Search API
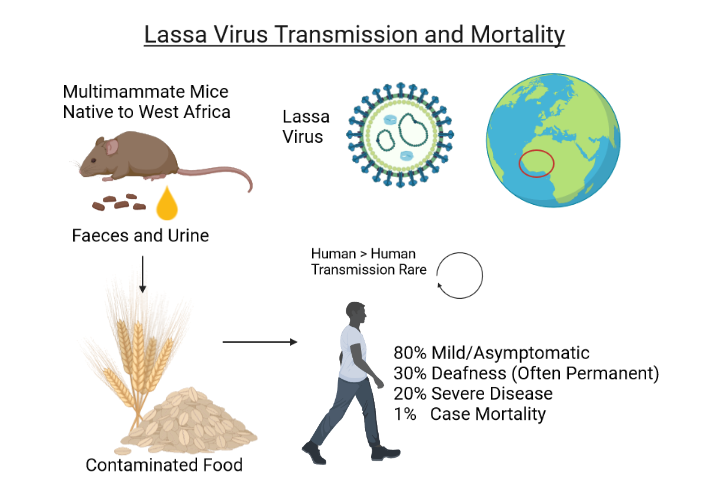
Despite extensive efforts, the Federal Republic of Nigeria continues to confront an expanding Lassa fever outbreak in 2024, reporting new cases and deaths.
Throughout 2024, Nigeria's Centre for Disease Control and Prevention (NCDC) confirmed 682 cases and 128 deaths, which is a Lassa fever case fatality rate of 18.8%, far exceeding the rate recorded in 2023.
The report identified 62% of confirmed cases originating from Ondo, Edo, and Bauchi states.
The NCDC wrote in 2023 that annual outbreaks of Lassa fever also involved infection and death of healthcare workers. This loss of life is not just a statistic but a significant loss of a loved family member, a spouse, a parent, and often a seasoned healthcare worker and team member. This exacerbates the challenge of insufficient human resources for health in the country.'
As of March 2024, the U.S. Food and Drug Administration and the European Medicines Agency had not approved a Lassa fever virus vaccine candidate.
However, four vaccine candidates (INO-4500, MV-LASV, rVSV∆G-LASV-GPC, and EBS-LASV) have entered the clinical clinical stage.
Without an available vaccine, the U.S. CDC says the risk of Lassa virus infection is associated with peridomestic rodent exposure, where inappropriate food storage increases the risk.
The CDC says people should practice safe food precautions and avoid handling, cooking, or eating raw or undercooked meat or animal products.
During the recent pandemic, many scientists were stunned by how fast government agencies authorized new vaccines. Traditionally, it took about eight years for a vaccine to progress through the various stages of clinical trials to obtain market approval.
Many scientists were amazed by the speed at which new vaccines were authorized by government agencies during the recent pandemic.
Traditionally, it took about eight years for a vaccine to progress through the various stages of clinical trials to obtain market approval.
However, new research published on March 21, 2024, suggests that the clinical development of innovative medicines and vaccines will continue to accelerate.
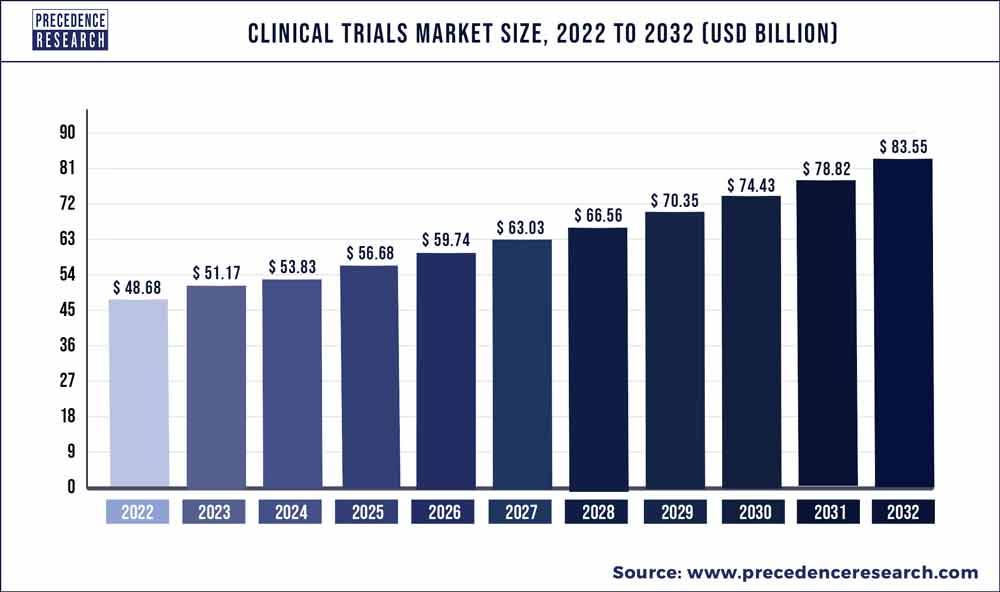
Precedence Research Pvt. Ltd. reported today that the global clinical trials market size was valued at $48.68 billion in 2022 and is predicted to reach about $83 billion by 2032. Clinical trial research was valued at $24.61 billion in the US sub-market in 2022.
