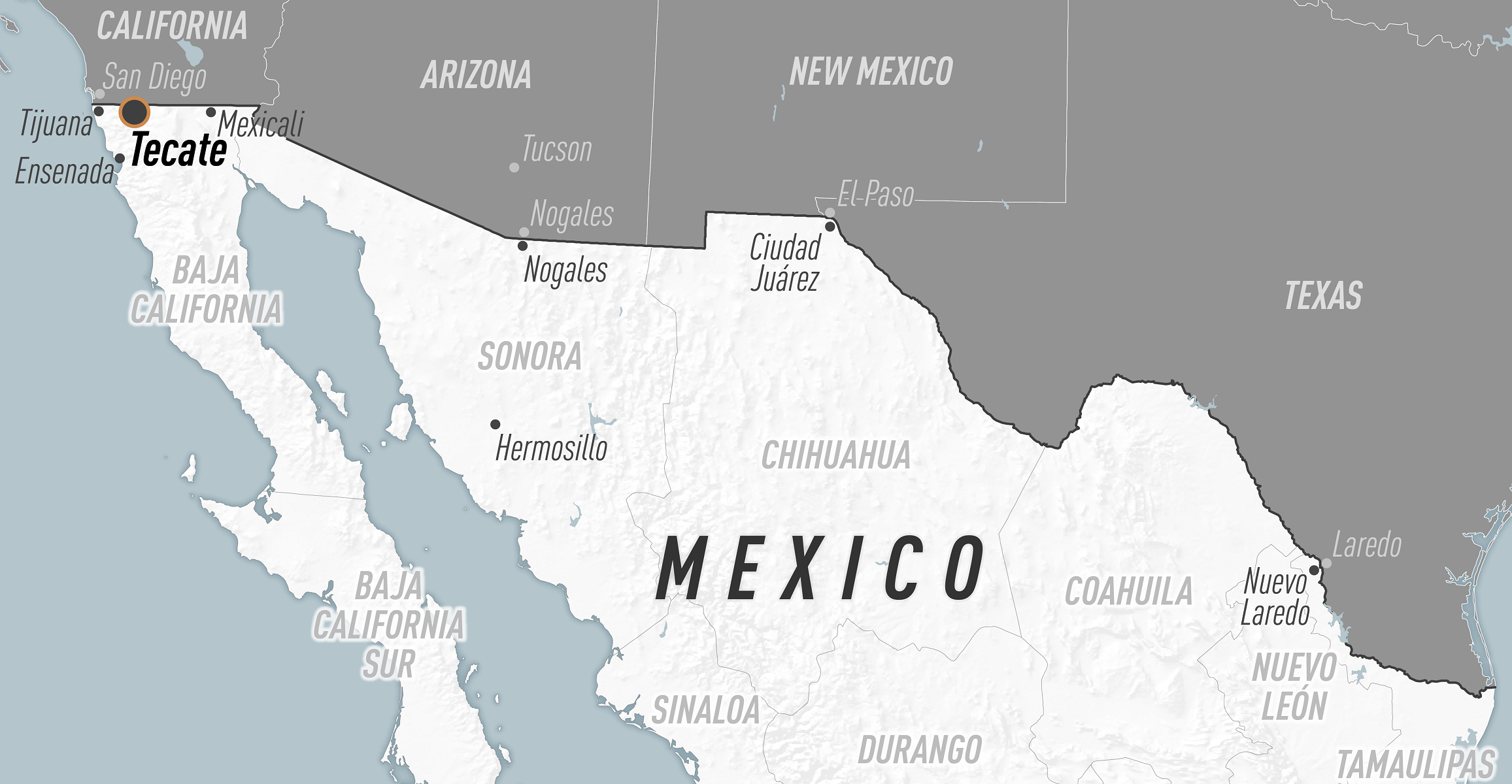
The Centers for Disease Control and Prevention (CDC) today issued a Health Alert Network Health Advisory confirming an outbreak of Rocky Mountain spotted fever (RMSF) among people in the United States with recent travel to or residence in the city of Tecate, state of Baja California, Mexico.
As of December 8, 2023, five patients have been confirmed with RMSF, and 3 have died since late July 2023.
Each patient had travel to or residence in Tecate within two weeks of illness onset.
RMSF is endemic in multiple border states in northern Mexico, including but not limited to Baja California, Sonora, Chihuahua, Coahuila, and Nuevo León.
RMSF is a severe, rapidly progressive, and often deadly disease transmitted by infected ticks, although many patients do not recall being bitten by a tick.
Furthermore, the CDC says dogs can carry infected ticks that can bite humans.
This disease progresses rapidly, with about half of all people who die from this disease succumbing within eight days of illness onset.
The CDC endorses Doxycycline as the treatment of choice for RMSF patients of all ages.
Doxycycline is offered in various pharmacies under different brand names.
As of December 9, 2023, there are no RMSF vaccines approved by the U.S. FDA.
The U.S. Centers for Disease Control and Prevention (CDC) today announced very few seniors have received an approved vaccine that protects against Respiratory syncytial virus (RSV).
This common respiratory virus can become severe and require hospitalization.
As of December 8, 2023, the CDC confirmed the percentage of adults 60+ reporting receiving an RSV vaccine was 15.9%.
This CDC data indicates an uptake gap compared to a recent survey.
A recent poll by the University of Michigan's Institute for Healthcare Policy and Innovation revealed that 21% of seniors want an RSV vaccination in 2023.
Unlike previous RSV seasons, there are two RSV vaccines endorsed by government agencies, which are as follows:
AREXVY™ RSV vaccine is approved for adults aged 60 and above.
ABRYSVO™ RSVpreF RSV bivalent vaccine is from Pfizer Inc.
The U.S. Food and Drug Administration, the U.K.'s Medicines and Healthcare products Regulatory Agency, and the European Commission recommend adults 60 years and older and pregnant women receive a single dose of an RSV vaccine based on discussions with healthcare providers and under certain conditions.
Dr. Mandy Cohen, Director of the CDC, commented during a media interview on December 8, 2023, "What we know is that several viruses are circulating, but we also have tools to protect ourselves."
"And we're hoping more can do that before the Christmas holiday."
In the U.S., RSV vaccines are generally available at clinics and pharmacies.