Search API
Sanofi and AstraZeneca recently announced they continue to engage with government officials, healthcare providers, and others in the United States regarding the supply of Beyfortus™ (nirsevimab-alip), a respiratory syncytial virus (RSV) monoclonal antibody therapy.
On December 14, 2023, the companies wrote, Since we launched in September 2023, hundreds of thousands of infants in the U.S. have received Beyfortus: 50 mg or 100mg Injection.
This is the first season in which passive immunization for babies against RSV has been provided broadly.
By the end of January 2024, a total of 1.4 million babies will be offered protection against RSV, a 27% increase over the initial supply forecast for the 2023-2024 RSV season in the U.S.
Beyfortus is a prescription injectable medicine used to help prevent a serious lung disease caused by RSV in babies under one year of age born during or entering their first RSV season and for children up to 24 months of age who remain at risk of severe RSV disease through their second RSV season.
It is a recombinant human immunoglobulin G1 kappa (IgG1ĸ) long-acting mAbs that binds to the prefusion conformation of the RSV F protein.
The most common side effects of Beyfortus include rash and pain, swelling, or hardness at the site of your child’s injection.
On October 23, 2023, the U.S. CDC published Interim Recommendations to Protect Infants from RSV (CDCHAN-00499) during the 2023–2024 RSV season. RSV antibody therapy was initially authorized in the U.S. in 1998.

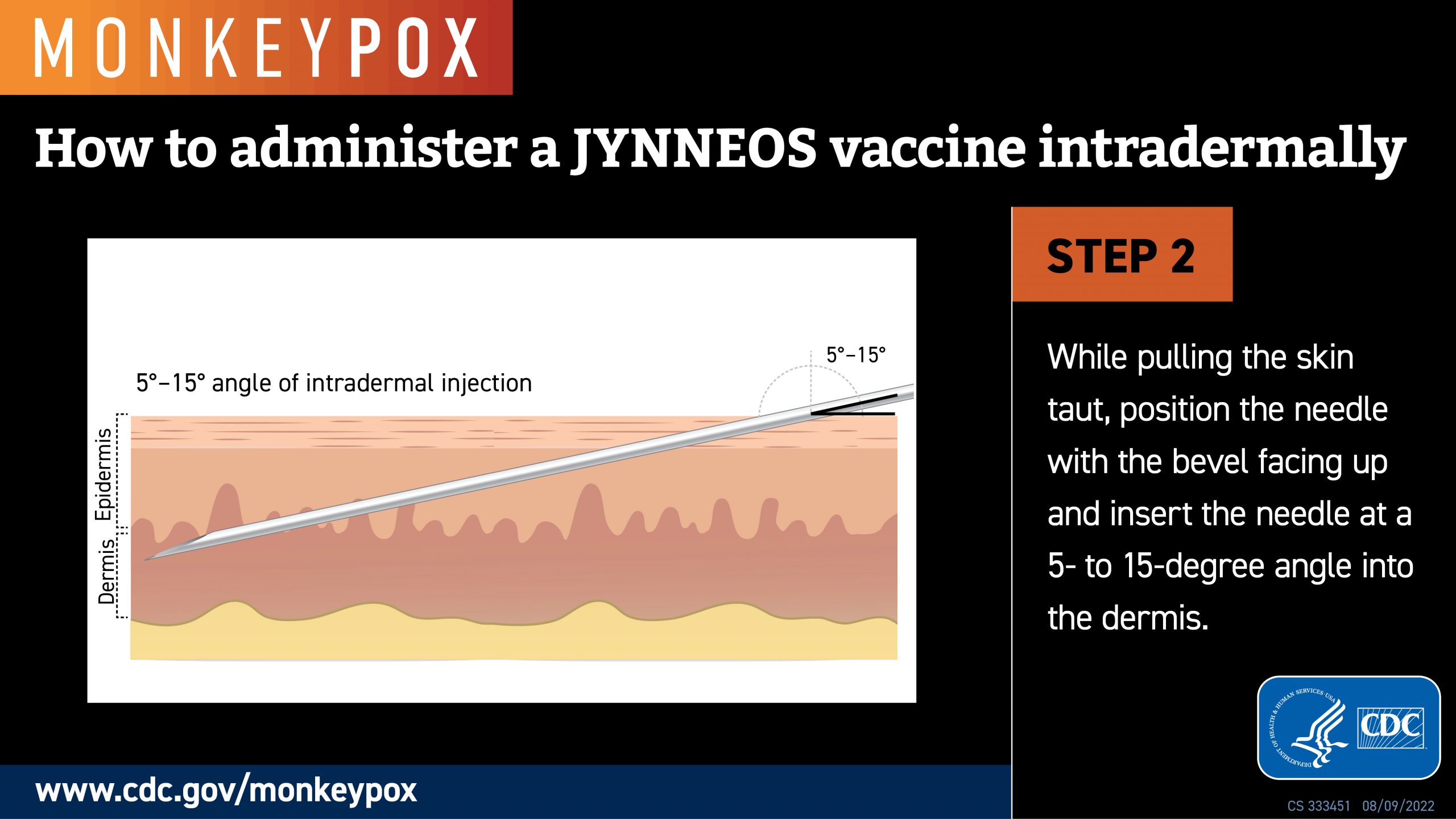
Led by researchers at NYU Grossman School of Medicine, a recent study showed no significant difference in the strength of the immune response (IgG antibodies) between most of those who received their vaccine injections in small doses between layers of the skin.
