Search API
The Coalition for Epidemic Preparedness Innovations (CEPI) recently announced it had partnered with Jurata Thin Film, Inc. to advance development of thermostable under-the-tongue vaccine films as a needle-free vaccine delivery platform.
On December 5, 2023, CEPI confirmed that it will provide up to an initial $1.2 million to support Jurata's proprietary innovative formulation platform, which, if shown to be successful, could help expand access to vaccines in underserved regions and advance the global response to future emerging infectious disease outbreaks.
CEPI's initial funding will support optimizing the composition and process of creating thin films and preclinical studies.
Under the agreement with CEPI, Jurata will create vaccine films to remain stable at 2-8 degrees, 25 degrees, and 40 degrees.
Jurata will optimise the composition of the films by testing various buffers, pH, stabilizers, sugars, salts, and different drying parameters and assessing how this affects vaccine stability and delivery.
Jurata aims to improve vaccine accessibility by stabilizing the 3D structure of mRNA-containing lipid nanoparticle vaccine materials, provided by Quantoom Biosciences, part of Univercells, into a thin thermostable film, thereby removing frozen storage needs.
The vaccine films are also lightweight and compact, simplifying the transportation process and potentially allowing for more doses to be shipped at any one time compared to current needle-and-syringe distribution.
Dr. Irnela Bajrovic, Chief Scientific Officer, Jurata, commented in a press release, "Our stabilising formulations have the potential to facilitate global access to mRNA vaccines, and our thin film delivery platform could make vaccine administration far easier than needle-and-syringe injections."
"We are grateful to CEPI for supporting our innovative technology and look forward to working with Quantoom to show the breadth of mRNA vaccines our technology can stabilize and deliver."
