Search API
The world's first and only licensed chikungunya vaccine today presented further positive pivotal Phase 3 data in adolescents.
On May 13, 2024, Valneva SE announced following the initial analysis up to Day 29 post-vaccination, the most recent analysis of study VLA1553-321 evaluated the safety and immunogenicity six months after vaccination with a single dose of the chikungunya virus (CHIKV) vaccine IXCHIQ®.
The Day 180 results confirm the initial positive immunogenicity and safety data Valneva reported previously and are intended to support filing for potential label extension for use in adolescents aged 12 to 17 years.
The data are also expected to support the licensure of IXCHIQ® in Brazil, which would be the first potential approval for use in endemic populations.
The U.S. FDA approved IXCHIQ in November 2023, and the Centers for Disease Control and Prevention (CDC) recently adopted the Advisory Committee on Immunization Practices' recommendations on the use of the vaccine in the U.S.
Juan Carlos Jaramillo, M.D., Chief Medical Officer of Valneva, said in a press release, "We are highly encouraged by these data, as they reinforce the strong immunity and safety observed in adults and the elderly, upon which FDA approval was granted."
"Given the substantial risk that chikungunya presents to individuals residing in or traveling to endemic regions, it's imperative to ensure the vaccine is available to all age groups. This broader accessibility can help provide protection and mitigate the burden of this debilitating illness."
Three marketing applications are under review by the European Medicines Agency, Health Canada, and the Brazilian Health Regulatory Agency, with potential approvals in 2024.
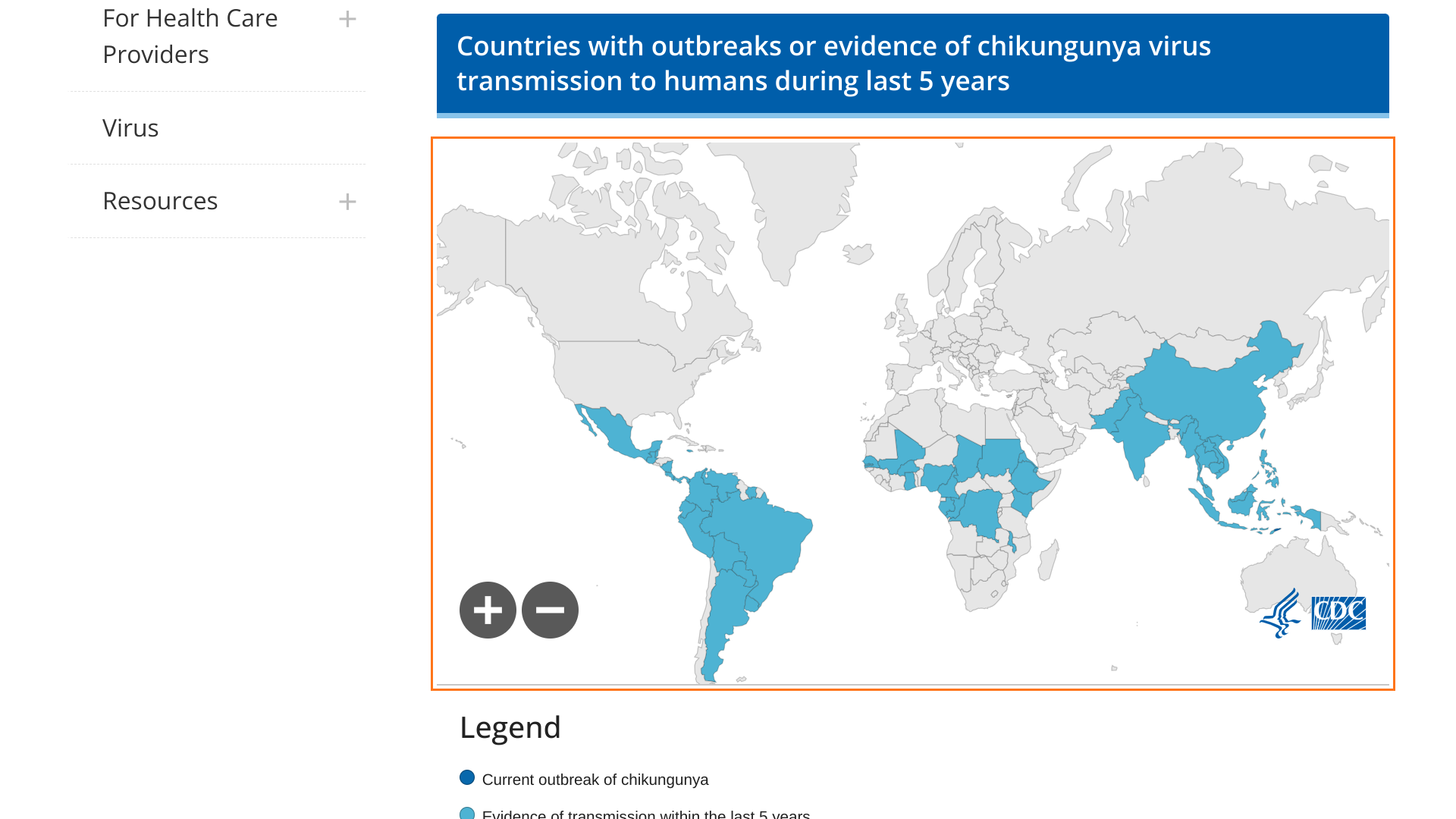
The CDC recently issued an updated Level 2 Travel Health Advisory confirming chikungunya vaccination is recommended for adults traveling to a destination with a current CHIKV outbreak.
Developed by Chengdu Weisjin Biomedical Technology Co., Ltd. (Wesjin Biotech), WGc-043 is an mRNA therapeutic cancer vaccine that recently received IND approval from the U.S. Food and Drug Administration (FDA).