In some cases, the IgG antibodies were detected more than six months after the second and final JYNNEOS® vaccination.
Because of limited space between skin layers, intradermal injections can only accommodate small doses, while larger doses generally require subcutaneous injections.
The smaller doses, about one-fifth of the usual full dose and spread out by as long as three months, were authorized by the U.S. FDA and CDC in August 2022.
About 155,000 New Yorkers have been vaccinated, mainly using smaller doses.
"Our study shows that smaller vaccine doses of mpox vaccine administered in two doses spread out over weeks to months were similar to the full (subcutaneous) FDA-approved dose," said study co-lead investigator and infectious disease specialist Angelica Cifuentes Kottkamp, MD, in a press release on December 14, 2023,
"Implementing the smaller dose was a good emergency measure in the face of immediate shortages of the vaccine," said Dr. Kottkamp, an assistant professor in the Department of Medicine at NYU Langone Health.
Additionally, the New England Journal of Medicine published a Correspondence that revealed people fully vaccinated with two smaller JYNNEOS doses had an immune response four times stronger than those who did not complete the vaccination series and had only one dose.
This study's finding is significant since The Lancet Infectious Diseases reported on December 7, 2023, that 12% of JYNNEOS vaccinated individuals were non-antibody responders.
Bavarian Nordic codeveloped JYNNEOS with the U.S. Government to ensure adult populations, including people with weakened immune systems, could be protected from smallpox.
On February 22, 2023, the U.S. CDC issued Interim Clinical Considerations confirming that mpox vaccination should continue to be offered to people with the highest potential for exposure to mpox. The U.K. and Europe have issued similar notices in 2023.