This is the fourth partner to be announced as part of CEPI's Call for Proposals for thermostable vaccine manufacturing innovations, announced in January 2022.
Thermostable vaccines are also identified as a preferred vaccine characteristic by the World Health Organization.

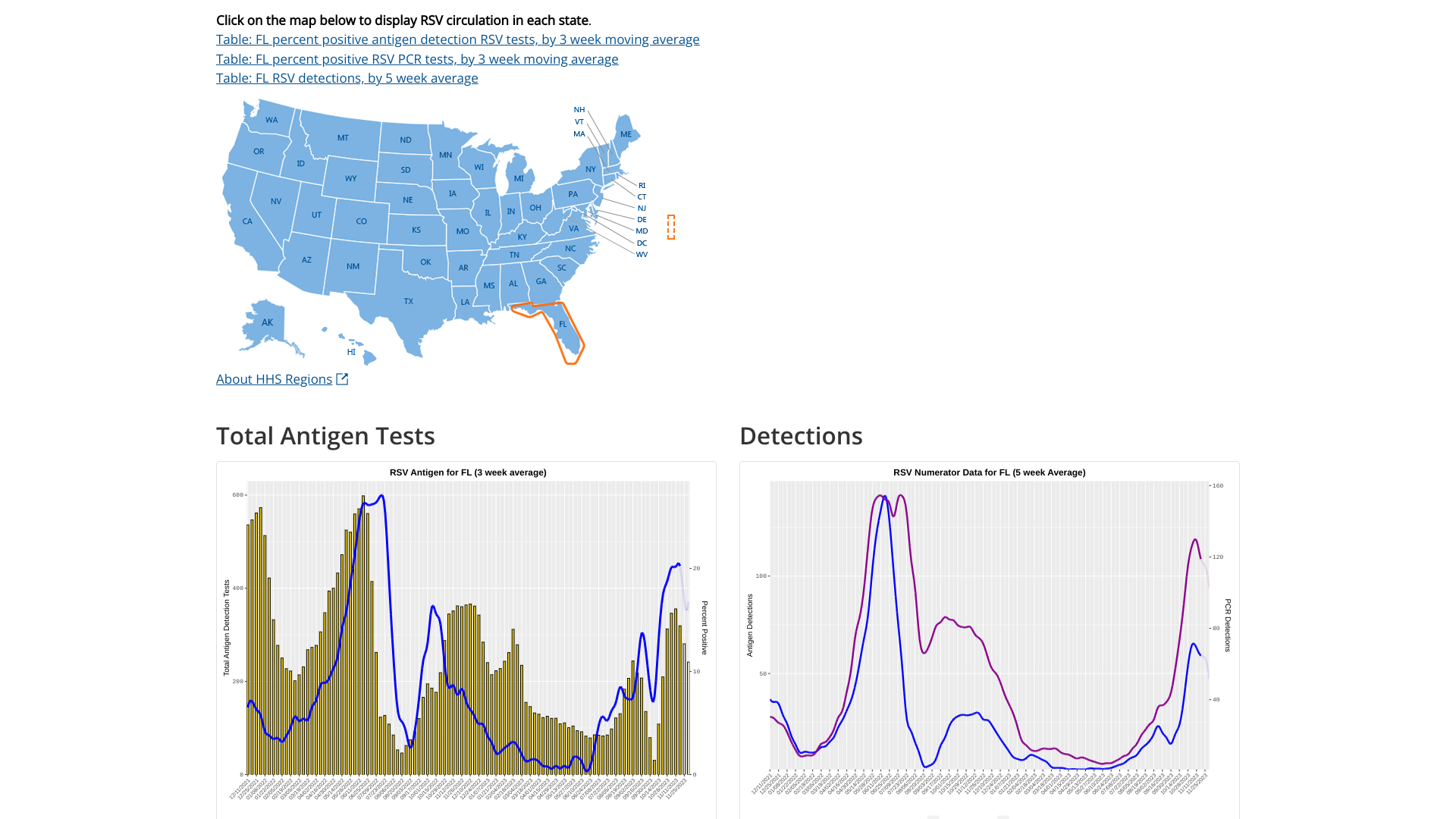
Since most respiratory syncytial virus (RSV) activity in the United States is initially recorded in the state of Florida, watching for the bending of the curve offers insights regarding future cases.
As of December 8, 2023, the U.S. Centers for Disease Control and Prevention (CDC) updated its RSV detection graphs to display the 5-week moving average. RSV infections typically occur during late fall, winter, and early spring.
There are variations in the timing of RSV outbreaks between regions and communities in the same region.
For Florida, the two charts indicate RSV's peak was in late November 2023.
Separately, the Florida Department of Health reported that as of December 2, 2023, RSV activity was decreasing in hospital admissions, positivity tests, and ER visits.
But, there was an RSV outbreak in Pinellas County.
From a prevention perspective, both RSV vaccines are available at most clinics and pharmacies in Florida. However, the CDC recently confirmed the percentage of seniors receiving an RSV vaccine was just 15.9%.
Unfortunately, the RSV antibody passive immunization for infants, Beyfortus™, remains in limited supply.
Virtually all children get an RSV infection by the time they are two years old. Most of the time, RSV will cause a mild, cold-like illness. RSV antibodies can help protect children from severe disease from an RSV infection.
RSV can be dangerous for infants and young children. Each year, thousands are hospitalized due to RSV infection, says the CDC.