The vaccine sub-market reached $102.9 billion in 2023 and is forecasted to expand by 47% to $35.1 billion by 2030.
Furthermore, there is plenty of investor interest in funding new, innovative vaccines.
In the past ten years, companies with infectious disease vaccine programs received 3.4% of the total ($6.5 billion) venture capital raised for biopharmaceutical companies.
Moreover, the promise of personalized vaccines tailored to subpopulations may disrupt the one-size-fits-all vaccination model, further expanding the need for clinical trial research.
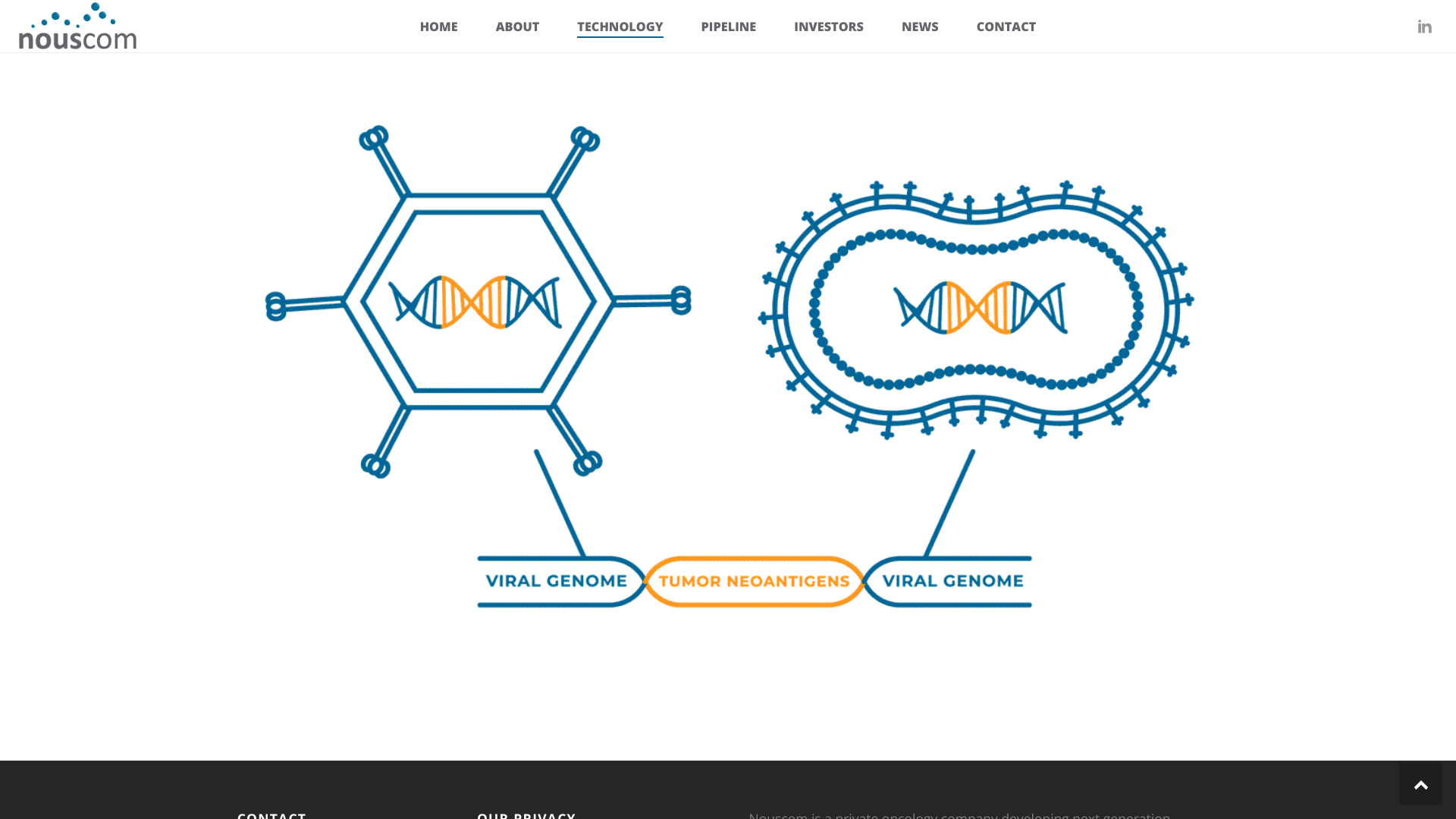
A corporate venture firm focused on investing in companies developing innovative solutions and ideas today announced its participation in an extension of the Series C financing round for Nouscom, a clinical-stage immuno-oncology company.
Nouscom raised approximately $82 million in its oversubscribed Series C round, which was first announced in November 2023.
Angelini Ventures joins a syndicate of top-tier international healthcare investors by participating in Nouscom’s financing.
This new financing will enable Nouscom, based in Basel, Switzerland. to continue advancing and expanding its wholly-owned clinical pipeline to achieve multiple clinical value catalysts, including:
- Readout from its ongoing randomized Phase 2 clinical trial for NOUS-209, an off-the-shelf cancer vaccine for treating Mismatch Repair/Microsatellite Instable Metastatic Colorectal Cancer.
- Phase 1b study completion and progression of NOUS-209 monotherapy in Lynch Syndrome carriers investigating the potential to intercept, prevent, or delay cancer in high-risk individuals.