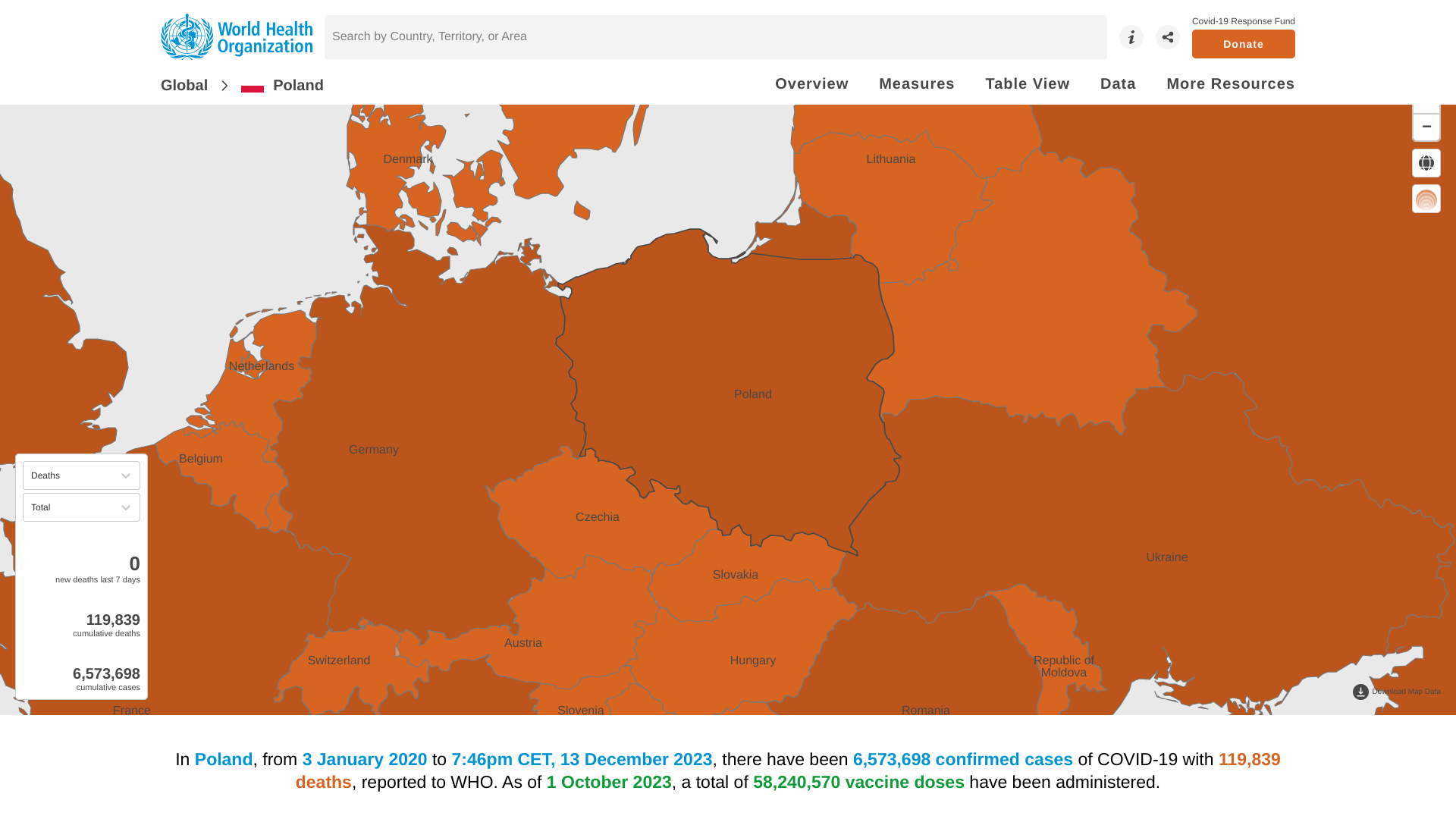
Novavax Inc. recently announced its updated protein-based COVID-19 vaccine is now the only COVID-19 vaccine option available in the Republic of Poland.
Doses were distributed by the appropriate Polish authorities in 2023 and made available for this season's vaccination campaign.
Novavax's vaccine is available in Polato to prevent COVID-19 in individuals aged 12 and older.
In Poland, from January 2020 to early December 2023, there have been 6,573,698 confirmed cases of COVID-19, with 119,839 related deaths.
As of October 2023, a total of 58,240,570 doses of various COVID-19 vaccines had been administered in Poland.
We are pleased that our updated vaccine is available in time for the upcoming Christmas and winter holidays, wrote the company on December 11, 2023. We are honored to support the Polish government and the country's healthcare workers in helping to protect Polish citizens and their loved ones against COVID-19.