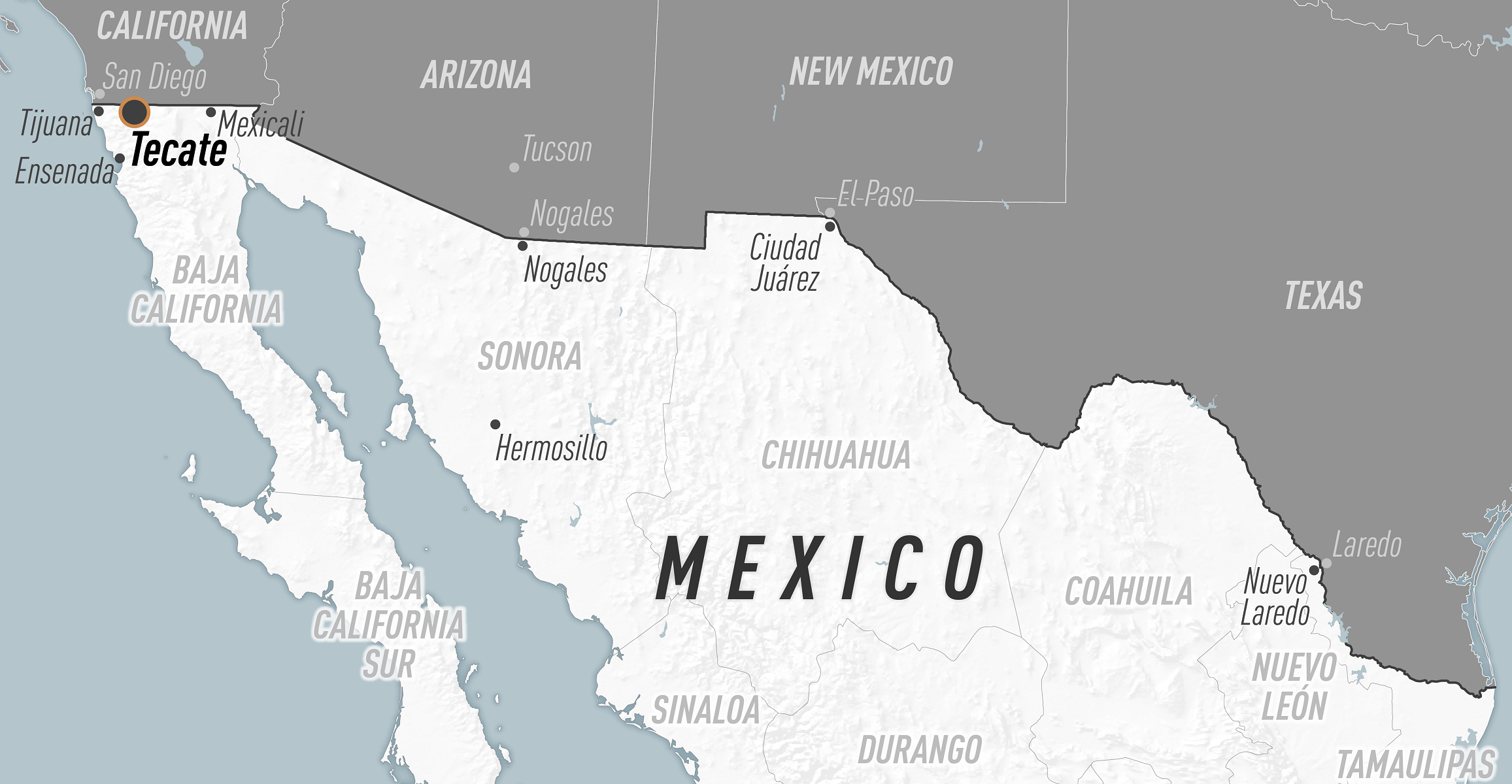
The Centers for Disease Control and Prevention (CDC) today issued a Health Alert Network Health Advisory confirming an outbreak of Rocky Mountain spotted fever (RMSF) among people in the United States with recent travel to or residence in the city of Tecate, state of Baja California, Mexico.
As of December 8, 2023, five patients have been confirmed with RMSF, and 3 have died since late July 2023.
Each patient had travel to or residence in Tecate within two weeks of illness onset.
RMSF is endemic in multiple border states in northern Mexico, including but not limited to Baja California, Sonora, Chihuahua, Coahuila, and Nuevo León.
RMSF is a severe, rapidly progressive, and often deadly disease transmitted by infected ticks, although many patients do not recall being bitten by a tick.
Furthermore, the CDC says dogs can carry infected ticks that can bite humans.
This disease progresses rapidly, with about half of all people who die from this disease succumbing within eight days of illness onset.
The CDC endorses Doxycycline as the treatment of choice for RMSF patients of all ages.
Doxycycline is offered in various pharmacies under different brand names.
As of December 9, 2023, there are no RMSF vaccines approved by the U.S. FDA.
The U.S. Centers for Disease Control and Prevention (CDC) today announced very few seniors have received an approved vaccine that protects against Respiratory syncytial virus (RSV).