- Completion of the Phase 1b study for NOUS-PEV, a personalized cancer immunotherapy, in combination with a checkpoint inhibitor in patients with advanced melanoma and entry into randomized Phase 2 trials in indications with high unmet medical needs.
Dr. Marina Udier, Chief Executive Officer at Nouscom, commented in a press release on March 21, 2024: “We are delighted to have extended our successful Series C financing round and excited to have Angelini Ventures on board. Their support strengthens our commitment to innovation and accelerates our journey towards delivering next-generation treatments for cancer patients.”
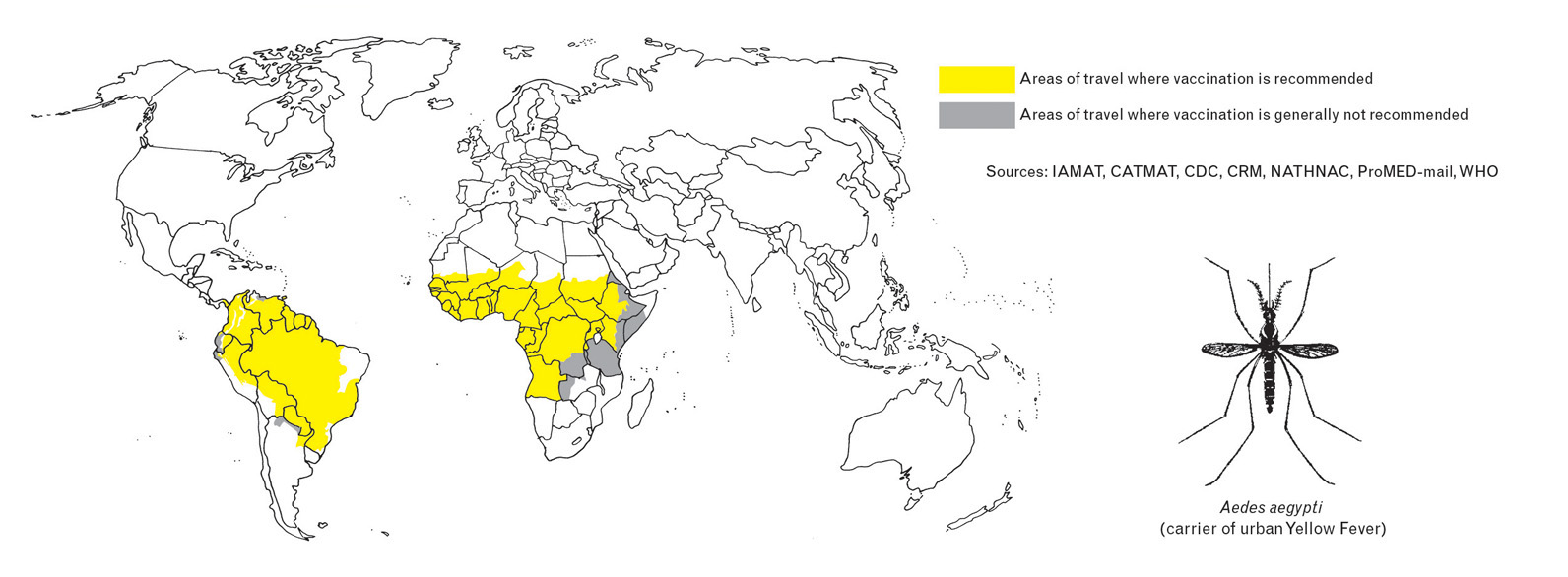
In 2023, the global Eliminate Yellow Fever Epidemics (EYE) secretariat led by the World Health Organization (WHO) coordinated preventive and responsive vaccination efforts.
As a result, about 62 million people in Africa received yellow fever vaccinations through EYE campaigns.
According to the WHO's Disease Outbreak News on March 20, 2024, a single yellow fever vaccination can provide lifelong protection and sustained immunity for around 90% of vaccinated individuals.
The WHO says yellow fever is an acute viral hemorrhagic disease. Cases can be difficult to distinguish from other viral hemorrhagic fevers such as arenavirus, hantavirus, or dengue. Related symptoms of yellow fever usually appear 3 to 6 days after the bite of an infected mosquito.
To notify international travelers, the U.S. Centers for Disease Control and Prevention advises against yellow fever vaccination when visiting countries with a low risk of virus exposure.