Recent non-clinical data showed that Novavax's updated COVID-19 vaccine induced functional immune responses against XBB.1.5, XBB.1.16 and XBB.2.3 variants. Additional non-clinical data demonstrated that Novavax's vaccine-induced neutralizing antibody responses to subvariants BA.2.86, EG.5.1, FL.1.5.1, and XBB.1.16.6 as well as CD4+ polyfunctional cellular (T-cell) responses against EG.5.1 and XBB.1.16.6.
These data indicate that Novavax's vaccine can stimulate both arms of the immune system and may induce a broad response against currently circulating variants.
As of December 14, 2023, Sweden and Italy have authorized Novavx's updated vaccine. Throughout the recent pandemic, Novavax COVID-19 Vaccines have been authorized in Europe, the U.S., the U.K., and numerous other countries under various brands.
The U.S. government today announced that Sanofi and AstraZeneca will make available 230,000 additional doses of a new passive immunization that prevents lower respiratory tract infections in infants caused by the respiratory syncytial virus (RSV).
Announcement on December 14, 2023, the additional doses of Beyfortus™ (Nirsevimab-alip) are scheduled for delivery in January 2024.
Beyfortus, an extended half-life monoclonal antibody, was approved by the U.S. FDA in July 2023.
This new allocation is in addition to the 77,000 Beyfortus doses released to the U.S. in November 2023.
Sanofi had previously confirmed that demand for this product, especially for the 100 mg doses, had been higher than anticipated.
In clinical trials, a single injection reduced the chances of severe RSV infection by 74.5%.
RSV disease can be prevented either by giving antibody products to infants and young children or by giving their mothers RSV vaccine during pregnancy, says the U.S. CDC.
According to a Johns Hopkins news article published in July 2023, monoclonal antibodies work by providing immediate and short-term protection, whereas vaccines “boost your immunity in the future.
David Dowdy, MD, professor in Epidemiology, explained that your immune system can’t “learn” anything from an antibody. The drug is “basically to protect during a time of life when people are most vulnerable.”
The protection that Beyfortus provides is called “passive immunity” because it does not come from the person’s immune system.

The Japan-based Global Health Innovative Technology Fund announced today that it will invest approximately $3.3 million in a Phase III clinical trial led by Fosun Pharma for a triple artemisinin combination drug (Artemether-Lumefantrine-Amodiaquine fixed-dose formulation) against malaria.
Importantly, this antimalarial drug includes developing a co-formulated child-friendly version, given that most malaria cases are in children.
This combination drug candidate should have a significant public health benefit as it is expected to play an essential role in the fight against artemisinin partial resistance (ART-R), which is now observed in Southeast Asia and Africa.
Wen Deyong, CEO of Fosun Pharma, commented in a press release on December 14, 2023, "The collaboration will accelerate the launch of this new drug to actively respond to the threat of artemisinin partial resistance and partner drug resistance of Plasmodium falciparum parasites and save more lives from malaria."