This common respiratory virus can become severe and require hospitalization.
As of December 8, 2023, the CDC confirmed the percentage of adults 60+ reporting receiving an RSV vaccine was 15.9%.
This CDC data indicates an uptake gap compared to a recent survey.
A recent poll by the University of Michigan's Institute for Healthcare Policy and Innovation revealed that 21% of seniors want an RSV vaccination in 2023.
Unlike previous RSV seasons, there are two RSV vaccines endorsed by government agencies, which are as follows:
AREXVY™ RSV vaccine is approved for adults aged 60 and above.
ABRYSVO™ RSVpreF RSV bivalent vaccine is from Pfizer Inc.
The U.S. Food and Drug Administration, the U.K.'s Medicines and Healthcare products Regulatory Agency, and the European Commission recommend adults 60 years and older and pregnant women receive a single dose of an RSV vaccine based on discussions with healthcare providers and under certain conditions.
Dr. Mandy Cohen, Director of the CDC, commented during a media interview on December 8, 2023, "What we know is that several viruses are circulating, but we also have tools to protect ourselves."
"And we're hoping more can do that before the Christmas holiday."
In the U.S., RSV vaccines are generally available at clinics and pharmacies.

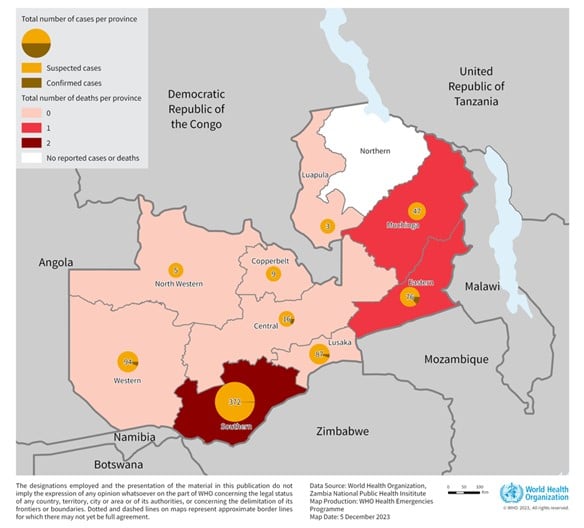
The ongoing anthrax outbreak in the Republic of Zambia has recently become a multi-country concern.
The World Health Organization (WHO today announced that as of November 20, 2023, 684 suspected human cases, including four deaths, have been reported in 2023.
This unprecedented anthrax outbreak marks the first significant occurrence spanning nine out of 10 country provinces. The last large-scale outbreak reported in Zambia occurred in 2011, with 511 suspected cases, wrote the WHO on December 8, 2023.
The risk of the outbreak spreading within Zambia is assessed to be high, and at the regional level is also considered high due to the frequent movement of both animals and people between Zambia and its neighboring countries, such as Angola, Botswana, the Democratic Republic of the Congo, Malawi, Mozambique, Namibia, Tanzania, Uganda, and Zimbabwe.
International travelers to anthrax-endemic countries should be aware of the current health risk, says the WHO. In 2021, about 554,000 tourists visited Zambia, formerly known as Northern Rhodesia, reported WorldData.
However, the WHO advises against implementing travel or trade restrictions with Zambia based on the current information on this event.
In addition to anthrax, the U.S. CDC has included Zambia in Travel Health Advisories in 2023 regarding measles and polio.
Humans usually acquire the infection after exposure to infected animals, carcasses, or animal products. More than 95% of human anthrax cases take the cutaneous form and result from handling infected carcasses or hides, hair, meat, or bones from such carcasses.
Anthrax is a zoonotic disease caused by Bacillus anthracis that typically affects ruminants (cows, sheep, and goats). The bacteria produce highly potent toxins responsible for the symptoms, causing a high lethality rate in the pulmonary form.
Humans can develop the disease from infected animals or through contaminated animal products. Hospitalization is required for all human cases identified. Vaccines are available for livestock.
However, humans have access to a limited supply.