According to public information from the FDA, WGc-043 Injection has been approved for two categories of indications: one is for adult patients with Epstein-Barr virus-positive advanced solid tumors who have undergone second-line systemic treatment.
The second indication is for adult patients with relapsed or refractory virus-positive hematoma.
This achievement, announced on May 9, 2024, marks the world's first approval of an Epstein-Barr virus (EBV)- related mRNA therapeutic cancer vaccine.
Once successfully launched, WGc-043 will provide a new treatment option for patients with advanced EB virus-positive solid tumors and hematologic malignancies.
EB virus is highly correlated with more than ten malignancies, including nasopharyngeal carcinoma, natural killer T-cell lymphoma, gastric cancer, lung cancer, liver cancer, esophageal cancer, breast cancer, cervical cancer, and autoimmune diseases such as multiple sclerosis and systemic lupus erythematosus.
According to a press release on May 10, 2024, WGc-043 shows promising efficacy, low toxicity, broad applicability, efficient scalability, and cost-effectiveness.
The company says that WGc-043 has already completed investigator-initiated trials, demonstrating superior safety and efficacy compared to other publicly available mRNA therapeutic cancer vaccines.
Specifically, the technical features of WGc-043 also include the antigen being the most broad-spectrum and safe protein sequence.
The originally designed immune enhancer (IE) is introduced into the mRNA molecule, and the mRNA delivery carrier is independently developed and obtained
A new type of LNP authorized by US and European patents (the safety of this LNP: has been verified in clinical trials of 3 varieties).
These designs enable WGc-043 to activate the patient's own anti-tumor immunity and generate tumor-killing cytotoxic T cells, antigen-specific antibodies, and memory T cells in the body, which is equivalent to CAR-T. The combined anti-tumor effect of monoclonal antibodies can also prevent tumor recurrence, have more efficient anti-cancer effects, and be superiorly safe.
Weisjin Biotechnology has filed over 60 invention patents, including the patent for ionizable lipids, which has been authorized by China, the United States, Europe, and other countries and regions.
As of May 12, 2024, no date has been announced regarding WGc-043's availability in the United States.