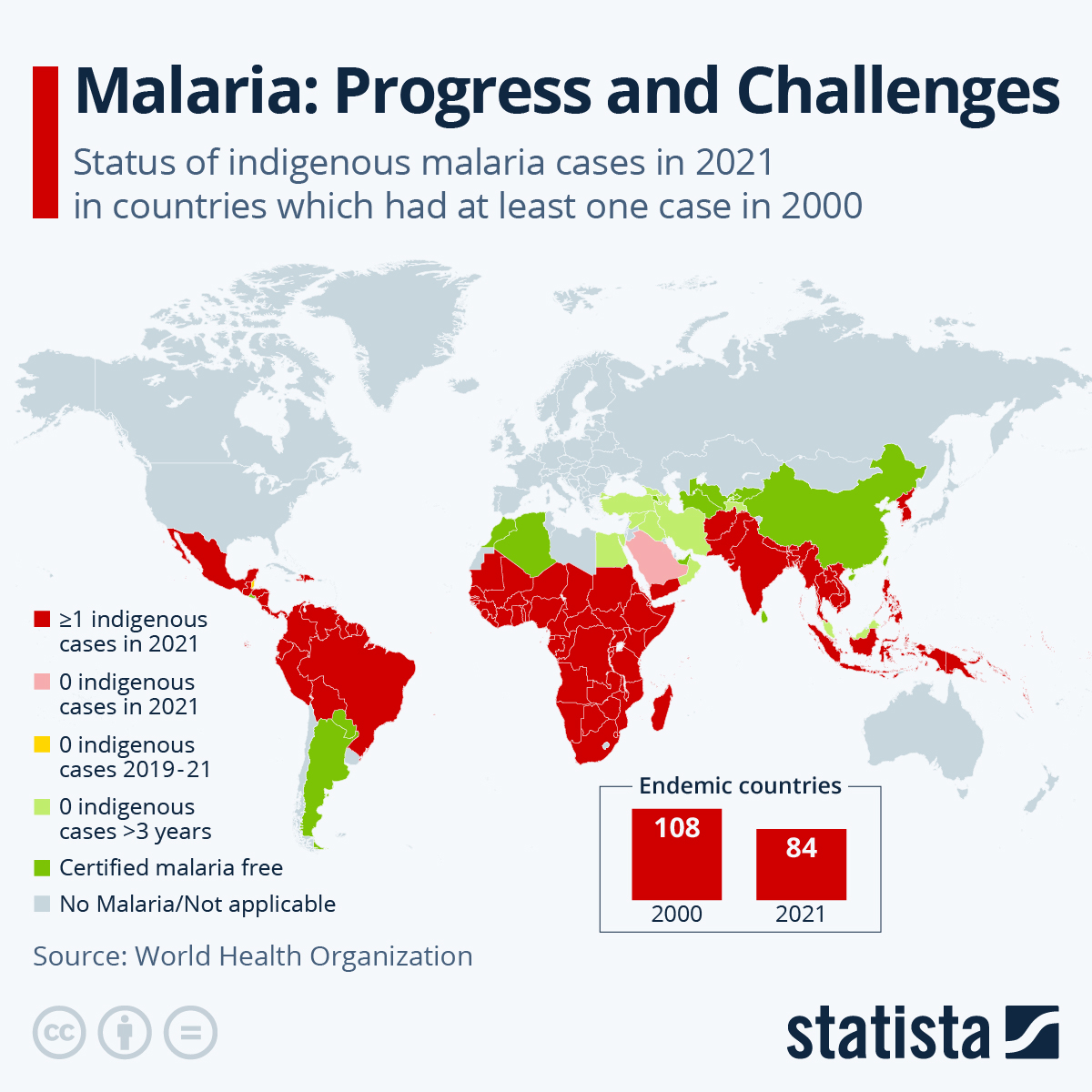
Malaria is an infectious parasitic disease transmitted by mosquitos that affects approximately 250 million people annually and was responsible for about 620,000 deaths in 2021.
Infection in the African region constitutes 95% of the total global malaria cases, and children under the age of five account for 80% of all malaria deaths in this region.
Currently, two malaria vaccines are being deployed in Africa.
Before 2023, there were no immunizing tools to protect older adults from illness and death due to respiratory syncytial virus (RSV). This year, the U.S. Food and Drug Administration approved two RSV vaccines for older adults.
According to an Original Article published today by the New England Journal of Medicine, an mRNA-based vaccine was found to be very effective at protecting healthy adults.
In this ModernaTX, Inc. sponsored ongoing, randomized, double-blind, placebo-controlled, phase 2/3 clinical trial, participants were randomly assigned, in a 1:1 ratio, adults 60 years of age or older to receive one dose of mRNA-1345 (50 μg) or placebo.
The two primary efficacy endpoints were preventing RSV-associated lower respiratory tract disease with at least two signs or symptoms and at least three signs or symptoms. A key secondary efficacy endpoint was the prevention of RSV-associated acute respiratory disease. Safety was also assessed.