From a prevention vaccination perspective, Emergent BioSolutions Inc. recently announced that the U.S. Biomedical Advanced Research and Development Authority awarded a $75 million contract option to acquire the newly licensed anthrax vaccine CYFENDUS™.
Deliveries of the two-dose vaccine are expected to begin in the U.S. in 2023 and be completed in the late first quarter of 2024. These vaccines are unavailable to the general public.
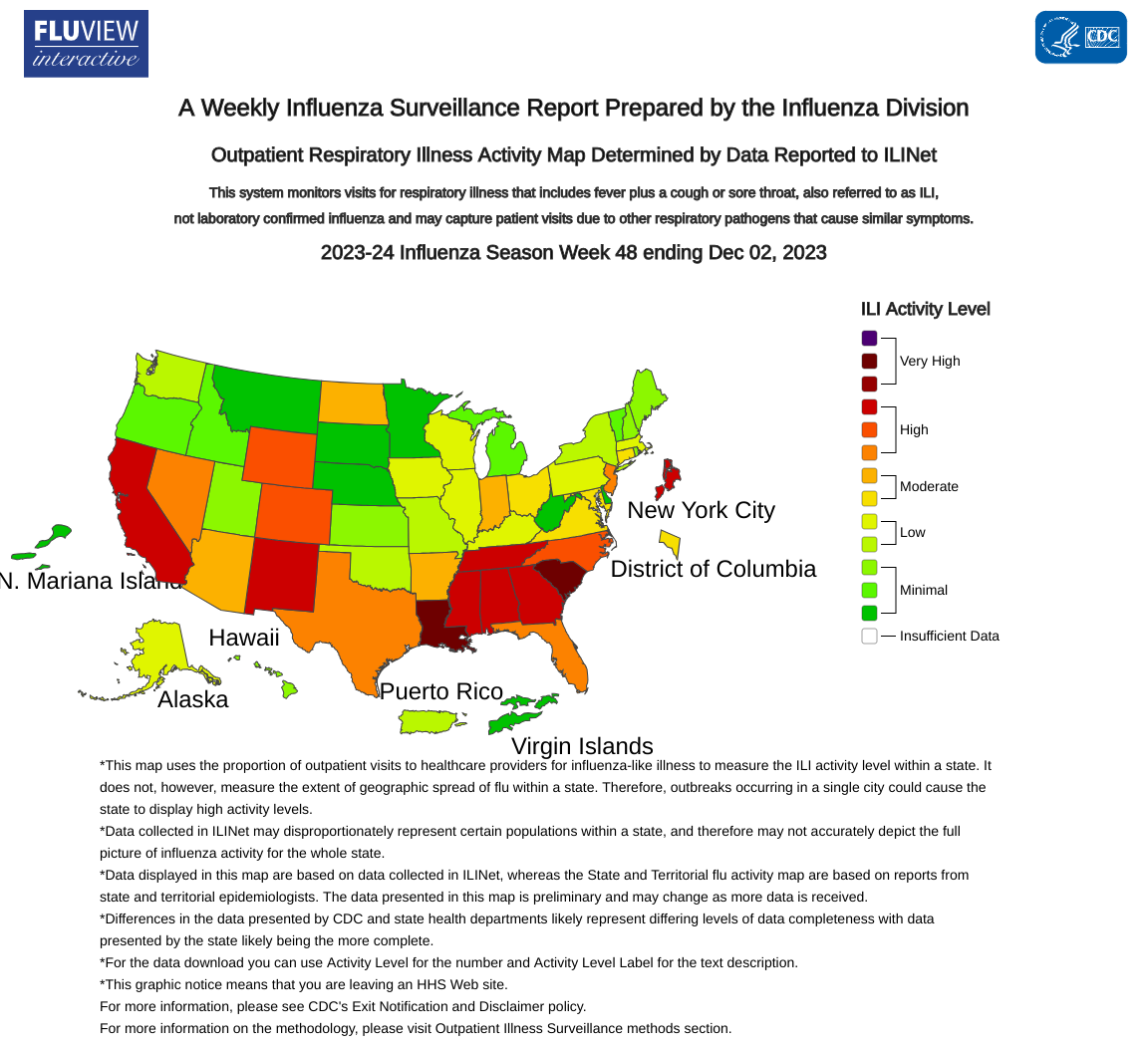
The U.S. Centers for Disease Control and Prevention (CDC) today announced seasonal flu activity continues to increase in most parts of the country, with the southeast and south-central areas of the country reporting the highest levels of activity.