Moderna, Inc., today announced that the U.S. Food and Drug Administration (FDA) has notified it that due to administrative constraints, the agency does not expect to complete its review of the Biologics License Application for mRNA-1345, Moderna's investigational respiratory syncytial virus (RSV) vaccine, by the previously communicated Prescription Drug User Fee Act date of May 12, 2024.
The FDA informed Moderna on May 10, 2024, that it is working to conclude its review of mRNA-1345 by the end of May 2024.
The FDA has not informed Moderna of any issues related to vaccine safety, efficacy, or quality that would prevent the approval of mRNA-1345.

The U.S. CDC reported today that seasonal influenza activity continues to decline nationally, with outpatient respiratory illness stable and below baseline for the fifth consecutive week.
As of Week #18, the CDC says that flu viruses are among the several contributing factors to respiratory disease activity. To help people understand their health risks, the CDC provides updated, integrated information about influenza, RSV, and other disease activity every week.
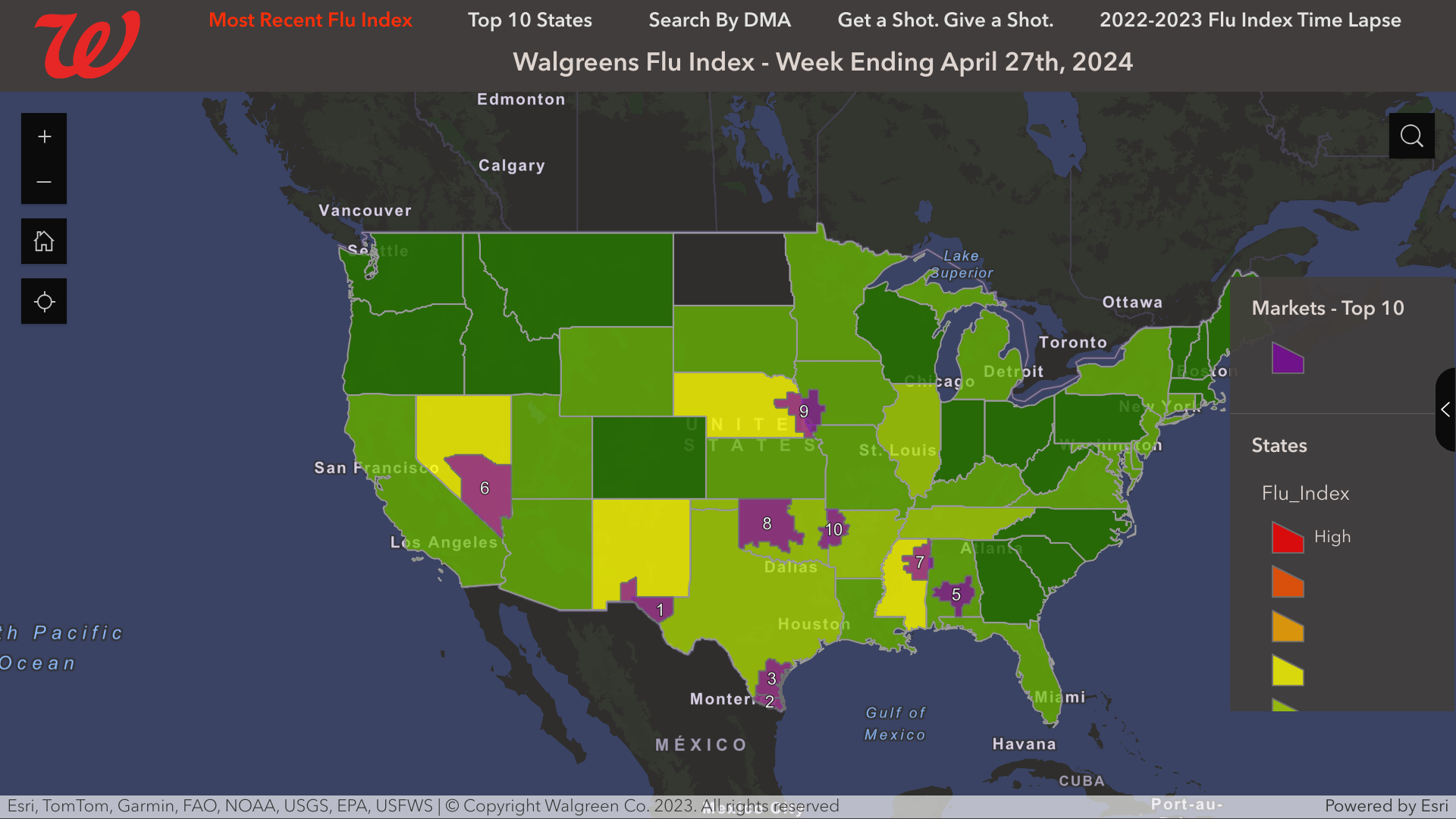
From a local perspective, the Walgreens Flu Index recently identified three Texas cities that led the USA with influenza activity in late April 2024.