Overall, 17,793 participants were assigned to receive the mRNA-1345 vaccine candidate. The median follow-up was 112 days (range, 1 to 379).
The primary analyses were conducted when at least 50% of the anticipated cases of RSV-associated lower respiratory tract disease had occurred.
Vaccine efficacy was 83.7% (95.88% confidence interval [CI], 66.0 to 92.2) against RSV-associated lower respiratory tract disease with at least two signs or symptoms and 82.4% (96.36% CI, 34.8 to 95.3) against the disease with at least three signs or symptoms. Vaccine efficacy was 68.4% (95% CI, 50.9 to 79.7) against RSV-associated acute respiratory disease.
Protection was observed against both RSV subtypes (A and B) and was generally consistent across subgroups defined according to age and coexisting conditions.
Participants in the mRNA-1345 group had a higher incidence than those in the placebo group of solicited local adverse reactions (58.7% vs. 16.2%) and systemic adverse reactions (47.7% vs. 32.9%). Most of these reactions were mild to moderate in severity and were transient.
However. serious adverse events occurred in 2.8% of the participants in each trial group.
In summary, these researchers wrote, 'This phase 2–3 efficacy trial showed that a single 50-μg dose of the mRNA-1345 vaccine in adults 60 or older was efficacious against a spectrum of RSV-confirmed respiratory disease.'

A Yale University-led study published today in the journal Pediatrics announced the rate of invasive pneumococcal disease (IPD) in children decreased by about 72% (incidence rate ratios 0.28, 95% CI 0.18–0.45) over twenty years.
This research found IPD rates continued to decline after the replacement of PCV7 with PCV13.
And during the recent pandemic, the rate of IPD among children aged <18 years reached 1.6 per 100,000, the lowest incidence observed over the 20 years.
In the PCV13 vaccine era, approximately one-third of the IPD cases in children aged >5 years had at least one underlying condition.
Serotypes 19A and 7F contributed 342 (48.9%) of all cases before implementation of PCV13 (2002–2010).
Serotype 3 (31, 8.6%), and non-PCV13 serotypes 15B/C (39, 10.8%), 33F (29, 8.0%), 23B (21, 0.8%), and 35B (17, 4.7%) were responsible for 37.8% of cases in PCV13 era (2011–2021).
Furthermore, penicillin nonsusceptibility declined (9.8% vs. 5.3% in the pre-/late PCV13 era, P = .003). However, it has become more common among non-PCV13 serotypes than vaccine serotypes (14.8% vs 1.4%, P < .001).
These Yale Child Health Research Center researchers concluded that robust ongoing surveillance networks are critical for identifying emerging pneumococcal serotypes and developing next-generation vaccine formulations.
As of December 2023, various pneumococcal vaccines are available at clinics and pharmacies in Asia, Europe, Japan, the U.S., and the United Kingdom.