For Americans seeking protection from this mosquito-transmitted disease, the YF-VAX® vaccine is available at certified travel vaccine clinics and pharmacies in the U.S.
The CDC recently reported no locally acquired yellow fever cases in 2024.
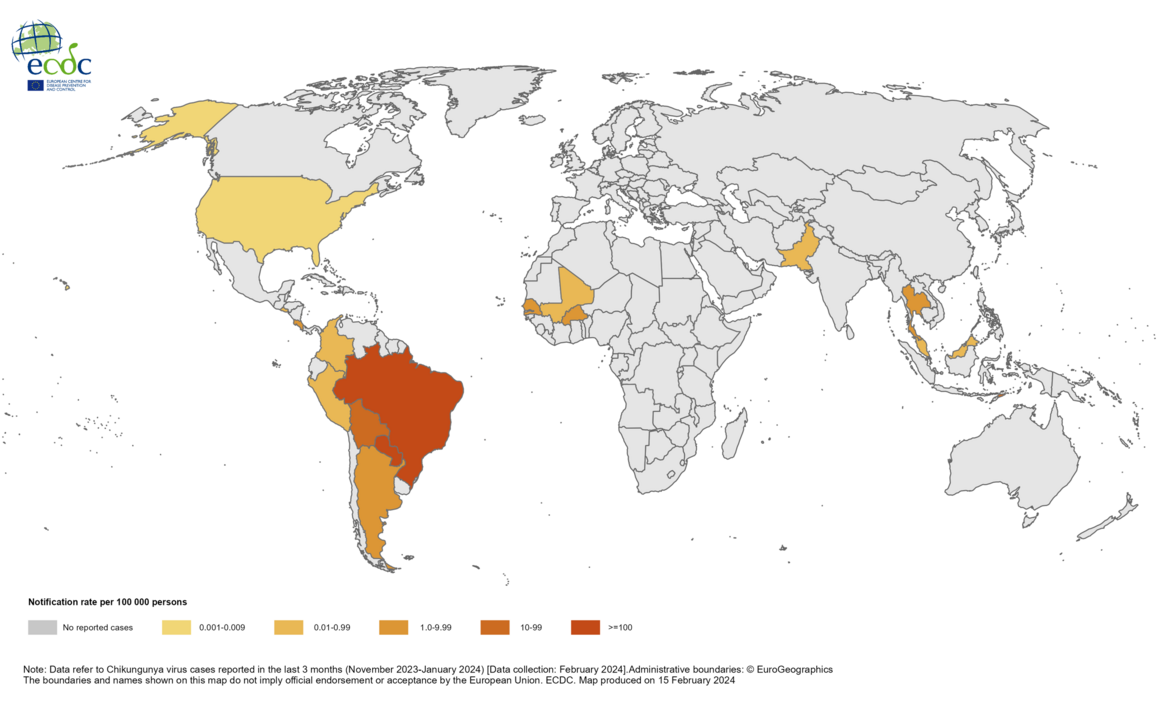
Millions of people's health is at risk by the mosquito-transmitted, vaccine-preventable chikungunya virus (CHIKV), but most Americans are unaware of its global impact.
In-person information about a newly approved chikungunya virus (CHIKV) vaccine will be shared at the Walter E. Washington Convention Center in Washington, D.C., from April 1-4, 2024.
Valneva SE, a France-based specialty vaccine company, today announced it will present its single-shot chikungunya vaccine for adults, IXCHIQ®, and participate in a panel discussion on efforts to eradicate chikungunya outbreaks at the 24th World Vaccine Congress.
XCHIQ®, the world's first and only chikungunya vaccine approved in the U.S., was recently recommended by the CDC's Advisory Committee on Immunization Practices for international travelers to at-risk areas.
As of March 21, 2024, CHIKV was identified in nearly 115 countries, primarily in the Region of the Americas. As of December 2023, approximately 460,000 CHIKV cases and 360 related deaths have been reported worldwide.
On the evening of April 2, Valneva will attend the Vaccine Industry Excellence Awards ceremony, where the vaccine is a finalist for the Best Prophylactic Vaccine award for IXCHIQ®.
And on April 3, Valneva's Chief Medical Officer, Dr. Juan Carlos Jaramillo, will participate in the "Vaccine Development and Efforts towards Eradicating Chikungunya" panel discussion.