These cities are:
- El Paso, Texas (Las Cruces, N.M.
- Harlingen-Weslaco-Brownsville-McAllen, Texas
- Corpus Christi, Texas
The CDC also disclosed that about 63 million flu shots have been administered at pharmacies and physician offices this flu season, which indicates about 6% fewer flu shots administered than last season.
These vaccines remain available at most pharmacies in the U.S.
The American Academy of Pediatrics journal recently confirmed data from the Vaccine Adverse Event Reporting System (VAERS) revealed over two dozen young children were incorrectly administered an adult RSV vaccine.
As reported in a Research Brief on May 9, 2024, these children were under the age of two between August 2023 and March 2024.
In January 2024, the Centers for Disease Control and Prevention (CDC) announced vaccine administration errors are known to occur.
Since the approval of RSV vaccines and the launch of a second-generation monoclonal antibody, VAERS has received reports of the Pfizer (Abrysvo) or GSK (Arexvy) vaccines being administered in error to children.
Additionally, the CDC and U.S. FDA have received reports of Arexvy being administered in error to pregnant women.
According to the U.S. CDC, no special monitoring is needed when an adult RSV vaccine is administered to children. However, if eligible, children who receive an RSV vaccine in error should receive Beyfortus™ to prevent severe RSV disease.
Beyfortus (Nirsevimab) is a long-acting monoclonal antibody that prevents severe RSV disease. As of May 10, 2024, Beyfortus is reported to be about 90% (95% CI = 75%–96%) protective against RSV-associated hospitalization in infants in their first RSV season.
The CDC says that although both monoclonal antibodies and vaccines provide protection, how they deliver protection differs.

The World Health Organization (WHO) today announced three human cases, including one death, of Middle East respiratory syndrome coronavirus (MERS-CoV) by the Ministry of Health of the Kingdom of Saudi Arabia (KSA).
According to the WHO's Disease Outbreak News on May 8, 2024, all three cases were middle-aged males with underlying health conditions.
The three cases are epidemiologically linked to exposures in a Riyadh healthcare facility, although investigations are ongoing to verify this and understand the transmission route.
In 2024, a total of four cases and two deaths have been reported from the KSA.
MERS is a viral respiratory infection caused by the MERS-CoV. Humans are infected with MERS-CoV from direct or indirect contact with dromedary camels, the virus's natural host and zoonotic source.
So far, non-sustained human-to-human transmission has occurred among close contacts and in healthcare settings. However, the WHO reports limited human-to-human transmission outside of healthcare settings.
Since 2012, when the first MERS-CoV case was reported in KSA, 2,613 MERS-CoV cases and 941 deaths (36%) have been reported from 27 countries in all six WHO regions.
Fortunately, no MERS-CoV cases have been reported from countries outside the Middle East since 2019.
The notification of the new cases does not change WHO's overall risk assessment, which remains moderate at both the global and regional levels.
As of May 9, 2024, no approved MERS vaccines are available.
However, in late 2023, Barinthus Biotherapeutics plc, the Coalition for Epidemic Preparedness Innovations, and the University of Oxford collaborated on fast-tracking the development of the VTP-500 vaccine candidate.
This $34.8 million project intends to take this MERS vaccine through Phase II clinical trials.