As of December 8, 2023, outpatient respiratory illness is above baseline nationally for the fifth week and is at or above baseline in all 10 HHS Regions.
Furthermore, the National Center for Health Statistics (NCHS) Mortality Surveillance data available on December 7, 2023, indicates that 0.2% of the deaths (48) that occurred during Week #48 were due to influenza.
Last week, in #47, there were 61 flu-related deaths and 2,373 pneumonia-related deaths.
Specifically, there were four influenza-associated pediatric deaths reported during Week #48, bringing the 2023-2024 season total to 12 pediatric deaths.
Last flu season, about 182 children died from influenza.
From a prevention perspective, the CDC recently confirmed that over 151 million flu shots had already been distributed in the U.S.
These influenza vaccines are generally available at clinics and pharmacies,
The journal Nature Medicine recently asked researchers to name their top clinical trial picks for 2024, from base editing and a vaccine against human immunodeficiency virus (HIV).
On December 7, 2023, Carrie Arnold and Paul Webster wrote ...with so many rollercoaster years since the start of the pandemic, it is impossible to predict exactly what the biomedical world will deliver in 2024.
Experts identified which trials will likely have an outsized impact on medicine in 2024.
One expert, Carey Hwang, a senior vice president and head of clinical research at Vir Biotechnologycommented, highlighted VIR-1388, a cytomegalovirus (CMV) vector vaccine that induces strong, unique, and sustained T cell responses that can potentially prevent the acquisition of HIV.
The HIV Vaccine Trials Network is conducting a clinical trial at ten sites in the U.S. and two sites in South Africa, with support from the U.S. National Institute of Allergy and Infectious Diseases and the Bill & Melinda Gates Foundation.
From a public health perspective, having a vaccine against HIV would have a tremendous impact, commented Hwang.
As of December 8, 2023, the U.S. FDA has not approved any HIV vaccine candidates.
The U.S. Centers for Disease Control and Prevention (CDC) today announced a Health Alert Network (HAN) Health Advisory about the occurrence, geographic spread, and sexually associated human-to-human transmission of Clade I Monkeypox virus (MPXV) in the Democratic Republic of the Congo (DRC).
Since January 2023, the DRC has reported 12,569 suspected mpox cases and 581 related deaths from 22 regions.
The new HAN says cases of Clade I MPXV have not been reported in the United States as of December 7, 2023. The global outbreak of Clade II MPXV was initially reported in May 2022.
However, clinicians should be aware of the possibility of Clade I MPXV in travelers who have been in DRC.
Third-party data indicate that the number of tourists arriving in the DRC was about 460,880 in 2021.
The CDC recently issued a Travel Health Notice (Level 2 - Practice Enhanced Precautions) for people traveling to DRC. Furthermore, there are no direct commercial passenger flights from DRC to the U.S. as of December 2023.
U.S. FDA-approved vaccines (JYNNEOS, ACAM2000) are expected to be effective for both Clade I and II MPXV infections.
Vaccination or prior MPXV infection should provide antibodies that will provide cross-protection to other orthopoxviruses, including Clade I MPXV, says the CDC.
However, clinical verification is under review.
The CDC recommends clinicians encourage vaccination for eligible patients.
Eligible patients who have only received one dose of Bavarian Nordic JYNNEOS® (MVA-BN®, IMVAMUNE®) vaccine, which is based on a live, attenuated vaccinia virus, should receive the second dose as soon as possible, regardless of the time that has elapsed since the first dose.
Mpox vaccines have limited availability in the U.S.
Furthermore, clinicians should notify their state health department if they have a patient with mpox-like symptoms and should submit lesion specimens for clade-specific testing for these patients.