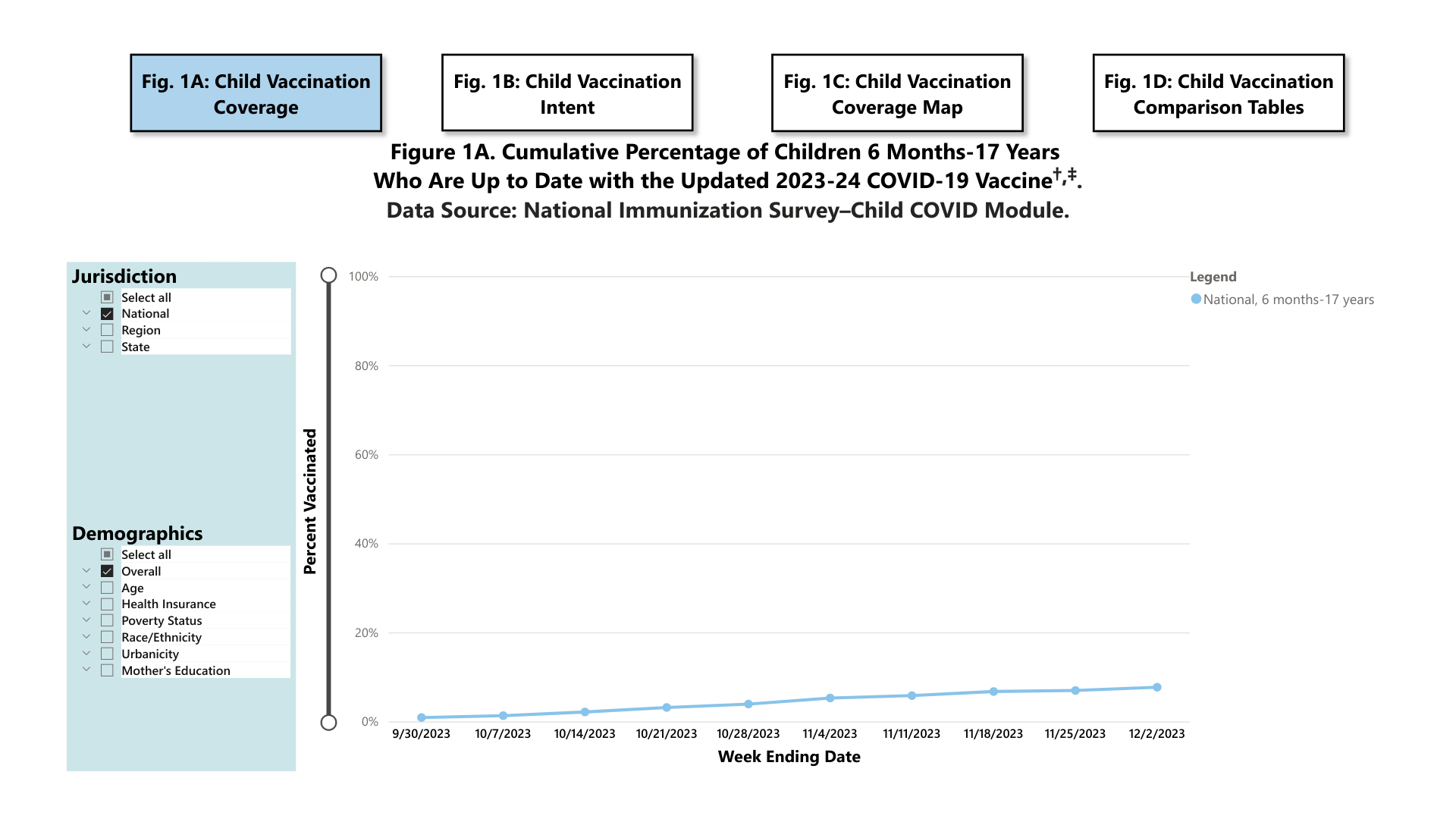
The U.S. CDC's COVIDVaxView COVID-19 Vaccination Dashboard was updated today, reflecting current trends in children's, adults, and pregnant women's vaccination rates.
As of December 13, 2023, the CDC confirmed:
As of December 2, 2023, 7.7% (95% Confidence Interval: 6.5%-8.9%) of children (six months to 17 years) were reported to be up to date with the 2023-24 COVID-19 vaccine. An additional 18.6% (15.9%-21.3%) of children had a parent who said they planned to get their child vaccinated.
And 17.2% (95% Confidence Interval: 16.3%-18.1%) of adults reported receiving an updated 2023-24 COVID-19 vaccine since September 14, 2023. An additional 14.6% (13.4%-15.9%) said they plan to get vaccinated.
Furthermore, 9.6% of pregnant women had received the updated 2023-24 COVID-19 vaccine. Vaccination coverage was highest among non-Hispanic Asian (16.5%) pregnant women and lowest among non-Hispanic Black (3%) women.
Additional COVID-19 vaccination data by demographic characteristics at the national level and overall estimates by jurisdiction are available at this CDC link.