For further details, contact Laetitia Bachelot-Fontaine, VP, Global Communications and European Investor Relations, at [email protected]

Valneva SE today reported its consolidated financial results for the year ending December 31, 2023, and provided several key corporate updates. Valneva's commercial portfolio is composed of three travel vaccines, IXIARO®/JESPECT®, DUKORAL®, and IXCHIQ®.
Valneva raised its 2024 product sales guidance to between €160 million and €180 million due to an improved outlook regarding the IXIARO® supply constraints anticipated in February 2024.
The Company made excellent progress across the R&D pipeline with the U.S. FDA's approval of the single-shot chikungunya vaccine IXCHIQ®, the world's first and only vaccine to address this significant unmet medical need.
In today's press release, Peter Bühler, Valneva's Chief Financial Officer, commented, "In 2023, Valneva successfully executed key strategic objectives despite a difficult economic environment."
"Our chikungunya vaccine IXCHIQ® became the world's first licensed chikungunya vaccine available to address this significant unmet medical need, and we also managed to surpass our pre-pandemic product sales."
Valneva will host a live webcast of its full-year 2023 results conference call on March 20, 2024, at 3 p.m. CET/10 a.m. EDT. The webcast will also be available on the Company's website. Please refer to this link: https://edge.media-server.com/mmc/p/hom3riyt.
Chikungunya is a viral disease transmitted by mosquitoes infected with the chikungunya virus. The World Health Organization says chikungunya outbreaks were identified in nearly 115 countries, primarily in the Region of the Americas, in 2023.